Keywords
3- or 2-(2-oxoimidazolidin-5-yl)indoles
amidoalkylation
anti-inflammatory activity
biheterocycles
imidazolylindoles
indoles
Abstract
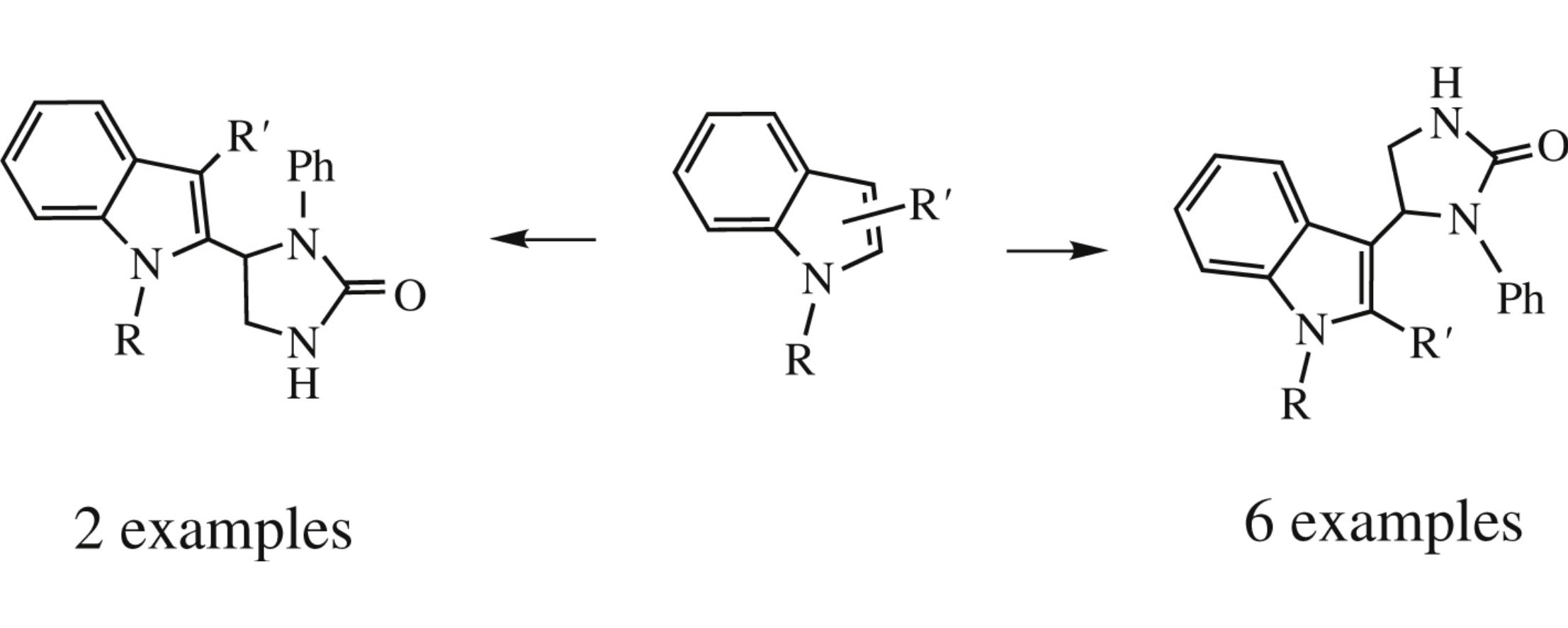
Boron trifluoride-catalyzed amidoalkylation of indole derivatives with 5-hydroxy-1-phenylimidozolidin-2-one affords new biheterocycles with a direct C–C bond. Among them, 3- or 2-(2-oxoimidazolidin-5-yl)indoles manifest antiinflammatory activity with relatively low toxicity.
References
1.

Indole in the target-based design of anticancer agents: A versatile scaffold with diverse mechanisms
Dadashpour S., Emami S.
European Journal of Medicinal Chemistry,
2018
2.

Thanikachalam P.V., Maurya R.K., Garg V., Monga V.
European Journal of Medicinal Chemistry,
2019
3.

Verma M., Tripathi M., Saxena A., Shanker K.
European Journal of Medicinal Chemistry,
1994
4.

Dousson C., Alexandre F., Amador A., Bonaric S., Bot S., Caillet C., Convard T., da Costa D., Lioure M., Roland A., Rosinovsky E., Maldonado S., Parsy C., Trochet C., Storer R., et. al.
Journal of Medicinal Chemistry,
2016
5.

Stec J., Onajole O.K., Lun S., Guo H., Merenbloom B., Vistoli G., Bishai W.R., Kozikowski A.P.
Journal of Medicinal Chemistry,
2016
6.

Yadav R.R., Khan S.I., Singh S., Khan I.A., Vishwakarma R.A., Bharate S.B.
European Journal of Medicinal Chemistry,
2015
7.

Praveen C., Ayyanar A., Perumal P.T.
Bioorganic and Medicinal Chemistry Letters,
2011
8.

Nomura S., Yamamoto Y., Matsumura Y., Ohba K., Sakamaki S., Kimata H., Nakayama K., Kuriyama C., Matsushita Y., Ueta K., Tsuda-Tsukimoto M.
ACS Medicinal Chemistry Letters,
2013
9.
![Synthesis under Microwave Irradiation of [1,2,4]Triazolo[3,4-b] [1,3,4]thiadiazoles and Other Diazoles Bearing Indole Moieties and Their Antimicrobial Evaluation](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
10.

Weng J., Tsai C., Kulp S.K., Chen C.
Cancer Letters,
2008
11.

Wan Y., Li Y., Yan C., Yan M., Tang Z.
European Journal of Medicinal Chemistry,
2019
12.

Zhang M., Jia C., Gu Y., Mulholland N., Turner S., Beattie D., Zhang W., Yang G., Clough J.
European Journal of Medicinal Chemistry,
2017
13.
Dutov M.D., Kachala V.V., Ugrak B.I., Korolev V.A., Popkov S.V., Aleksanyan D.R., Rusina O.N., Aleksanyan K.G., Koshelev V.N.
Mendeleev Communications,
2018
14.

Rajan S., Puri S., Kumar D., Babu M.H., Shankar K., Varshney S., Srivastava A., Gupta A., Reddy M.S., Gaikwad A.N.
European Journal of Medicinal Chemistry,
2018
15.
Vereshchagin A.N., Frolov N.A., Konyuhova V.Y., Hansford K.A., Egorov M.P.
Mendeleev Communications,
2019
16.
Lemport P.S., Roznyatovsky V.A., Tarasevich B.N., Khromov O.V., Khrustalev V.N., Rozentsveig I.B., Nenajdenko V.G.
Mendeleev Communications,
2019
17.
Epishina M.A., Kulikov A.S., Fershtat L.L., Ananyev I.V., Makhova N.N.
Mendeleev Communications,
2019
18.

Reader J.C., Matthews T.P., Klair S., Cheung K.J., Scanlon J., Proisy N., Addison G., Ellard J., Piton N., Taylor S., Cherry M., Fisher M., Boxall K., Burns S., Walton M.I., et. al.
Journal of Medicinal Chemistry,
2011
19.
K. Karabelas, A. Astra, M. Lepistö and P. Sj, WO Patent 99/32483, C07D 403/04, 1998.
20.
10.1016/j.mencom.2020.05.029_bib0100
Lewis
Cochrane Database of Systematic Reviews,
2005
21.

Archana S., Ranganathan R., Dinesh M., Arul P., Ponnuswamy A., Kalaiselvi P., Chellammal S., Subramanian G.
Research on Chemical Intermediates,
2016
22.

Cortes S., Kohn H.
Journal of Organic Chemistry,
1983
23.

Sviridova L.A., Aganas'eva S.V., Golubeva G.A., Terent'ev P.B., Bundel' Y.G.
Chemistry of Heterocyclic Compounds,
1990
24.

Sadovoy A.V., Kovrov A.E., Golubeva G.A., Sviridova L.A.
Chemistry of Heterocyclic Compounds,
2011
25.

PEREIRA E.R., SANCELME M., TOWA J., PRUDHOMME M., MARTRE A., MOUSSET G., RAPP M.
Journal of Antibiotics,
1996
26.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
27.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
28.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007