Keywords
aminoisoxazoles
Antibacterial activity
chemoselectivity
isoxazoles
nitration
nitroisoxazoles
quantum chemical calculations
trifluoroacetyl nitrate
Abstract
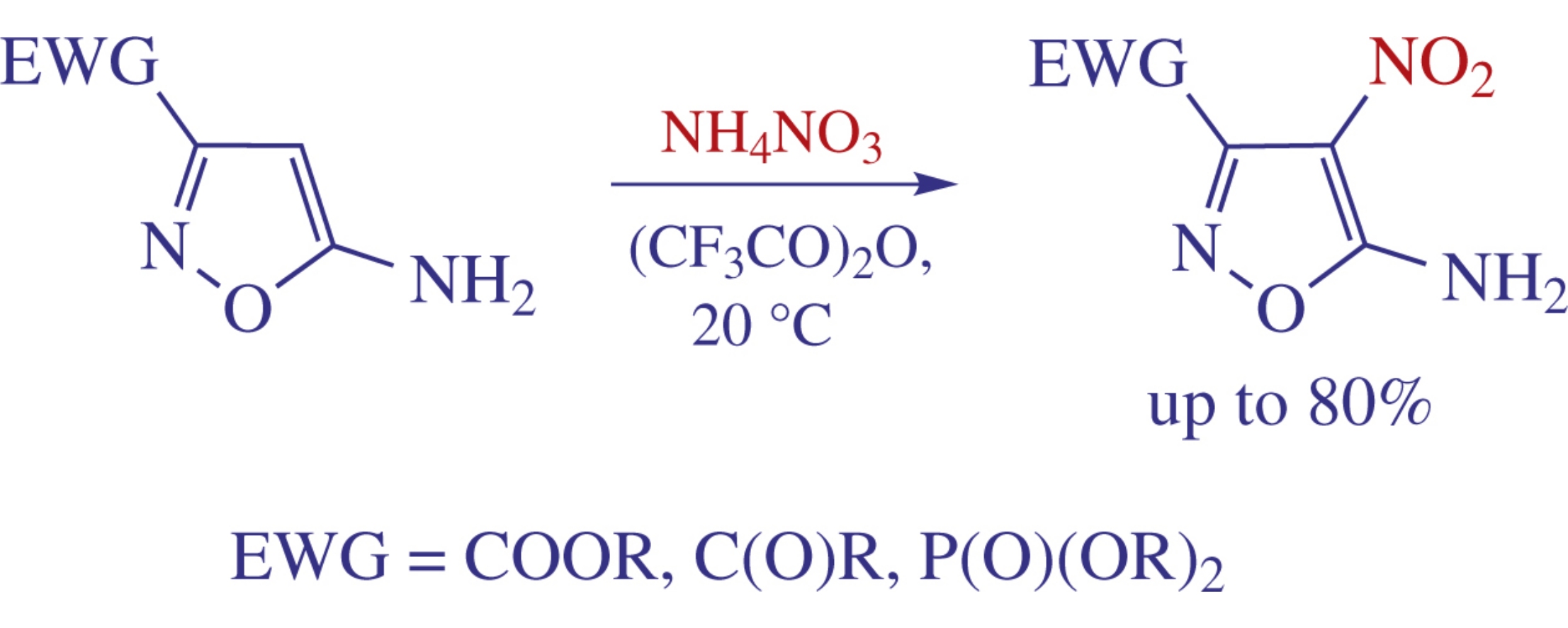
5-Aminoisoxazoles bearing 3-positioned electron-withdrawing group react chemoselectively with trifluoroacetyl nitrate (generated from ammonium nitrate and trifluoroacetic anhydride) to give the corresponding 4-nitro derivatives in yields up to 80%. The results were rationalized by computation studies. Several 5-amino-4-nitroisoxazole-3-carboxilic acid derivatives revealed moderate antibacterial activity.
References
1.

A. Barmade M., R. Murumkar P., Kumar Sharma M., Ram Yadav M.
Current Topics in Medicinal Chemistry,
2016
2.

Uto Y.
Current Pharmaceutical Design,
2016
3.

Sysak A., Obmińska-Mrukowicz B.
European Journal of Medicinal Chemistry,
2017
4.

Zhu J., Mo J., Lin H., Chen Y., Sun H.
Bioorganic and Medicinal Chemistry,
2018
5.
Tsyganov D.V., Semenova M.N., Konyushkin L.D., Ushkarov V.I., Raihstat M.M., Semenov V.V.
Mendeleev Communications,
2019
6.
10.1016/j.mencom.2020.07.027_sbref0010a
Doi
2015
7.
10.1016/j.mencom.2020.07.027_sbref0010b
Farrington
Clinical Pharmacology,
2012
8.

Leppik I.E.
Seizure : the journal of the British Epilepsy Association,
2004
9.

Leucht S., Cipriani A., Spineli L., Mavridis D., Örey D., Richter F., Samara M., Barbui C., Engel R.R., Geddes J.R., Kissling W., Stapf M.P., Lässig B., Salanti G., Davis J.M., et. al.
The Lancet,
2013
10.

Larsen J.K., Rafaelsen O.J.
Acta Psychiatrica Scandinavica,
1980
11.

Liu Y., Hsiao H., Wang J.C., Liu Y., Wu S.
European Journal of Pharmacology,
2019
12.

Sanders S., Harisdangkul V.
American Journal of the Medical Sciences,
2002
13.

Fuse S., Morita T., Nakamura H.
Synthesis,
2017
14.

Volkova Y.A., Averina E.B., Vasilenko D.A., Sedenkova K.N., Grishin Y.K., Bruheim P., Kuznetsova T.S., Zefirov N.S.
Journal of Organic Chemistry,
2019
15.

Averina E.B., Samoilichenko Y.V., Volkova Y.A., Grishin Y.K., Rybakov V.B., Kutateladze A.G., Elyashberg M.E., Kuznetsova T.S., Zefirov N.S.
Tetrahedron Letters,
2012
16.
![Chemoselective Reduction of Functionalized 5-Nitroisoxazoles: Synthesis of 5-Amino- and 5-[Hydroxy(tetrahydrofuran-2-yl)amino]isoxazoles](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Averina E., Vasilenko D., Samoilichenko Y., Grishin Y., Rybakov V., Kuznetsova T., Zefirov N.
Synthesis,
2014
17.

Averina E., Vasilenko D., Sedenkova K., Kuznetsova T.
Synthesis,
2019
18.

Averina E.B., Vasilenko D.A., Sadovnikov K.S., Sedenkova K.N., Kurova A.V., Grishin Y.K., Kuznetsova T.S., Rybakov V.B., Volkova Y.A.
Synthesis,
2020
19.

Bartoli S., Jensen K.B., Kilburn J.D.
Journal of Organic Chemistry,
2003