Keywords
Abstract
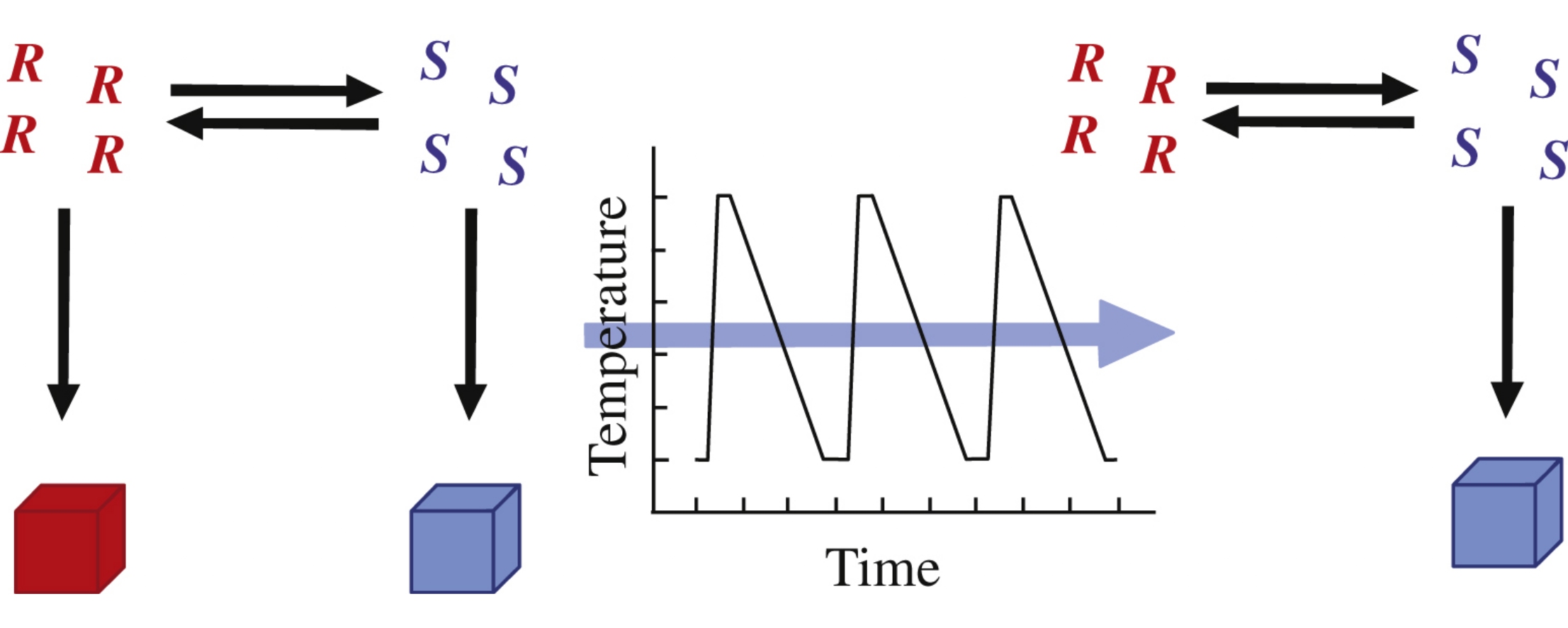
The problem of separation and purification of the enantiomers of chiral species is a significant issue in the production of modern chemicals of pharmaceutical, agricultural and food industries. Efficient methods enabling a complete conversion of a racemic mixture into the desired enantiomer would be of great benefit to industry. Temperature cycle induced deracemization (TCID), a process allowing an initially racemic crystal phase of a suspension to be converted into an enantiopure state, combines solution phase racemization of the solute molecules and a series of temperature cycles inducing dissolution and crystal growth. The process first described as a more convenient and scalable alternative to Viedma ripening, has now been successfully tested on a wide range of chiral components that are conglomerate forming and racemizable. This review discusses the origins of TCID, potential mechanisms responsible for the deracemization, and also some related processes.
References