Keywords
cysteine
Griess method
nitrite ion
organic nitrate
thionitrate
Abstract
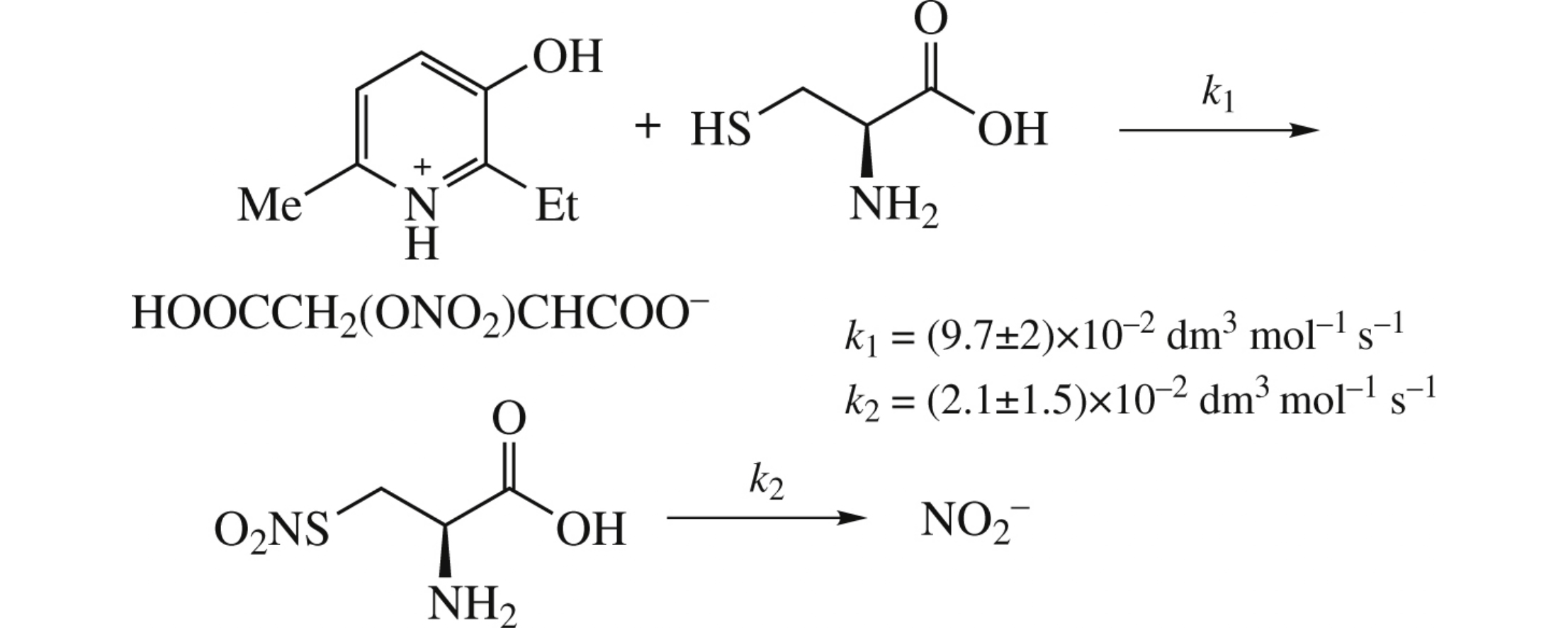
The nitrite-generating activity of 2-ethyl-3-hydroxy-6-methylpyridinium 2-nitroxysuccinate as a promising NO-donor has been investigated in reactions with various reducing agents. Reduction of the NO-donor with cysteine was analyzed using a kinetic modeling method. The calculated rate constants satisfactorily describe the experimental data, thereby confirming the proposed reaction mechanism.
References
1.
10.1016/j.mencom.2020.07.025_bib0005
Nitric Oxide: Biology and Pathobiology,
2000
2.
10.1016/j.mencom.2020.07.025_bib0010
Nitric Oxide Donors for Pharmaceutical and Biological Applications,
2005
3.

Kuropteva Z.V., Baider L.M., Nagler L.G., Bogatyrenko T.N., Belaia O.L.
Russian Chemical Bulletin,
2019
4.
B. S. Fedorov, M.A. Fadeev, V.N. Varfolomeev, M.I. Retskij, G.N. Bliznetsova and E. V. Neborak, RU Patent 2394815 C2, 2010.
5.

Antioxidant Properties of a Pharmaceutical Substance Hypocard, a Potential Drug for Ischemic Disease
Neganova M.E., Klochkov S.G., Shevtsova E.F., Bogatyrenko T.N., Mishchenko D.V.
Bulletin of Experimental Biology and Medicine,
2018
6.

Pokidova O.V., Batova E.V., Sadkov A.P., Eremeev A.B., Fedorov B.S., Kotelnikov A.I.
Doklady Chemistry,
2019
7.

Tat’yanenko L.V., Dobrokhotova O.V., Varfolomeev V.N., Fadeev M.A., Fedorov B.S., Shtol’ko V.N., Mishchenko D.V.
Pharmaceutical Chemistry Journal,
2014
8.

Page N.A., Fung H.
Journal of Pharmaceutical Sciences,
2013
9.

Tsou P., Page N.A., Lee S.G., Fung S.M., Keung W.M., Fung H.
AAPS Journal,
2011
10.

Feelisch M., Noack E.
European Journal of Pharmacology,
1987
11.

Saeho C., Ho-leung F.
Biochemical Pharmacology,
1991
12.

Sano T., Shimada K., Aoki Y., Kawashima T., Sase S., Goto K.
Molecules,
2016
13.

Feelisch M.
Journal of Cardiovascular Pharmacology,
2008
14.

R. J. Thatcher G.
Chemical Society Reviews,
1998
15.

Gladwin M.T., Schechter A.N., Kim-Shapiro D.B., Patel R.P., Hogg N., Shiva S., Cannon R.O., Kelm M., Wink D.A., Espey M.G., Oldfield E.H., Pluta R.M., Freeman B.A., Lancaster J.R., Feelisch M., et. al.
Nature Chemical Biology,
2005
16.

Huang Z.
Journal of Clinical Investigation,
2005
17.
10.1016/j.mencom.2020.07.025_bib0085
Granik
Oksid azota (NO). Novyi put’ k poisku lekarstv [Nitric Oxide (NO). A New Way to Drug Search],
2004
18.
10.1016/j.mencom.2020.07.025_bib0090
Musil
Biochemistry in Schematic Perspective, Avicenum,
1980