Keywords
1,3-benzothiazin-4-ones
amino defluorination
benzoyl isothiocyanates
heterocyclization
nitro compounds
organofluorine compounds
tuberculostatic activity
Abstract
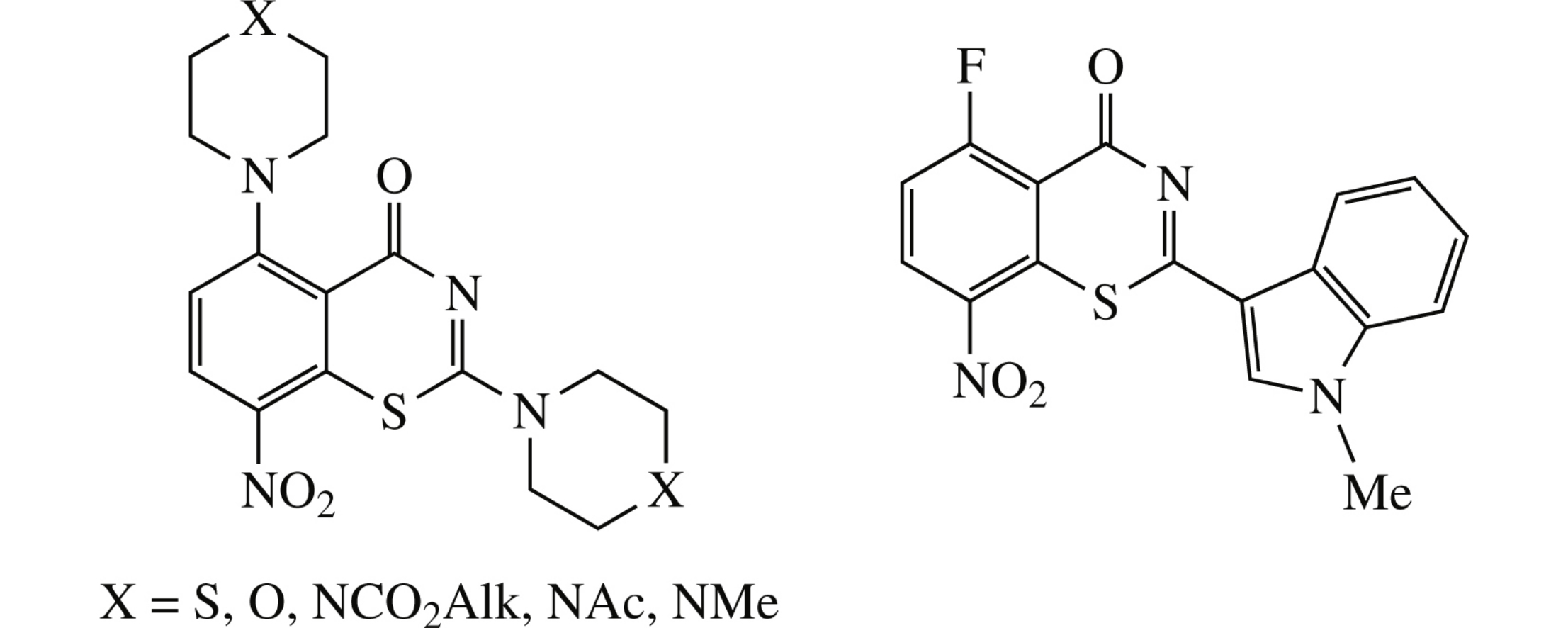
New 2,5-bis(azacyclohex-1-yl)-8-nitro-1,3-benzothiazin-4-ones were synthesized from 2,6-difluorobenzoic acid in two preparative stages. The ethoxycarbonylpiperazino derivative surpasses in tuberculostatic activity (MIC 4μgml−1) its 5-fluoro-8-H-counterpart. The first representative of 5-fluoro-8-nitro-1,3-benzothiazin-4-ones was obtained through the condensation of 2,6-difluoro-3-nitrobenzoyl isothiocyanate and N-methylindole.
References
1.

Kumar V., Patel S., Jain R.
Medicinal Research Reviews,
2017
2.

Nosova E.V., Lipunova G.N., Charushin V.N., Chupakhin O.N.
Mini-Reviews in Medicinal Chemistry,
2018
3.

Chetty S., Ramesh M., Singh-Pillay A., Soliman M.E.
Bioorganic and Medicinal Chemistry Letters,
2017
4.

D. Joshi S., Kumar D., R. Dixit S., S. Joshi A., M. Aminabhavi T.
Mini-Reviews in Organic Chemistry,
2016
5.

Branco F., Pinto A., Boechat N.
Current Topics in Medicinal Chemistry,
2013
6.

Makarov V., Lechartier B., Zhang M., Neres J., Sar A.M., Raadsen S.A., Hartkoorn R.C., Ryabova O.B., Vocat A., Decosterd L.A., Widmer N., Buclin T., Bitter W., Andries K., Pojer F., et. al.
EMBO Molecular Medicine,
2014
7.

Batt S.M., Cacho Izquierdo M., Castro Pichel J., Stubbs C.J., Vela-Glez Del Peral L., Pérez-Herrán E., Dhar N., Mouzon B., Rees M., Hutchinson J.P., Young R.J., McKinney J.D., Barros Aguirre D., Ballell L., Besra G.S., et. al.
ACS Infectious Diseases,
2015
8.

Trefzer C., Rengifo-Gonzalez M., Hinner M.J., Schneider P., Makarov V., Cole S.T., Johnsson K.
Journal of the American Chemical Society,
2010
9.

Nosova E.V., Batanova O.A., Lipunova G.N., Kotovskaya S.K., Slepukhin P.A., Kravchenko M.A., Charushin V.N.
Journal of Fluorine Chemistry,
2019
10.

Nosova E.V., Poteeva A.D., Lipunova G.N., Slepukhin P.A., Charushin V.N.
Russian Journal of Organic Chemistry,
2019
11.

Yoshida Y., Barrett D., Azami H., Morinaga C., Matsumoto S., Matsumoto Y., Takasugi H.
Bioorganic and Medicinal Chemistry,
1999