Keywords
catalysis
heterocyclization
hexaazadibenzotetracenes
polyazapolycycles
tetraazatetracenes
trans-1,2-diaminocyclohexane
Abstract
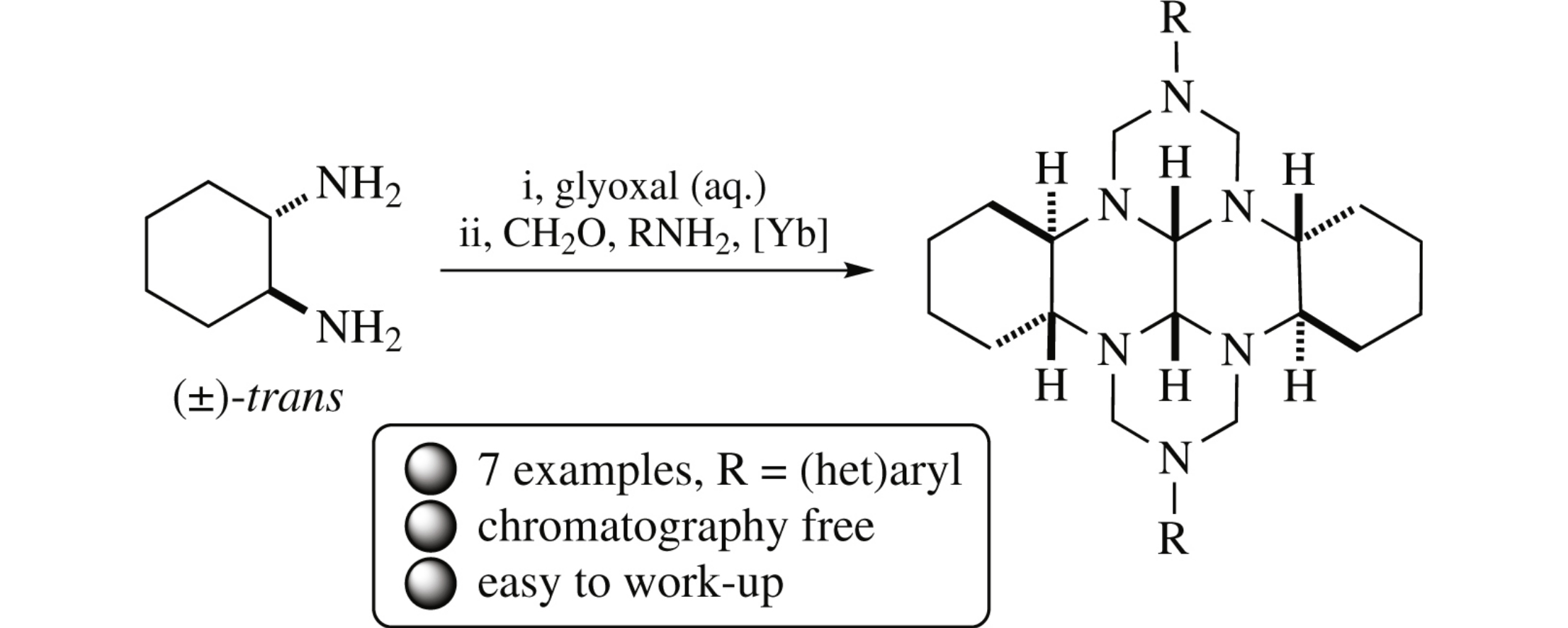
A one-pot reaction between (±)-trans-1,2-diaminocyclohexane and glyoxal affords intermediate perhydro-1,6,7,12-tetraazatetracene whose further treatment with formaldehyde and (het)arylamines in the presence of YbCl3·6H2O gives 2,9-di(het)aryl substituted (3bR*,7aR*,10bR*,14aR*)-perhydro-2,3a,7b,9,10a,14b-hexaazadibenzo[fg,op]tetracenes with cis-junction of the cycles along the C(14c)–C(14d) bond.
References
1.
P. Neumann, A. Aumueller, H. Trauth, US Patent 4,904,779, 1990.
2.

Rakhimova E., Kirsanov V., Mescheryakova E., Khalilov L., Ibragimov A., Dzhemilev U.
Synlett,
2018
3.

Rakhimova E.B., Kirsanov V.Y., Meshcheryakova E.S., Khalilov L.M., Kutepov B.I., Ibragimov A.G., Dzhemilev U.M.
Tetrahedron,
2017
4.

Rakhimova E.B., Kirsanov V.Y., Mescheryakova E.S., Khalilov L.M., Ibragimov A.G., Dzhemileva L.U., D’yakonov V.A., Dzhemilev U.M.
ACS Medicinal Chemistry Letters,
2019
5.

Dragoun M., Günther T., Frias C., Berkessel A., Prokop A.
Journal of Cancer Research and Clinical Oncology,
2018
6.

Khokhar A.R., Al-Baker S., Shamsuddin S., Siddik Z.H.
Journal of Medicinal Chemistry,
1997
7.

Morales F., Ramírez A., Morata-Tarifa C., Navarro S.A., Marchal J.A., Campos J.M., Conejo-García A.
Future Medicinal Chemistry,
2017
8.

Omer K.H., Seliman A.A., Altaf M., Casagrande N., Aldinucci D., Altuwaijri S., Isab A.A.
Polyhedron,
2015
9.

Iwanejko J., Wojaczyńska E., Trynda J., Maciejewska M., Wietrzyk J., Kochel A., Wojaczyński J.
Tetrahedron,
2017
10.

Rakhimova E.B., Kirsanov V.Y., Ibragimov A.G., Dzhemilev U.M.
Russian Journal of Organic Chemistry,
2018
11.
![Synthesis, Structure, and Biological Activity of 2,6-Disubstituted 2,3a,4a,6,7a,8a-Hexaazaperhydrocyclopenta[ def ]fluorene-4-thioxo-8-ones](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Barsegyan Y., Baranov V., Kravchenko A., Strelenko Y., Anikina L., Karnoukhova V., Kolotyrkina N.
Synthesis,
2018
12.

Yamaguchi T., Ito S., Iwase Y., Watanabe K., Harano K.
Heterocycles,
1999
13.

Yamaguchi T., Ito S., Iwase Y., Watanabe K., Harano K.
Heterocycles,
2000
14.

Désogère P., Bernhard C., Goze C., Penouilh M., Rousselin Y., Denat F.
European Journal of Organic Chemistry,
2013
15.

Lei X., Jalla A., Abou Shama M., Stafford J., Cao B.
Synthesis,
2015
16.
10.1016/j.mencom.2020.05.015_bib0080
CrysAlis
Agilent Technologies,
2012
17.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
18.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
19.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015