Keywords
1,3-diynes
9-azabicyclo[4.2.1]nona-2,4,7-trienes
antitumor activity
azepine-1-carboxylates
catalytic systems
cobalt complexes
cycloaddition
Abstract
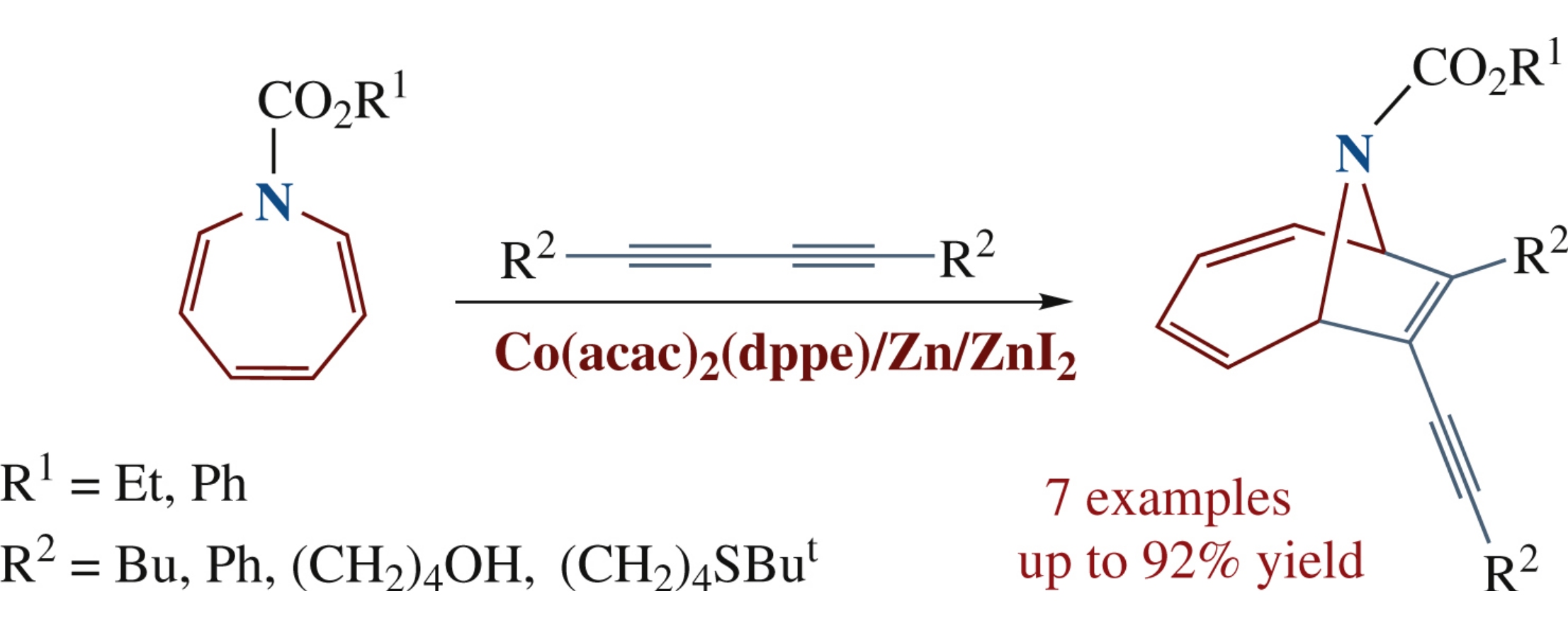
New 7-alkynyl-9-azabicyclo[4.2.1]nona-2,4,7-triene-9carboxylates were obtained upon the Co(acac)2(dppe)/Zn/ZnI2catalyzed [6p+2p]-cycloaddition of 1,3-diynes to ethyl/phenyl azepine-1-carboxylates. The structures of the obtained azacyclic compounds were established by 1D and 2D NMR spectroscopy. The compounds prepared were tested for antitumor activities in vitro against Jurkat, K562, U937 and HL60 cancer cell lines.
References
1.

Dyakonov V.A., Kadikova G.N., Dzhemilev U.M.
Russian Chemical Reviews,
2018
2.

Yu Z., Wang Y., Wang Y.
Chemistry - An Asian Journal,
2010
3.

Rigby J.H.
Tetrahedron,
1999
4.

Lautens M., Klute W., Tam W.
Chemical Reviews,
1996
5.

Frühauf H.
Chemical Reviews,
1997
6.
![Targeted Synthesis of 9-Azabicyclo[4.2.1]nona-2,4,7-trienes by Cobalt(I)-Catalyzed [6π+2π]-Cycloaddition of Alkynes to N -Substituted Azepines and Their Antitumor Activity](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
D'yakonov V.A., Kadikova G.N., Nasretdinov R.N., Dzhemileva L.U., Dzhemilev U.M.
European Journal of Organic Chemistry,
2020
7.

Green M., Heathcock S.M., Turney T.W., Mingos D.M.
Journal of the Chemical Society Dalton Transactions,
1977
8.
![Dienophile cycloaddition to cycloheptatriene and related complexes of tricarbonyliron. X-Ray crystal structures of [Fe(CO)3(η4-C7H7CN)]·C2(CN)4and [ Fe(CO)3(η4-C7H7–C7H7)]](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Chopra S.K., Cunningham D., Kavanagh S., McArdle P., Moran G.
Journal of the Chemical Society Dalton Transactions,
1987
9.

Rigby J.H., Ateeq H.S., Krueger A.C.
Tetrahedron Letters,
1992
10.

Rigby J.H.
Accounts of Chemical Research,
1993
11.
![Metal-promoted higher-order cycloaddition reactions. Stereochemical, regiochemical, and mechanistic aspects of the [6.pi. + 4.pi.] reaction](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Ateeq H.S., Charles N.R., Cuisiat S.V., Ferguson M.D., Henshilwood J.A., Krueger A.C., Ogbu C.O., Short K.M., Heeg M.J.
Journal of the American Chemical Society,
1993
12.
![Chromium(0) promoted [6π+2π] cycloaddition reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Rigby J.H., Ateeq H.S., Charles N.R., Henshilwood J.A., Short K.M., Sugathapala P.M.
Tetrahedron,
1993
13.
![Metal-Mediated [6 + 2] Cycloadditions of Alkynes to Cycloheptatriene and N-Carbethoxyazepine](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chaffee K., Huo P., Sheridan J.B., Barbieri A., Aistars A., Lalancette R.A., Ostrander R.L., Rheingold A.L.
Journal of the American Chemical Society,
1995
14.
![Preparation of a Resin-Based Chromium Catalyst for Effecting [6π + 2π] Cycloaddition Reactions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rigby J.H., Kondratenko M.A., Fiedler C.
Organic Letters,
2000
15.

16.

Devlin J.P., Edwards O.E., Gorham P.R., Hunter N.R., Pike R.K., Stavric B.
Canadian Journal of Chemistry,
1977
17.

Parsons P.J., Camp N.P., Edwards N., Ravi Sumoreeah L.
Tetrahedron,
2000
18.

Hjelmgaard T., Søtofte I., Tanner D.
Journal of Organic Chemistry,
2005
19.
![Application of Intramolecular Enyne Metathesis to the Synthesis of Aza[4.2.1]bicyclics: Enantiospecific Total Synthesis of (+)-Anatoxin-a](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Brenneman J.B., Martin S.F.
Organic Letters,
2004
20.
![Synthesis of the first highly potent bridged nicotinoid 9-Azabicyclo[4.2.1]nona[2,3-c]pyridine (pyrido[3,4-b]homotropane)](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kanne D.B., Ashworth D.J., Cheng M.T., Mutter L.C., Abood L.G.
Journal of the American Chemical Society,
1986
21.

Wright E., Gallagher T., Sharples C.G., Wonnacott S.
Bioorganic and Medicinal Chemistry Letters,
1997
22.
![Synthesis and Nicotinic Binding Studies on Enantiopure Diazine Analogues of the Novel (2-Chloro-5-pyridyl)-9-azabicyclo[4.2.1]non-2-ene UB-165](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Gohlke H., Gündisch D., Schwarz S., Seitz G., Tilotta M.C., Wegge T.
Journal of Medicinal Chemistry,
2002
23.
![Syntheses and evaluation of pyridazine and pyrimidine Containing bioisosteres of (±)-Pyrido[3.4-b]homotropane and Pyrido-[3.4-b]tropane as novel nAChR ligands](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gündisch D., Kämpchen T., Schwarz S., Seitz G., Siegl J., Wegge T.
Bioorganic and Medicinal Chemistry,
2002
24.

Takada N., Iwatsuki M., Suenaga K., Uemura D.
Tetrahedron Letters,
2000
25.

Kigoshi H., Hayashi N., Uemura D.
Tetrahedron Letters,
2001
26.

Malpass J.R., Hemmings D.A., Wallis A.L.
Tetrahedron Letters,
1996
27.

Malpass J.R., Hemmings D.A., Wallis A.L., Fletcher S.R., Patel S.
Journal of the Chemical Society Perkin Transactions 1,
2001
28.

Holladay M.W., Dart M.J., Lynch J.K.
Journal of Medicinal Chemistry,
1997
29.

Lin N., Meyer M.D.
Expert Opinion on Therapeutic Patents,
1998
30.
![The Synthesis of Bicyclo[4.2.1]nona-2,4,7-trienes by [6π + 2π]-Cycloaddition of 1-Substituted 1,3,5-Cycloheptatrienes Catalyzed by Titanium and Cobalt Complexes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
D’yakonov V.A., Kadikova G.N., Nasretdinov R.N., Dzhemileva L.U., Dzhemilev U.M.
Journal of Organic Chemistry,
2019
31.
![First Cobalt(I)-Catalyzed [6 + 2] Cycloadditions of Cycloheptatriene with Alkynes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Achard M., Tenaglia A., Buono G.
Organic Letters,
2005
32.

Hilt G., Smolko K.I.
Synthesis,
2002
33.

Hilt G., Smolko K.I.
Angewandte Chemie - International Edition,
2003
34.

Hilt G., Smolko K.I., Lotsch B.V.
Synlett,
2002
35.
D’yakonov V.A., Kadikova G.N., Gazizullina G.F., Ramazanov I.R., Dzhemileva L.U., Dzhemilev U.M.
Mendeleev Communications,
2019