Keywords
[3+2]-cycloaddition
azomethine ylides
C60 fullerene
fulleropyrrolidines
norbornadiene
Prato reaction
pyrrolidines
quadricyclane
Abstract
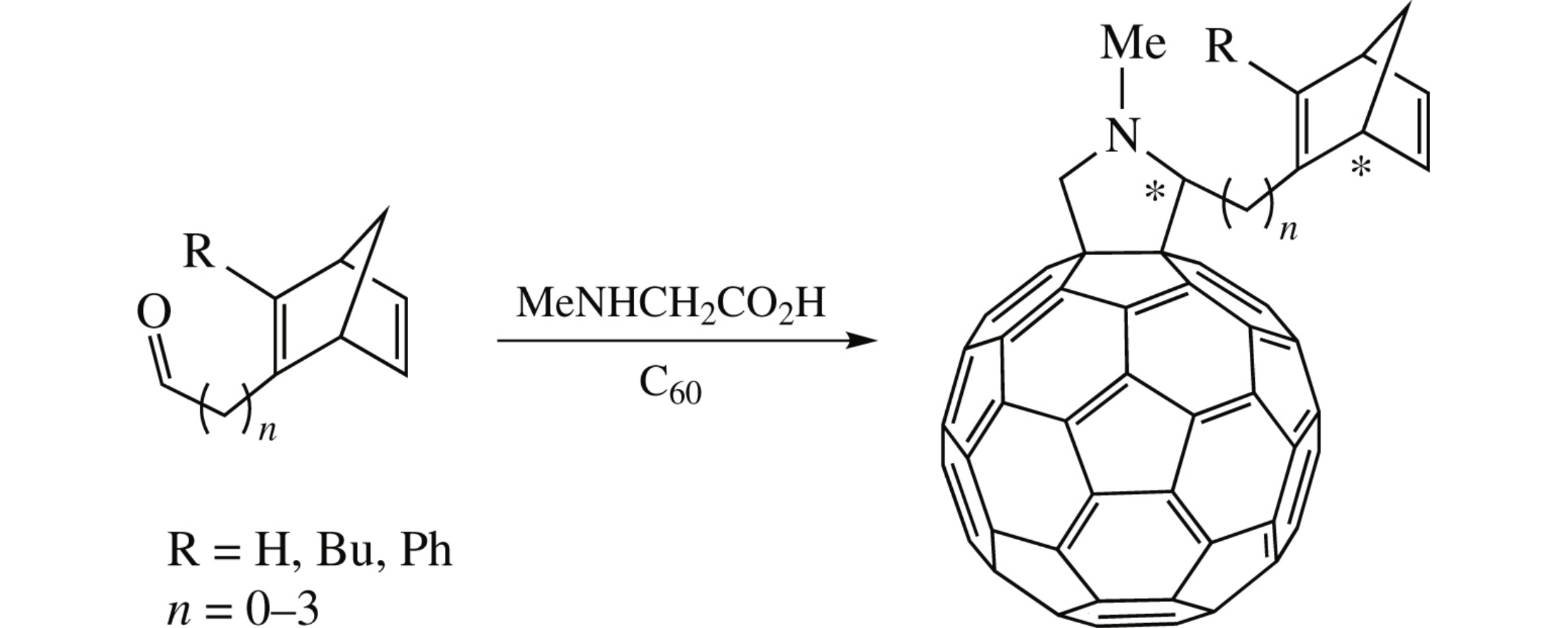
N-Methyl-3,4-fulleropyrrolidine–norbornadiene conjugates with or without (CH2)n (n=0–3) spacers were synthesized by the Prato reaction from the corresponding norbornadiene aldehydes. The influence of the spacer length on the diastereomer composition of the conjugates with two chiral centres studied by 13C NMR revealed a pronounced diastereotopic splitting of the fullerene carbon signals relating to four diastereomers in the case when the norbornadiene substituent was directly attached to the pyrrolidine moiety. When norbornadiene moiety was separated from the pyrrolidine one by three methylene units, the diastereotopic difference was lower and some of the signals were grouped.
References
1.

Hammond G.S., Wyatt P., DeBoer C.D., Turro N.J.
Journal of the American Chemical Society,
1964
2.

Bren' V.A., Dubonosov A.D., Minkin V.I., Chernoivanov V.A.
Russian Chemical Reviews,
1991
3.

Dubonosov A.D., Bren V.A., Chernoivanov V.A.
Russian Chemical Reviews,
2002
4.
![Enthalpy of the metal catalyzed isomerizations of quadricyclane and of tricyclo[4.1.0.02,7]heptane](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Wiberg K.B., Connon H.A.
Journal of the American Chemical Society,
1976
5.

Bonfantini E.E., Officer D.L.
Journal of the Chemical Society Chemical Communications,
1994
6.

Lainé P., Marvaud V., Gourdon A., Launay J., Argazzi R., Bignozzi C.
Inorganic Chemistry,
1996
7.

Fraysse S., Coudret C., Launay J.
European Journal of Inorganic Chemistry,
2000
8.
Morino S., Watanabe T., Magaya Y., Yamashita T., Horie K., Nishikubo T.
Journal of Photopolymer Science and Technology,
1994
9.

Takahashi S., Samata K., Muta H., Machida S., Horie K.
Applied Physics Letters,
2001
10.

Herges R., Reif W.
European Journal of Organic Chemistry,
2006
11.

Starck F., Jones P.G., Herges R.
European Journal of Organic Chemistry,
1998
12.

Nakatsuji S., Takeuchi S., Ojima T., Ogawa Y., Akutsu H., Yamada J.
Molecular Crystals and Liquid Crystals Science and Technology Section A Molecular Crystals and Liquid Crystals,
2001
13.

Takeuchi S., Ogawa Y., Naito A., Sudo K., Yasuoka N., Akutsu H., Yamada J., Nakatsuji S.
Molecular Crystals and Liquid Crystals Science and Technology Section A Molecular Crystals and Liquid Crystals,
2000
14.

Nakatsuji S., Ogawa Y., Takeuchi S., Akutsu H., Yamada J., Naito A., Sudo K., Yasuoka N.
Journal of the Chemical Society Perkin Transactions 2,
2000
15.

Wright M.E., Allred G.D., Wardle R.B., Cannizzo L.F.
Journal of Organic Chemistry,
1993
16.
![An ambipolar organic field-effect transistor using oligothiophene incorporated with two [60]fullerenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Kunugi Y., Takimiya K., Negishi N., Otsubo T., Aso Y.
Journal of Materials Chemistry A,
2004
17.

Matsumoto K., Hashimoto K., Kamo M., Uetani Y., Hayase S., Kawatsura M., Itoh T.
Journal of Materials Chemistry A,
2010
18.
10.1016/j.mencom.2020.05.031_bib0090
Carini
Handbook of Carbon Nano Materials,
2012
19.

Castro E., Garcia A.H., Zavala G., Echegoyen L.
Journal of Materials Chemistry B,
2017
20.

Flid V.R., Gringolts M.L., Shamsiev R.S., Finkelshtein E.S.
Russian Chemical Reviews,
2018
21.

Durakov S.A., Shamsiev R.S., Flid V.R., Gekhman A.E.
Russian Chemical Bulletin,
2018
22.

Tuktarov A.R., Akhmetov A.R., Khuzin A.A., Dzhemilev U.M.
Journal of Organic Chemistry,
2018
23.

Maggini M., Scorrano G., Prato M.
Journal of the American Chemical Society,
1993
24.

Tanyeli C., Çelikel G., Akhmedov İ.M.
Tetrahedron Asymmetry,
2001
25.

Ishihara K., Kondo S., Kurihara H., Yamamoto H., Ohashi S., Inagaki S.
Journal of Organic Chemistry,
1997
26.

Tulyabaev A.R., Tuktarov A.R., Khalilov L.M.
Magnetic Resonance in Chemistry,
2013
27.

Tranmer G.K., Tam W.
Journal of Organic Chemistry,
2001
28.
![Nickel-catalyzed [2.pi. + 2.pi. + 2.pi.] (homo-Diels-Alder) and [2.pi. + 2.pi.] cycloadditions of bicyclo[2.2.1]hepta-2,5-dienes.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Lautens M., Edwards L.G., Tam W., Lough A.J.
Journal of the American Chemical Society,
1995
29.

Petrov V., Vasil'ev N.
Current Organic Synthesis,
2006