Keywords
CDK7 inhibitors
cyclin-dependent kinase 7
free energy perturbation
hepatotoxicity
molecular dynamics
non-hepatotoxic scaffold
PHA-793887
relative binding free energy
Abstract
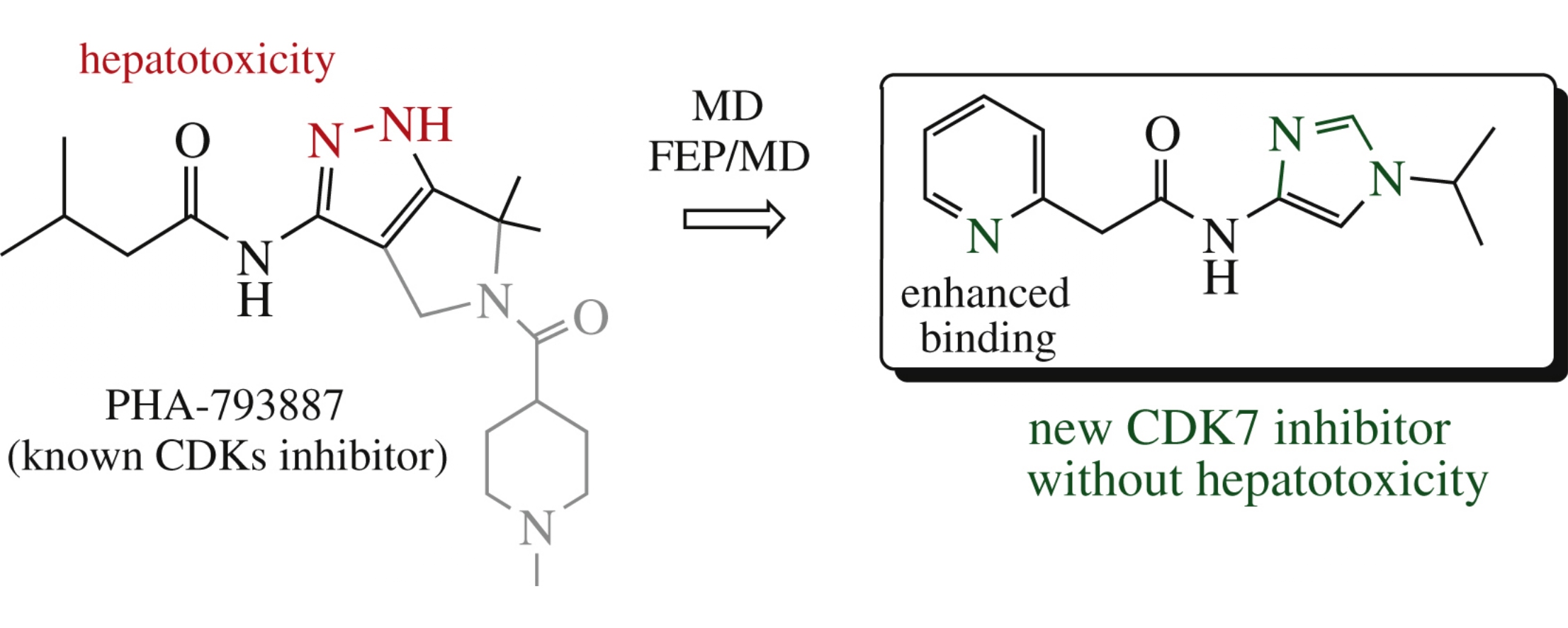
Although CDK7 inhibitors are considered to be potential anticancer drugs, all inhibitors developed so far have significant disadvantages preventing their further use. We have developed a new CDK7 inhibitor scaffold lacking hepatotoxicity using molecular dynamics (MD) and free energy perturbation (FEP/MD) methods, and were able to double its binding affinity after additional research. The combination of MD and FEP/MD methods was shown to be a valuable instrument for the development of novel and potent CDK7 inhibitors for anticancer therapy.
References
1.

Fisher R.P.
Transcription,
2018
2.

Sun B., Mason S., Wilson R.C., Hazard S.E., Wang Y., Fang R., Wang Q., Yeh E.S., Yang M., Roberts T.M., Zhao J.J., Wang Q.
Oncogene,
2019
3.

Sánchez-Martínez C., Lallena M.J., Sanfeliciano S.G., de Dios A.
Bioorganic and Medicinal Chemistry Letters,
2019
4.

Polshakov V.I., Batuev E.A., Mantsyzov A.B.
Russian Chemical Reviews,
2019
5.
Grigorenko B.L., Kots E.D., Krylov A.I., Nemukhin A.V.
Mendeleev Communications,
2019
6.
Lavrov M.I., Karlov D.S., Palyulin V.A., Grigoriev V.V., Zamoyski V.L., Brkich G.E., Pyatigorskaya N.V., Zapolskiy M.E.
Mendeleev Communications,
2018
7.

Magdesieva T.V., Levitskiy O.A.
Russian Chemical Reviews,
2018
8.
Kuznetsov N.Y., Malishev V.I., Medvedev M.G., Bubnov Y.N.
Mendeleev Communications,
2019
9.

Ustynyuk L.Y., Tikhonov A.N.
Journal of Organometallic Chemistry,
2018
10.
Marjewski A.A., Medvedev M.G., Gerasimov I.S., Panova M.V., Perdew J.P., Lyssenko K.A., Dmitrienko A.O.
Mendeleev Communications,
2018
11.

Stroganov O.V., Novikov F.N., Stroylov V.S., Kulkov V., Chilov G.G.
Journal of Chemical Information and Modeling,
2008
12.

Wang L., Wu Y., Deng Y., Kim B., Pierce L., Krilov G., Lupyan D., Robinson S., Dahlgren M.K., Greenwood J., Romero D.L., Masse C., Knight J.L., Steinbrecher T., Beuming T., et. al.
Journal of the American Chemical Society,
2015
13.
![Optimization of 6,6-dimethyl pyrrolo[3,4-c]pyrazoles: Identification of PHA-793887, a potent CDK inhibitor suitable for intravenous dosing](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Brasca M.G., Albanese C., Alzani R., Amici R., Avanzi N., Ballinari D., Bischoff J., Borghi D., Casale E., Croci V., Fiorentini F., Isacchi A., Mercurio C., Nesi M., Orsini P., et. al.
Bioorganic and Medicinal Chemistry,
2010
14.

Massard C., Soria J., Anthoney D.A., Proctor A., Scaburri A., Pacciarini M.A., Laffranchi B., Pellizzoni C., Kroemer G., Armand J., Balheda R., Twelves C.J.
Cell Cycle,
2011
15.

Lu Y., Gong P., Cederbaum A.I.
Toxicology,
2008
16.

Wang C., Jin H., Gao D., Wang L., Evers B., Xue Z., Jin G., Lieftink C., Beijersbergen R.L., Qin W., Bernards R.
Cell Research,
2018
17.

Schönherr H., Cernak T.
Angewandte Chemie - International Edition,
2013
18.
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD, 2012.