Keywords
coordination
dimethyl sulfoxide
hydrogen bond
hydrophilic core
hydrophobic shell
monoethanolamine
stability
Abstract
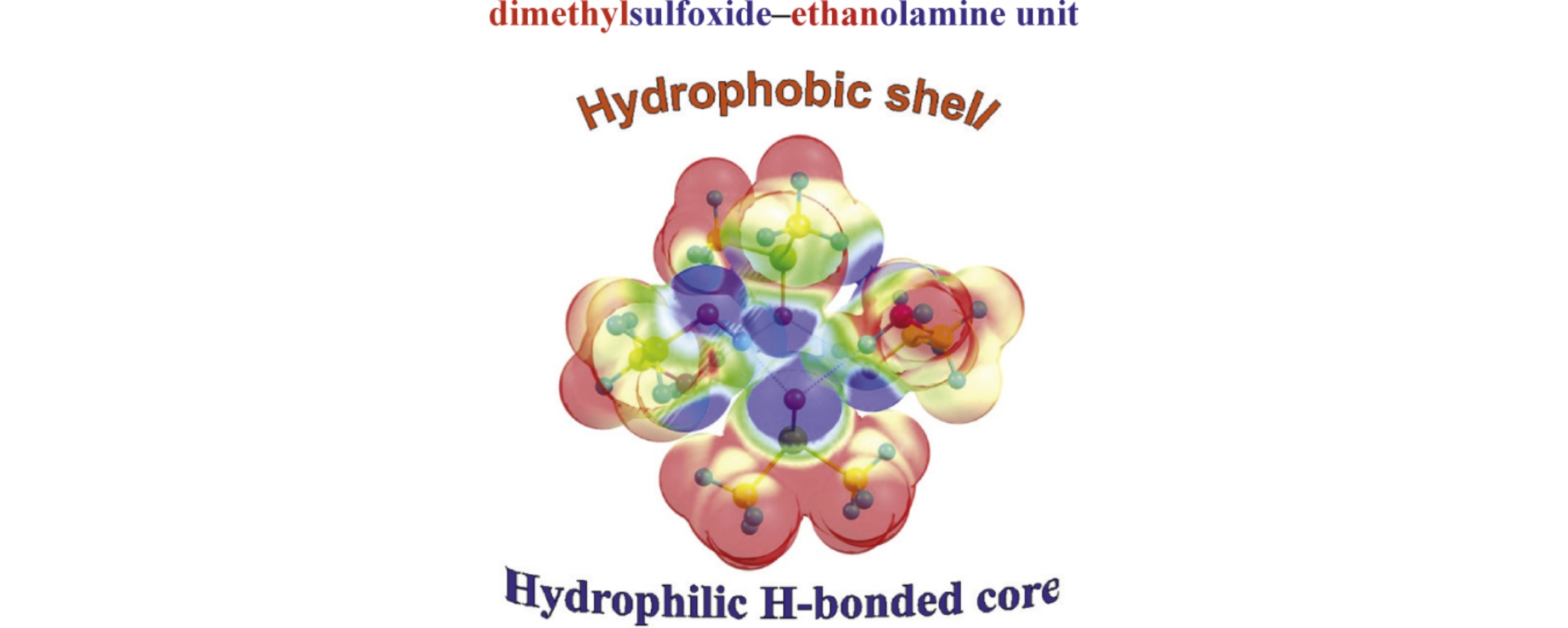
Nonempirical simulations at the MP2/6-31++G(d,p) level revealed favorable building blocks in the dimethyl sulfoxide (DMSO)–monoethanolamine (MEA) binary system. The analysis of diverse coordination motifs of MEA and DMSO molecules showed that energetically preferable configurations are those composed of equal numbers of DMSO and gauche MEA molecules, where OH, NH2, and SO groups of all molecules are conglomerated in a hydrophilic core surrounded with a hydrophobic methyl–methylene shell, and amino groups act as bridges between the two parts.
References
1.

Havemeyer R.N.
Journal of Pharmaceutical Sciences,
1966
2.

Baudot A., Cacela C., Duarte M.L., Fausto R.
Cryobiology,
2002
3.

Yu Z.-., Quinn P.J.
Bioscience Reports,
1994
4.

Anchordoguy T.J., Cecchini C.A., Crowe J.H., Crowe L.M.
Cryobiology,
1991
5.

RASMUSSEN D.H., MACKENZIE A.P.
Nature,
1968
6.
M. N. Rodnikova, Dr. Sci. Thesis, Moscow, 1998.
7.

Solonina I.A., Rodnikova M.N., Kiselev M.R., Khoroshilov A.V., Makaev S.V.
Russian Journal of Inorganic Chemistry,
2019
8.

Tarakeshwar P., Kim K.S., Brutschy B.
Journal of Chemical Physics,
2000
9.
A. A. Granovsky, Firefly version 8, http://classic.chem.msu.su/gran/firefly/index.html.
10.
Chemcraft, https://www.chemcraftprog.com.
11.
10.1016/j.mencom.2020.05.039_bib0055
Faber
Z. Naturforsch.,
1969
12.

Chang Y.-., Su T.-., Li T.-., Chao I.
Journal of Physical Chemistry A,
1997
13.

Vorobyov I., Yappert M.C., DuPré D.B.
Journal of Physical Chemistry A,
2002
14.

Wang K., Shan X., Chen X.
Journal of Molecular Structure THEOCHEM,
2009
15.
10.1016/j.mencom.2020.05.039_bib0075
Sun
J. Comput. Sci. Eng.,
2011
16.

da Silva E.F., Kuznetsova T., Kvamme B., Merz K.M.
Journal of Physical Chemistry B,
2007
17.

Tubergen M.J., Torok C.R., Lavrich R.J.
Journal of Chemical Physics,
2003
18.

Penn R.E., Curl R.F.
Journal of Chemical Physics,
1971
19.

Penn R.E., Olsen R.J.
Journal of Molecular Spectroscopy,
1976
20.

Widicus S.L., Drouin B.J., Dyl K.A., Blake G.A.
Journal of Molecular Spectroscopy,
2003
21.

Novakovskaya Y.V., Rodnikova M.N.
Structural Chemistry,
2014
22.

Novakovskaya Y.V., Rodnikova M.N.
Doklady Physical Chemistry,
2016