Keywords
azomethine ylides
dispiro compounds
indolinones
pyrrolidines
selenohydantoins
spiro compounds
X-ray
Abstract
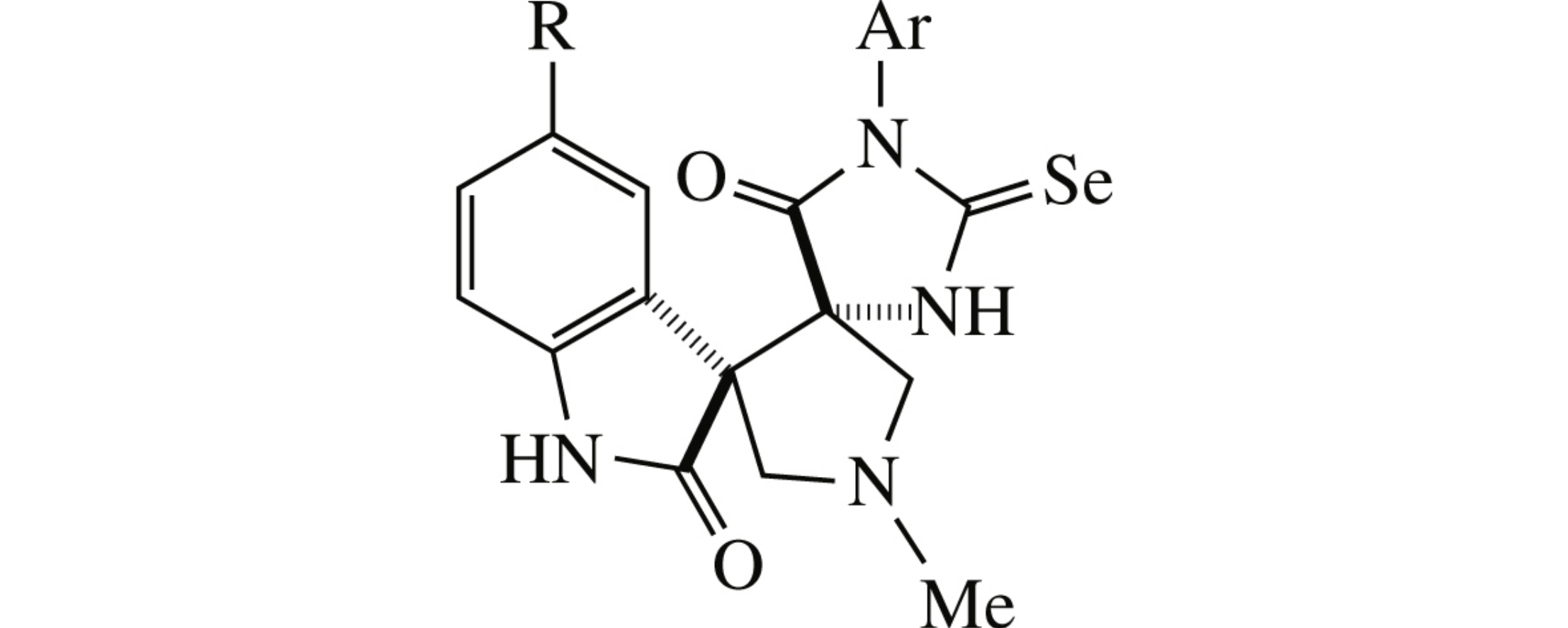
New 2-selenoxodispiro[imidazolidine-4,3′-pyrrolidine-4′,3″-indoline]-2″,5-dione system was accessed by addition of azomethine ylide at the double bond of (Z)-(2-oxoindolin-3-ylidene)-2-selenoxoimidazolidin-4-one derivatives. The latter were stereoselectively obtained in two steps from ethyl isoselenocyanatoacetate and isatins.
References
1.

Wu X., Bayle J.H., Olson D., Levine A.J.
Genes and Development,
1993
2.

3.

Ding K., Lu Y., Nikolovska-Coleska Z., Qiu S., Ding Y., Gao W., Stuckey J., Krajewski K., Roller P.P., Tomita Y., Parrish D.A., Deschamps J.R., Wang S.
Journal of the American Chemical Society,
2005
4.

Ivanenkov Y.A., Vasilevski S.V., Beloglazkina E.K., Kukushkin M.E., Machulkin A.E., Veselov M.S., Chufarova N.V., Chernyaginab E.S., Vanzcool A.S., Zyk N.V., Skvortsov D.A., Khutornenko A.A., Rusanov A.L., Tonevitsky A.G., Dontsova O.A., et. al.
Bioorganic and Medicinal Chemistry Letters,
2015
5.

Beloglazkina A.А., Skvortsov D.А., Tafeenko V.A., Majouga А.G., Zyk N.V., Beloglazkina Е.К.
Russian Chemical Bulletin,
2018
6.

Beloglazkina A.A., Karpov N.A., Mefedova S.R., Polyakov V.S., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Majouga A.G., Zyk N.V., Beloglazkina E.K.
Russian Chemical Bulletin,
2019
7.

Wirth T.
Angewandte Chemie - International Edition,
2015
8.

Müller A., Cadenas E., Graf P., Sies H.
Biochemical Pharmacology,
1984
9.

Schewe T.
General Pharmacology The Vascular System,
1995
10.
10.1016/j.mencom.2020.05.020_bib0050
Batist
Cancer Res.,
1986
11.

Ivanenkov Y.A., Veselov M.S., Rezekin I.G., Skvortsov D.A., Sandulenko Y.B., Polyakova M.V., Bezrukov D.S., Vasilevsky S.V., Kukushkin M.E., Moiseeva A.A., Finko A.V., Koteliansky V.E., Klyachko N.L., Filatova L.A., Beloglazkina E.K., et. al.
Bioorganic and Medicinal Chemistry,
2016