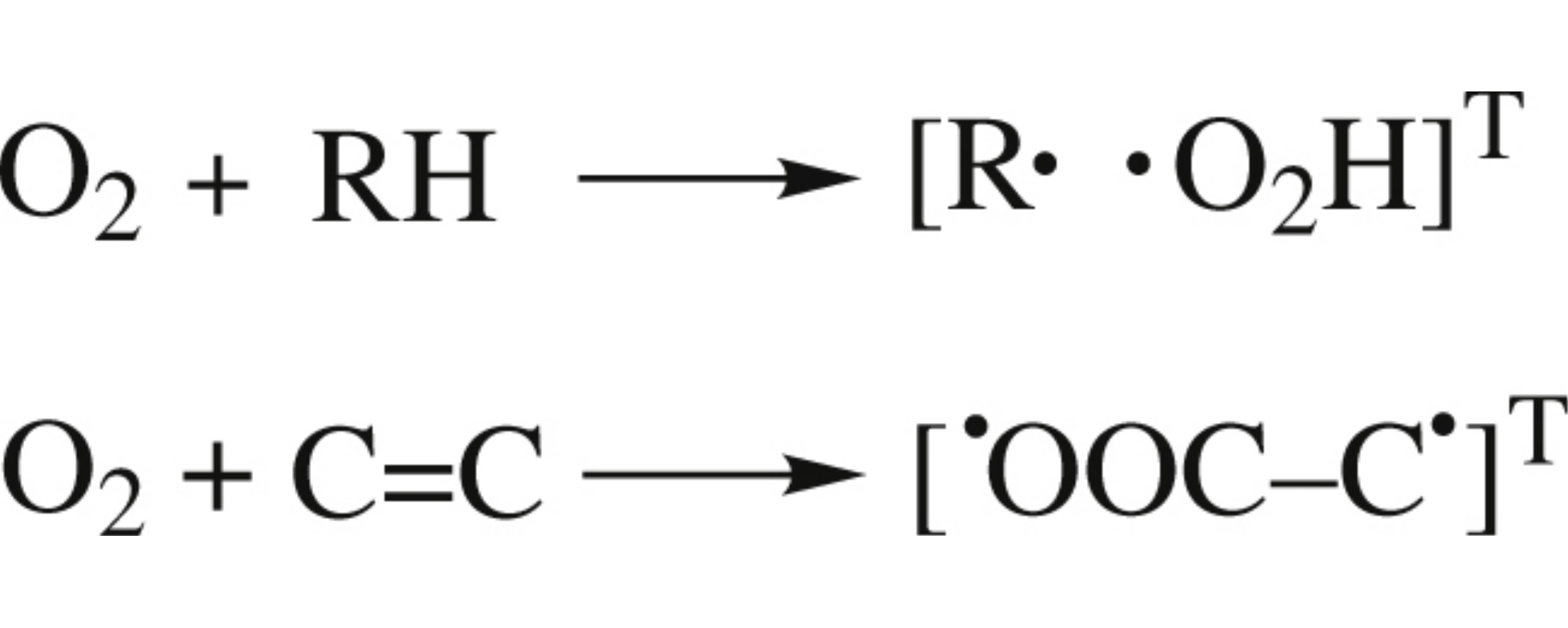Keywords
chain oxidation mechanism
hydrogen atom abstraction
magnetic field effect
oxygen
triplet biradical
Zeeman interaction
Abstract
Oxidation of olefins is initiated by addition of oxygen at the π-bond with formation of triplet biradical; therefore, dioxetane formation becomes dominant due to the Zeeman interaction stimulating triplet–singlet conversion through the T–S transition, with the contribution of the radical initiation being lower. In the case of saturated compounds, the Zeeman interaction results in the absence of the magnetic field effect on the rate of radical formation since the chain initiation is ruled by the hydrogen atom abstraction.
References
1.
10.1016/j.mencom.2020.07.009_bib0005
Oxidation and Antioxidants in Organic Chemistry and Biology,
2005
2.
10.1016/j.mencom.2020.07.009_bib0010
Buchachenko
Magneto-Biology and Medicine,
2014
3.
10.1016/j.mencom.2020.07.009_bib0015
Niki
Encyclopedia of Radicals in Chemistry, Biology and Materials,
2012
4.

Yin H., Xu L., Porter N.A.
Chemical Reviews,
2011
5.

Pratt D.A., Tallman K.A., Porter N.A.
Accounts of Chemical Research,
2011
6.

Porter N.A.
Journal of Organic Chemistry,
2013
7.
10.1016/j.mencom.2020.07.009_bib0035
Hayashi
Introduction to Dynamic Spin Chemistry: Magnetic Field Effects on Chemical and Biochemical Reactions,
2004
8.

Sampson C., Keens R.H., Kattnig D.R.
Physical Chemistry Chemical Physics,
2019
9.

Lukzen N.N., Ivanov K.L., Sadovsky V.M., Sagdeev R.Z.
Journal of Chemical Physics,
2020
10.
Pliss E.M., Grobov A.M., Kuzaev A.K., Buchachenko A.L.
Mendeleev Communications,
2017
11.

Pliss E.M., Grobov A.M., Kuzaev A.K., Buchachenko A.L.
Journal of Physical Organic Chemistry,
2018
12.

Sirick A., Lednev S., Moskalenko I., Machtin V., Pliss E.
Reaction Kinetics, Mechanisms and Catalysis,
2015