Keywords
azetidin-2-ones
chemodivergent synthesis
dehydrobromination
diastereomers
α-methylidene-β-lactams
β-bromomethacryloylamides
β-lactams
Abstract
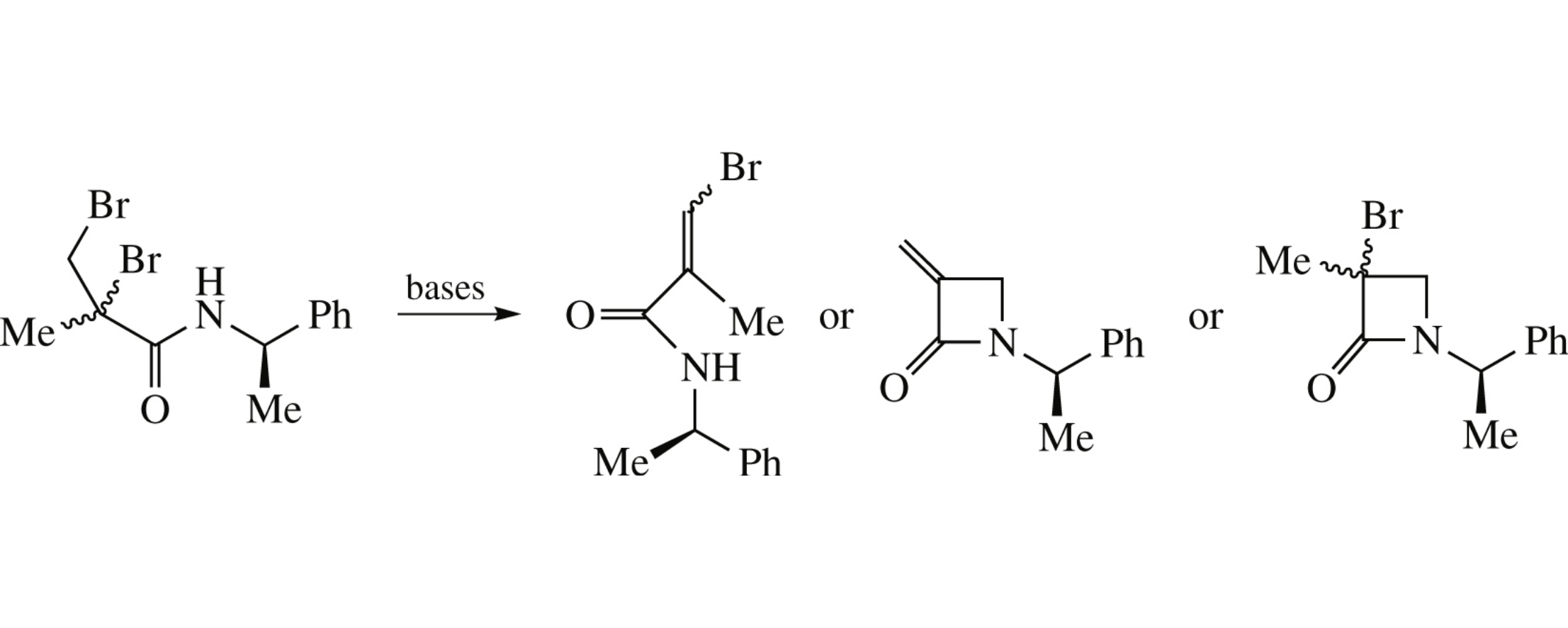
Depending on the base used, reactions of 2,3-dibromo-2-methyl-N-[(1R)-1-phenylethyl]propanamide with DBU, ButOK and NaH in THF lead to β-bromomethacryloylamides, α-methylidene-β-lactam and azetidin-2-ones, respectively.
References
1.

Singh G S
Mini-Reviews in Medicinal Chemistry,
2004
2.

Śnieżek M., Stecko S., Panfil I., Furman B., Chmielewski M.
Journal of Organic Chemistry,
2013
3.

4.
Selezneva N.K., Valiullina Z.R., Khasanova L.S., Gimalova F.A., Biglova R.Z., Miftakhov M.S.
Mendeleev Communications,
2018
5.

Nicolaou K.C., Ueno H., Liu J.-., Nantermet P.G., Yang Z., Renaud J., Paulvannan K., Chadha R.
Journal of the American Chemical Society,
1995
6.

Islami M., Hosseinkhani B., Hosseinkhani S.
Synlett,
2015
7.
Khrenova M.G., Tsirelson V.G.
Mendeleev Communications,
2019
8.

Dao Thi H., Danneels B., Desmet T., Van Hecke K., Van Nguyen T., D'hooghe M.
Asian Journal of Organic Chemistry,
2016
9.

Li W., Liu C., Zhang H., Ye K., Zhang G., Zhang W., Duan Z., You S., Lei A.
Angewandte Chemie - International Edition,
2014
10.

Tavani C., Bianchi L., Giorgi G., Maccagno M., Petrillo G.
European Journal of Organic Chemistry,
2018
11.

Wang X., Meng F., Wang Y., Han Z., Chen Y., Liu L., Wang Z., Ding K.
Angewandte Chemie - International Edition,
2012
12.

Zhu L., Xiong Y., Li C.
Journal of Organic Chemistry,
2014
13.

Beletskaya I.P., Cheprakov A.V.
Chemical Reviews,
2000
14.

Kishi Y.
Pure and Applied Chemistry,
1992
15.
CrysAlis PRO, Agilent Technologies, Yarnton, Oxfordshire, UK, 2014.
16.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
17.
10.1016/j.mencom.2020.05.017_bib0085
Sheldrick
Acta Crystallogr.,
2015