Keywords
biphenyl scaffold
docking
electrophysiological investigations
negative allosteric modulator
NMDA receptors
Abstract
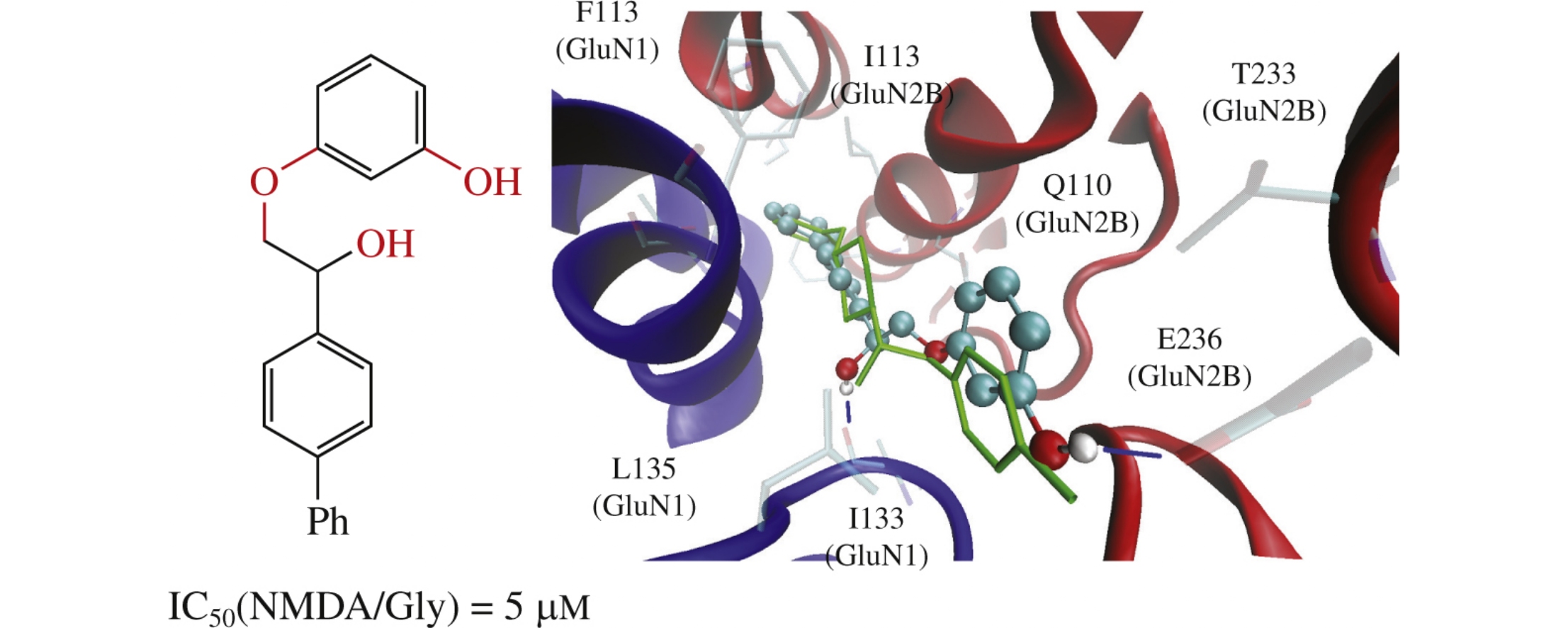
Ifenprodil-like NMDA receptor negative allosteric modulator based on the biphenyl scaffold has been identified using virtual screening. Docking approach demonstrated that the modulator maintains some characteristic interactions with protein similar to ifenprodil. Electrophysiological and radioligand investigations demonstrated the concentration-dependent NMDA/Gly-induced currents inhibition with IC50=5μM and significant displacement of [H3]ifenprodil with EC50=30μM, respectively.
References
1.

Traynelis S.F., Wollmuth L.P., McBain C.J., Menniti F.S., Vance K.M., Ogden K.K., Hansen K.B., Yuan H., Myers S.J., Dingledine R.
Pharmacological Reviews,
2010
2.

Watkins J.C., Jane D.E.
British Journal of Pharmacology,
2006
3.

Olney J.W., Labruyere J., Price M.T.
Science,
1989
4.

Berman R.M., Cappiello A., Anand A., Oren D.A., Heninger G.R., Charney D.S., Krystal J.H.
Biological Psychiatry,
2000
5.

Moghaddam B., Adams B., Verma A., Daly D.
Journal of Neuroscience,
1997
6.

Regan M.C., Zhu Z., Yuan H., Myers S.J., Menaldino D.S., Tahirovic Y.A., Liotta D.C., Traynelis S.F., Furukawa H.
Nature Communications,
2019
7.

Stroebel D., Buhl D.L., Knafels J.D., Chanda P.K., Green M., Sciabola S., Mony L., Paoletti P., Pandit J.
Molecular Pharmacology,
2016
8.

Karakas E., Simorowski N., Furukawa H.
Nature,
2011
9.

Tajima N., Karakas E., Grant T., Simorowski N., Diaz-Avalos R., Grigorieff N., Furukawa H.
Nature,
2016
10.

Karlov D.S., Radchenko E.V., Zefirov A.N., Palyulin V.A., Pentkovski V.M., Zefirov N.S.
Biochemical and Biophysical Research Communications,
2012
11.

Borza I., Kolok S., Gere A., Nagy J., Fodor L., Galgóczy K., Fetter J., Bertha F., Ágai B., Horváth C., Farkas S., Domány G.
Bioorganic and Medicinal Chemistry Letters,
2006
12.

McCauley J.A., Theberge C.R., Romano J.J., Billings S.B., Anderson K.D., Claremon D.A., Freidinger R.M., Bednar R.A., Mosser S.D., Gaul S.L., Connolly T.M., Condra C.L., Xia M., Cunningham M.E., Bednar B., et. al.
Journal of Medicinal Chemistry,
2004
13.

Sokolov V.B., Aksinenko A.Y., Gabrel’yan A.V., Grigoriev V.V.
Russian Chemical Bulletin,
2019
14.

Kobayashi T., Washiyama K., Ikeda K.
Neuropsychopharmacology,
2005
15.

Finlayson K., Witchel H.J., McCulloch J., Sharkey J.
European Journal of Pharmacology,
2004
16.

Cavalli A., Poluzzi E., De Ponti F., Recanatini M.
Journal of Medicinal Chemistry,
2002
17.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
18.

Kew J.N., Richards J.G., Mutel V., Kemp J.A.
Journal of Neuroscience,
1998
19.

Radchenko E.V., Rulev Y.A., Safanyaev A.Y., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2017