Keywords
diazo transfer
insertion reactions
methoxycarbonylation
SAFE cocktail
β-oxopropionates
Abstract
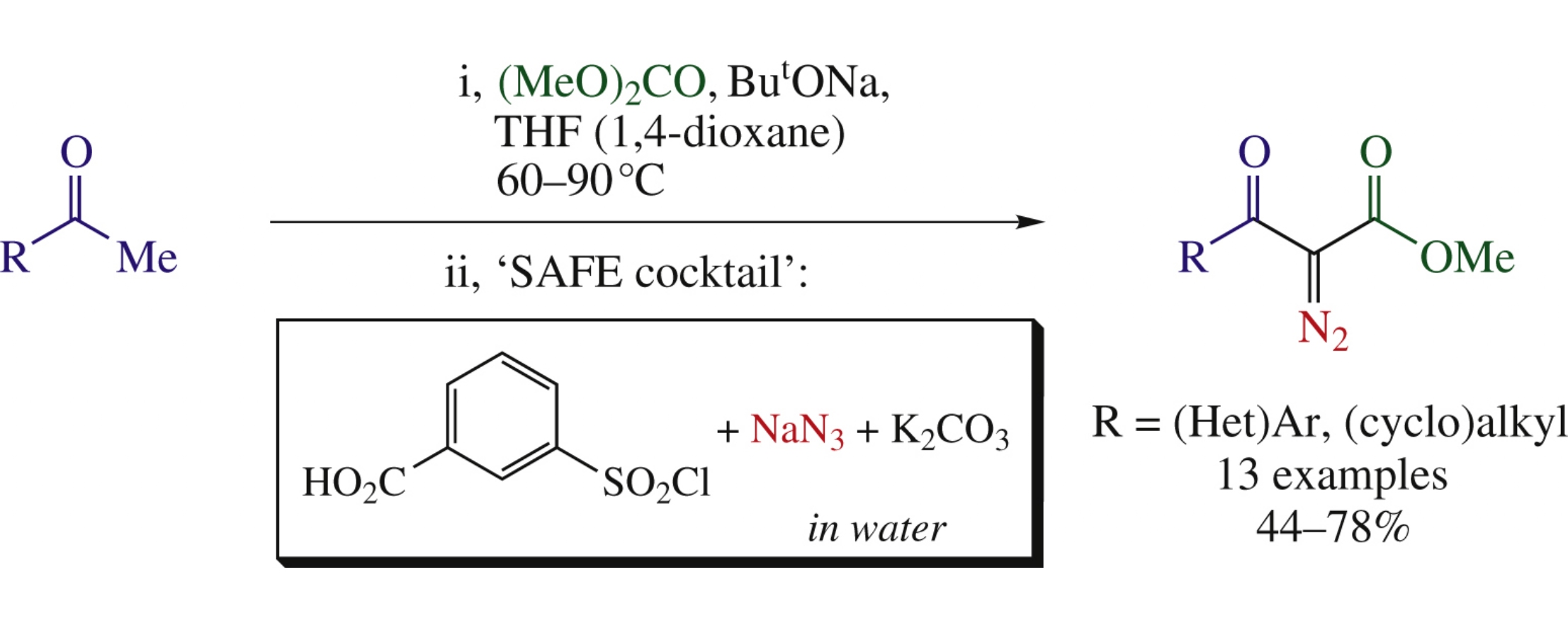
Methyl 2-diazo-3-oxopropionates were obtained by in situ methoxycarbonylation of methyl ketones followed by diazo transfer onto active methylene group of the intermediate β-oxo esters. At the second stage, ‘sulfonyl-azide-free’ (SAFE) diazo transfer protocol in aqueous medium was employed.
References
1.

Ford A., Miel H., Ring A., Slattery C.N., Maguire A.R., McKervey M.A.
Chemical Reviews,
2015
2.
10.1016/j.mencom.2020.05.016_bib0010
Doyle
Modern Catalytic Methods for Organic Synthesis with Diazo Compounds,
1998
3.
10.1016/j.mencom.2020.05.016_bib0015
Regitz
Chem.,
1964
4.
10.1016/j.mencom.2020.05.016_bib0020
Regitz
Diazo Compounds: Properties and Synthesis,
1986
5.

6.

Curphey T.J.
Organic Preparations and Procedures International,
1981
7.

Dar’in D., Kantin G., Krasavin M.
Chemical Communications,
2019
8.
![A novel approach to biologically relevant oxazolo[5,4-d]pyrimidine-5,7-diones via readily available diazobarbituric acid derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gecht M., Kantin G., Dar'in D., Krasavin M.
Tetrahedron Letters,
2019
9.

Shershnev I., Dar'in D., Chuprun S., Kantin G., Bakulina O., Krasavin M.
Tetrahedron Letters,
2019
10.

Dar’in D., Kantin G., Krasavin M.
Synthesis,
2019
11.

Marcoux D., Goudreau S.R., Charette A.B.
Journal of Organic Chemistry,
2009
12.

Phun L.H., Patil D.V., Cavitt M.A., France S.
Organic Letters,
2011
13.

Yasui K., Kato T., Kojima K., Nagasawa K.
Chemical Communications,
2015