Keywords
bacteriochlorins
chlorins
fluorescence diagnostics
nanoparticles
photodynamic therapy
targeted photosensitizers
Abstract
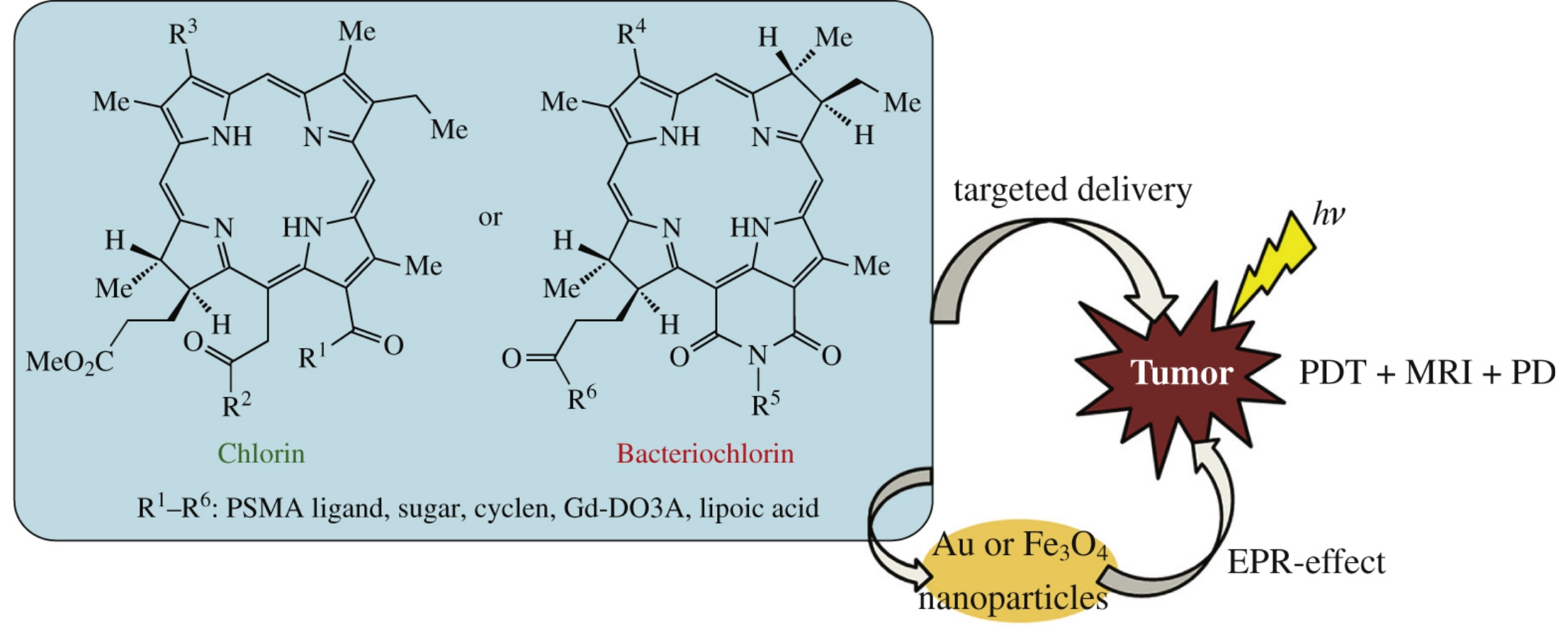
This review deals with the progress in the chemistry of natural chlorins and the prospects of using their derivatives as theranostics. Particular attention is paid to the creation of targeted photosensitizers by addition of vector molecules to chlorins and bacteriochlorins, or by immobilization onto nanoparticles of various nature. The results of biological tests that establish relationship between the structure, antitumor activity, and diagnostic potential of the resulting photosensitizers are presented.
References
1.
10.1016/j.mencom.2020.07.003_bib0005
Bonnett
Chemical Aspects of Photodynamic Therapy,
2000
2.

Celli J.P., Spring B.Q., Rizvi I., Evans C.L., Samkoe K.S., Verma S., Pogue B.W., Hasan T.
Chemical Reviews,
2010
3.

Agostinis P., Berg K., Cengel K.A., Foster T.H., Girotti A.W., Gollnick S.O., Hahn S.M., Hamblin M.R., Juzeniene A., Kessel D., Korbelik M., Moan J., Mroz P., Nowis D., Piette J., et. al.
Ca-A Cancer Journal for Clinicians,
2011
4.

Dabrowski J.M., Arnaut L.G.
Photochemical and Photobiological Sciences,
2015
5.

Dougherty T.J., Gomer C.J., Henderson B.W., Jori G., Kessel D., Korbelik M., Moan J., Peng Q.
Journal of the National Cancer Institute,
1998
6.

Mironov A.F.
Russian Journal of General Chemistry,
2019
7.
10.1016/j.mencom.2020.07.003_bib0035
Pushpan
Curr. Med. Chem.: Anti-Cancer Agents,
2002
8.

9.

Abrahamse H., Hamblin M.
Biochemical Journal,
2016
10.
10.1016/j.mencom.2020.07.003_bib0050
Mironov
Itogi Nauki i Tekhniki. Sovremennie Problemy Lazernoi Fiziki,
1989
11.
10.1016/j.mencom.2020.07.003_bib0055
Moan
Photodynamic Therapy,
2003
12.

Trindade F.Z., Pavarina A.C., Ribeiro A.P., Bagnato V.S., Vergani C.E., de Souza Costa C.A.
Lasers in Medical Science,
2011
13.

Zang L., Zhao H., Fang Q., Fan M., Chen T., Tian Y., Yao J., Zheng Y., Zhang Z., Cao W.
Journal of Porphyrins and Phthalocyanines,
2017
14.
Hu D., Chen L., Qu Y., Peng J., Chu B., Shi K., Hao Y., Zhong L., Wang M., Qian Z.
Theranostics,
2018
15.
![2-[1-hexyloxyethyl]-2-devinyl pyropheophorbide-a (HPPH) in a nude rat glioma model: Implications for photodynamic therapy](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Lobel J., MacDonald I.J., Ciesielski M.J., Barone T., Potter W.R., Pollina J., Plunkett R.J., Fenstermaker R.A., Dougherty T.J.
Lasers in Surgery and Medicine,
2001
16.
10.1016/j.mencom.2020.07.003_bib0080
Partrice
Photodynamic Therapy,
2003
17.

Zverev V.V., Makarov O.V., Khashukoeva A.Z., Svitich O.A., Dobrokhotova Y.E., Markova E.A., Labginov P.A., Khlinova S.A., Shulenina E.A., Gankovskaya L.V.
Lasers in Medical Science,
2016
18.

Dolmans D.E., Fukumura D., Jain R.K.
Nature Reviews Cancer,
2003
19.

Brilkina A.A., Dubasova L.V., Sergeeva E.A., Pospelov A.J., Shilyagina N.Y., Shakhova N.M., Balalaeva I.V.
Journal of Photochemistry and Photobiology B: Biology,
2019
20.

Brun P.H., De Groot J.L., Gudgin Dickson E.F., Farahani M., Pottier R.H.
Photochemical and Photobiological Sciences,
2004
21.

Chevalier S., Anidjar M., Scarlata E., Hamel L., Scherz A., Ficheux H., Borenstein N., Fiette L., Elhilali M.
Journal of Urology,
2011
22.

Scherz A., Salomon Y., Coleman J.
Photodiagnosis and Photodynamic Therapy,
2017
23.

Yang N.J., Hinner M.J.
Methods in Molecular Biology,
2014
24.

Parslow A., Parakh S., Lee F., Gan H., Scott A.
Biomedicines,
2016
25.

Casi G., Neri D.
Journal of Medicinal Chemistry,
2015
26.

Kurzrock R., Gabrail N., Chandhasin C., Moulder S., Smith C., Brenner A., Sankhala K., Mita A., Elian K., Bouchard D., Sarantopoulos J.
Molecular Cancer Therapeutics,
2012
27.

Liu X., Guo J., Han S., Yao L., Chen A., Yang Q., Bo H., Xu P., Yin J., Zhang Z.
Vaccine,
2012
28.

Manzoor A.A., Lindner L.H., Landon C.D., Park J., Simnick A.J., Dreher M.R., Das S., Hanna G., Park W., Chilkoti A., Koning G.A., ten Hagen T.L., Needham D., Dewhirst M.W.
Cancer Research,
2012
29.

Yoo J., Chambers E., Mitragotri S.
Current Pharmaceutical Design,
2010
30.

Zheng X., Pandey R.
Anti-Cancer Agents in Medicinal Chemistry,
2008
31.

Aksenova A.A., Sebyakin Y.L., Mironov A.F.
Russian Journal of Bioorganic Chemistry,
2000
32.

Aksenova A.A., Sebyakin Y.L., Mironov A.F.
Russian Journal of Bioorganic Chemistry,
2001
33.
Lebedeva V.S., Ruziev R.D., Popov A.V., Sebyakin Y.L., Mironov A.F.
Mendeleev Communications,
2007
34.

Mironov A.F., Lebedeva V.S.
Tetrahedron Letters,
1998
35.
Grin M.A., Lonin I.S., Makarov A.I., Lakhina A.A., Toukach F.V., Kachala V.V., Orlova A.V., Mironov A.F.
Mendeleev Communications,
2008
36.
10.1016/j.mencom.2020.07.003_bib0180
Lonin
J. Porphyrins Phthalocyanines,
2008
37.

Grin M.A., Lonin I.S., Lakhina A.A., Ol'shanskaya E.S., Makarov A.I., Sebyakin Y.L., Guryeva L.Y., Toukach P.V., Kononikhin A.S., Kuzmin V.A., Mironov A.F.
Journal of Porphyrins and Phthalocyanines,
2009
38.

Pandey S.K., Zheng X., Morgan J., Missert J.R., Liu T., Shibata M., Bellnier D.A., Oseroff A.R., Henderson B.W., Dougherty T.J., Pandey R.K.
Molecular Pharmaceutics,
2007
39.
Lonin I.S., Grin M.A., Lakhina A.A., Mironov A.F.
Mendeleev Communications,
2012
40.
10.1016/j.mencom.2020.07.003_bib0200
Plotnikova
Rossiiskii Bioterapevticheskii Zhurnal,
2012
41.

Grin M.A., Lonin I.S., Likhosherstov L.M., Novikova O.S., Plyutinskaya A.D., Plotnikova E.A., Kachala V.V., Yakubovskaya R.I., Mironov A.F.
Journal of Porphyrins and Phthalocyanines,
2012
42.

Mironov A.F., Kozyrev A.N., Brandis A.S.
Proceedings of SPIE - The International Society for Optical Engineering,
1993
43.

Mironov A.F., Grin M.A., Tsiprovskiy A.G., Kachala V.V., Karmakova T.A., Plyutinskaya A.D., Yakubovskaya R.I.
Journal of Porphyrins and Phthalocyanines,
2009
44.

Stoermer D., Liu Q., Hall M.R., Flanary J.M., Thomas A.G., Rojas C., Slusher B.S., Tsukamoto T.
Bioorganic and Medicinal Chemistry Letters,
2003
45.

Synthesis and Biological Analysis of Prostate-Specific Membrane Antigen-Targeted Anticancer Prodrugs
Kularatne S.A., Venkatesh C., Santhapuram H.R., Wang K., Vaitilingam B., Henne W.A., Low P.S.
Journal of Medicinal Chemistry,
2010
46.

Hillier S.M., Maresca K.P., Lu G., Merkin R.D., Marquis J.C., Zimmerman C.N., Eckelman W.C., Joyal J.L., Babich J.W.
Journal of Nuclear Medicine,
2013
47.
M. A. Grin, N.V. Suvorov, A.E. Machulkin, E.A. Plotnikova, R.I. Yakubovskaya, A.G. Majouga, A.F. Mironov and E. V. Filonenko, Patent RU 2670087C1, 2018.
48.

Hargus J.A., Fronczek F.R., Vicente M.G., Smith K.M.
Photochemistry and Photobiology,
2007
49.

Suvorov N.V., Machulkin A.E., Ivanova A.V., Popkov A.M., Bondareva E.A., Plotnikova E.A., Yakubovskaya R.I., Majouga A.G., Mironov A.F., Grin M.A.
Journal of Porphyrins and Phthalocyanines,
2018
50.
Suvorov N.V., Cheskov D.A., Mironov A.F., Grin M.A.
Mendeleev Communications,
2019
51.

Oliveira B.L., Guo Z., Bernardes G.J.
Chemical Society Reviews,
2017
52.

Sharonov G.V., Karmakova T.A., Kassies R., Pljutinskaya A.D., Grin M.A., Refregiers M., Yakubovskaya R.I., Mironov A.F., Maurizot J., Vigny P., Otto C., Feofanov A.V.
Free Radical Biology and Medicine,
2006
53.
V. I. Chissov, R.I. Yakubovskaya, A.F. Mironov, M.A. Grin, E.A. Plotnikova, N.B. Morozova and A. A. Tsygankov, Patent RU 2521327C1, 2012.
54.

Zhao Z., Zhou Z., Bao J., Wang Z., Hu J., Chi X., Ni K., Wang R., Chen X., Chen Z., Gao J.
Nature Communications,
2013
55.

Ostroverkhov P.V., Semkina A.S., Naumenko V.A., Plotnikova E.A., Melnikov P.A., Abakumova T.O., Yakubovskaya R.I., Mironov A.F., Vodopyanov S.S., Abakumov A.M., Majouga A.G., Grin M.A., Chekhonin V.P., Abakumov M.A.
Journal of Colloid and Interface Science,
2019
56.

Ostroverkhov P., Semkina A., Nikitin A., Smirnov A., Vedenyapina D., Vlasova K., Kireev I., Grin M., Chekhonin V., Majouga A., Abakumov M.
Journal of Magnetism and Magnetic Materials,
2019
57.

Peer D., Karp J.M., Hong S., Farokhzad O.C., Margalit R., Langer R.
Nature Nanotechnology,
2007
58.

GHOSH P., HAN G., DE M., KIM C., ROTELLO V.
Advanced Drug Delivery Reviews,
2008
59.

Rana S., Bajaj A., Mout R., Rotello V.M.
Advanced Drug Delivery Reviews,
2012
60.

61.

Suvorov N.V., Grin M.A., Popkov A.M., Garanina A.S., Mironov A.F., Majouga A.G.
Macroheterocycles,
2016
62.

Dreaden E.C., Alkilany A.M., Huang X., Murphy C.J., El-Sayed M.A.
Chemical Society Reviews,
2012
63.

Bardhan R., Lal S., Joshi A., Halas N.J.
Accounts of Chemical Research,
2011
64.

Xia Y., Li W., Cobley C.M., Chen J., Xia X., Zhang Q., Yang M., Cho E.C., Brown P.K.
Accounts of Chemical Research,
2011
65.

Pantiushenko I.V., Rudakovskaya P.G., Starovoytova A.V., Mikhaylovskaya A.A., Abakumov M.A., Kaplan M.A., Tsygankov A.A., Majouga A.G., Grin M.A., Mironov A.F.
Biochemistry (Moscow),
2015
66.

Murray K.S., Winter A.G., Corradi R.B., LaRosa S., Jebiwott S., Somma A., Takaki H., Srimathveeravalli G., Lepherd M., Monette S., Kim K., Scherz A., Coleman J.A.
Journal of Urology,
2016
67.
Neuschmelting V., Kim K., Malekzadeh-Najafabadi J., Jebiwott S., Prakash J., Scherz A., Coleman J.A., Kircher M.F., Ntziachristos V.
Theranostics,
2017
68.
10.1016/j.mencom.2020.07.003_bib0340
Rashid
Rev. Roum. Chim.,
2014
69.
10.1016/j.mencom.2020.07.003_bib0345
Thomsen
Image Principles, Neck, and the Brain,
2016
70.

Rashid H.U., Martines M.A., Jorge J., de Moraes P.M., Umar M.N., Khan K., Rehman H.U.
Bioorganic and Medicinal Chemistry,
2016
71.

Cakić N., Gündüz S., Rengarasu R., Angelovski G.
Tetrahedron Letters,
2015
72.
10.1016/j.mencom.2020.07.003_bib0360
Tóth
2003
73.
Grin M.A., Brusov S.S., Shchepelina E.Y., Ponomarev P.V., Khrenova M.K., Smirnov A.S., Lebedeva V.S., Mironov A.F.
Mendeleev Communications,
2017
74.
10.1016/j.mencom.2020.07.003_bib0370
Smirnov
Tonkie Khim. Tekhnol. (Fine Chem. Technol.),
2019