Keywords
Barton–Zard reaction
isomerization
lamellarins
propenylbenzenes
pseudonitrosites
pyrroles
Abstract
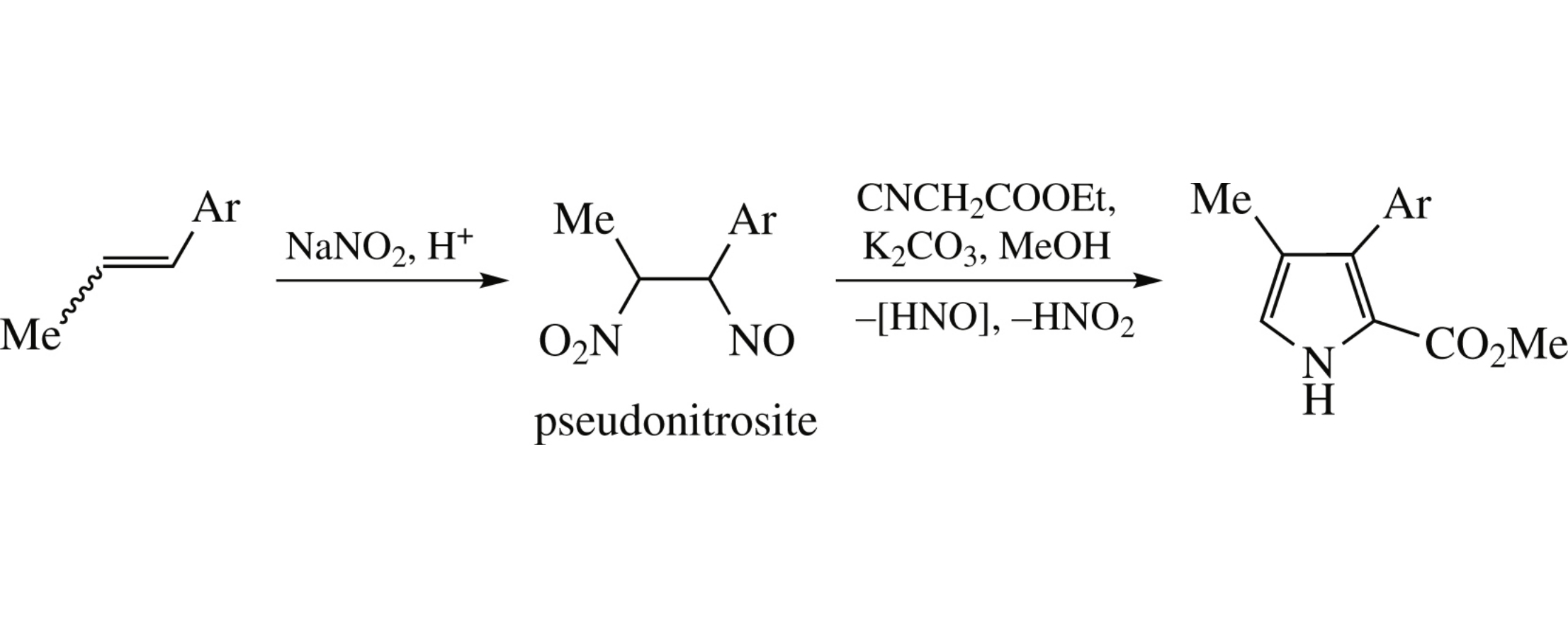
3-Aryl-4-methylpyrrole-2-carboxylates were prepared via the Barton–Zard reaction using pseudonitrosites as a source of the corresponding nitroalkenes. The starting pseudonitrosites were, in turn, obtained via addition of N2O3 to propenylbenzenes available from the natural plant essential oils.
References
1.

Barton D.H., Zard S.Z.
Journal of the Chemical Society Chemical Communications,
1985
2.

H.R. Barton D., Kervagoret J., Zard S.Z.
Tetrahedron,
1990
3.

4.

He X., Zhao H., Song X., Jiang B., Du W., Chen Y.
ACS Catalysis,
2019
5.

Zheng S., Wang Q., Zhu J.
Angewandte Chemie - International Edition,
2019
6.

Kaufman N.E., Meng Q., Griffin K.E., Singh S.S., Dahal A., Zhou Z., Fronczek F.R., Mathis J.M., Jois S.D., Vicente M.G.
Journal of Medicinal Chemistry,
2019
7.

Johnson R.E., de Rond T., Lindsay V.N., Keasling J.D., Sarpong R.
Organic Letters,
2015
8.

Bullington J.L., Wolff R.R., Jackson P.F.
Journal of Organic Chemistry,
2002
9.

Myśliwiec D., Donnio B., Chmielewski P.J., Heinrich B., Stępień M.
Journal of the American Chemical Society,
2012
10.

Silyanova E.A., Samet A.V., Semenov V.V.
Russian Chemical Bulletin,
2018
11.

Samet A.V., Sil’yanova E.A., Ushkarov V.I., Semenova M.N., Semenov V.V.
Russian Chemical Bulletin,
2018
12.

Semenova M.N., Demchuk D.V., Tsyganov D.V., Chernysheva N.B., Samet A.V., Silyanova E.A., Kislyi V.P., Maksimenko A.S., Varakutin A.E., Konyushkin L.D., Raihstat M.M., Kiselyov A.S., Semenov V.V.
ACS Combinatorial Science,
2018
13.

Silyanova E.A., Samet A.V., Salamandra L.K., Khrustalev V.N., Semenov V.V.
European Journal of Organic Chemistry,
2020
14.
10.1016/j.mencom.2020.07.026_sbref0020a
Fukuda
Alkaloids,
2020
15.
10.1016/j.mencom.2020.07.026_sbref0020b
Chittchang
2018
16.

Imbri D., Tauber J., Opatz T.
Marine Drugs,
2014
17.

Iwao M., Fukuda T., Ishibashi F.
Heterocycles,
2011
18.

Ohta T., Fukuda T., Ishibashi F., Iwao M.
Journal of Organic Chemistry,
2009
19.

Tangdenpaisal K., Worayuthakarn R., Karnkla S., Ploypradith P., Intachote P., Sengsai S., Saimanee B., Ruchirawat S., Chittchang M.
Chemistry - An Asian Journal,
2015
20.

Angeli A.
Berichte der deutschen chemischen Gesellschaft,
1891
21.
22.

Demjanow N.J.
Berichte der deutschen chemischen Gesellschaft,
1907
23.

Semenov V.V., Rusak V.V., Chartov E.M., Zaretskii M.I., Konyushkin L.D., Firgang S.I., Chizhov A.O., Elkin V.V., Latin N.N., Bonashek V.M., Stas’eva O.N.
Russian Chemical Bulletin,
2007
24.

Semenov V.V., Kiselyov A.S., Titov I.Y., Sagamanova I.K., Ikizalp N.N., Chernysheva N.B., Tsyganov D.V., Konyushkin L.D., Firgang S.I., Semenov R.V., Karmanova I.B., Raihstat M.M., Semenova M.N.
Journal of Natural Products,
2010
25.

Titov I.Y., Sagamanova I.K., Gritsenko R.T., Karmanova I.B., Atamanenko O.P., Semenova M.N., Semenov V.V.
Bioorganic and Medicinal Chemistry Letters,
2011
26.

Samet A.V., Shevchenko O.G., Rusak V.V., Chartov E.M., Myshlyavtsev A.B., Rusanov D.A., Semenova M.N., Semenov V.V.
Journal of Natural Products,
2019
27.

Stepanov A.V., Veselovsky V.V.
Russian Chemical Reviews,
2003
28.

Riebsomer J.L.
Chemical Reviews,
1945
29.
10.1016/j.mencom.2020.07.026_sbref0035c
Aitken
Sci. Synth.,
2010
30.

Kametani T., Sugahara H., Yagi H.
Journal of the Chemical Society C Organic,
1966
31.

Kurihara Y., Yamagishi K.
Bulletin of the Chemical Society of Japan,
1965
32.
10.1016/j.mencom.2020.07.026_sbref0040c
Savitskii
Zh. Obsch. Khim.,
1940
33.

Fumoto Y., Uno H., Ito S., Tsugumi Y., Sasaki M., Kitawaki Y., Ono N.
Journal of the Chemical Society Perkin Transactions 1,
2000
34.

Schramm M., Pham C., Park M., Pham J., Martin S.
Synthesis,
2013
35.

Kancharla P., Reynolds K.A.
Tetrahedron,
2013
36.

Abeywardana S., Cantu A.L., Nedic T., Schramm M.P.
Tetrahedron Letters,
2018
37.

Sy W., By A.W.
Tetrahedron Letters,
1985
38.

Nichols D.E., Snyder S.E., Oberlender R., Johnson M.P., Huang X.
Journal of Medicinal Chemistry,
1991
39.
10.1016/j.mencom.2020.07.026_sbref0055a
Schmidt
Ber.,
1922
40.

Shulgin A.T.
Journal of Medicinal Chemistry,
1966
41.

Gooden D., Chakrapani H., Toone E.
Current Topics in Medicinal Chemistry,
2005
42.

Chiu K.W., Savage P.D., Wilkinson G., Williams D.J.
Polyhedron,
1985
43.

Shaabani A., Ameri M., Bijanzadeh H.R.
Journal of Chemical Research,
1998
44.

Shaabani A., Bijanzadeh H.R., Karimi A.R., Teimouri M.B., Soleimani K.
Canadian Journal of Chemistry,
2008