Keywords
DFT
silver–gold alloy
surface enrichment
surface segregation
water adsorption
Abstract
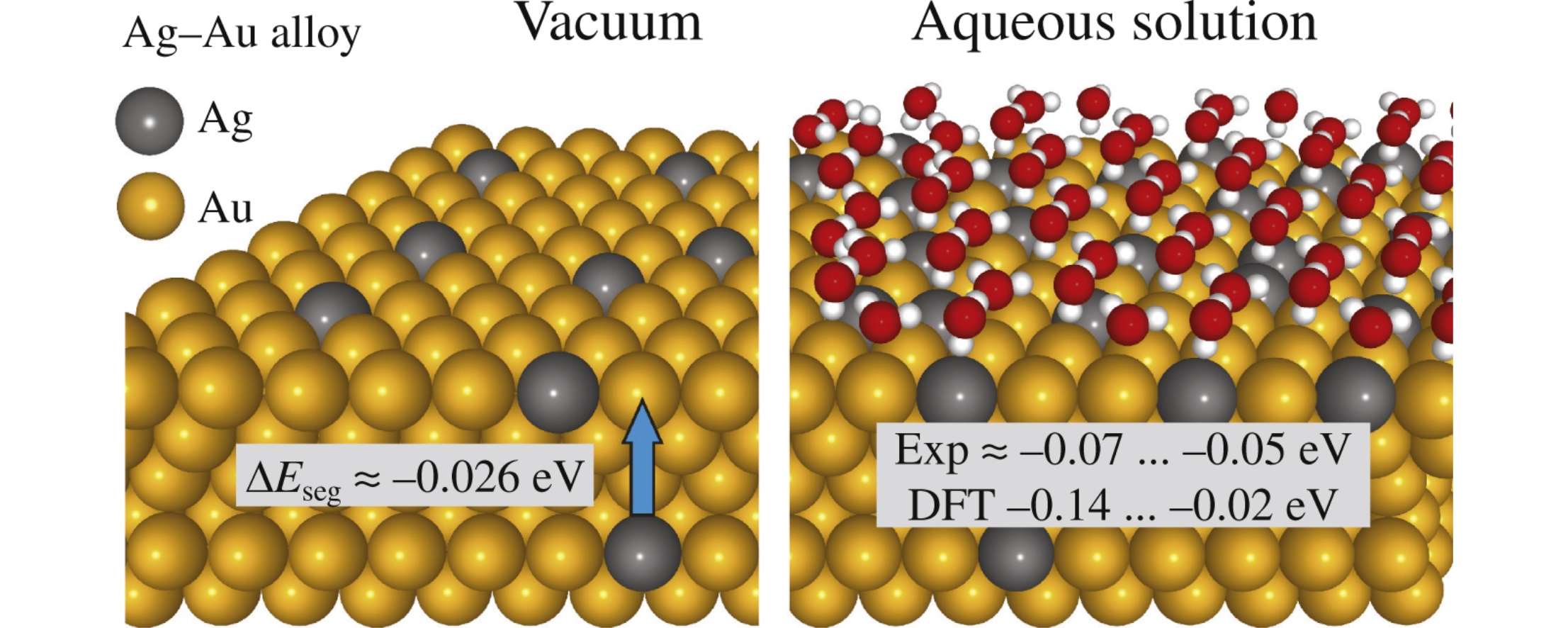
Segregation energy of Ag atoms in Ag(3%) and Ag(10%) silver–gold alloys is more than two times decreased upon the transition from vacuum to aqueous solutions. The DFT calculations using the (111) face of Au as an example have revealed that the effect of OH radical adsorbed together with H2O molecules on the surface Ag atoms on the segregation energy is comparable to that of a water monolayer. Both experimental and model dependences of the segregation energy and the coverage with Ag on the cluster charge and electrode potential were analyzed.
References
1.

2.

Kelley M.J., Swartzfager D.G., Sundaram V.S.
Journal of Vacuum Science and Technology,
1979
3.

Meinel K., Klaua M., Bethge H.
phys stat sol (a),
1988
4.

Overbury S.H., Somorjai G.A.
Surface Science,
1976
5.

King T.S., Donnelly R.G.
Surface Science,
1985
6.

King T.S., Donnelly R.G.
Surface Science,
1984
7.

Manzhos R.A., Krivenko A.G., Doronin S.V., Choba M.A., Safonov V.A.
Journal of Electroanalytical Chemistry,
2013
8.
Karavanov A.N., Batirev I.G.
Mendeleev Communications,
1993
9.
Stakheev A.Y., Smirnova N.S., Krivoruchenko D.S., Baeva G.N., Mashkovsky I.S., Yakushev I.A., Vargaftik M.N.
Mendeleev Communications,
2017
10.
Mashkovsky I.S., Smirnova N.S., Markov P.V., Baeva G.N., Bragina G.O., Bukhtiyarov A.V., Prosvirin I.P., Stakheev A.Y.
Mendeleev Communications,
2018
11.

Déronzier T., Morfin F., Lomello M., Rousset J.
Journal of Catalysis,
2014
12.

Zugic B., Wang L., Heine C., Zakharov D.N., Lechner B.A., Stach E.A., Biener J., Salmeron M., Madix R.J., Friend C.M.
Nature Materials,
2016
13.

Stamenković V., Schmidt T.J., Ross P.N., Marković N.M.
Journal of Electroanalytical Chemistry,
2003
14.

Cui C., Gan L., Heggen M., Rudi S., Strasser P.
Nature Materials,
2013
15.

Friesen C., Dimitrov N., Cammarata R.C., Sieradzki K.
Langmuir,
2001
16.

Bozzolo G., Garcés J.E., Derry G.N.
Surface Science,
2007
17.

Qi W.H., Lee S.T.
Journal of Physical Chemistry C,
2010
18.

Deng L., Hu W., Deng H., Xiao S., Tang J.
Journal of Physical Chemistry C,
2011
19.

20.

Doronin S.V., Manzhos R.A., Krivenko A.G.
Electrochemistry Communications,
2015
21.
10.1016/j.mencom.2020.05.008_bib0105
McLean
Grain Boundaries in Metals,
1957
22.

Dowben P.A., Miller A.H., Vook R.W.
Gold Bulletin,
1987
23.

Creuze J., Berthier F., Tétot R., Legrand B.
Surface Science,
2003
24.

Roudgar A., Groß A.
Chemical Physics Letters,
2005
25.

Sánchez C.G.
Surface Science,
2003
26.

Chen J., Nie L., Yao S.
Journal of Electroanalytical Chemistry,
1996
27.

Diaz-Morales O., Calle-Vallejo F., de Munck C., Koper M.T.
Chemical Science,
2013
28.

Giannozzi P., Baroni S., Bonini N., Calandra M., Car R., Cavazzoni C., Ceresoli D., Chiarotti G.L., Cococcioni M., Dabo I., Dal Corso A., de Gironcoli S., Fabris S., Fratesi G., Gebauer R., et. al.
Journal of Physics Condensed Matter,
2009
29.

Perdew J.P., Ruzsinszky A., Csonka G.I., Vydrov O.A., Scuseria G.E., Constantin L.A., Zhou X., Burke K.
Physical Review Letters,
2008
30.

Vanderbilt D.
Physical Review B,
1990
31.

Liu S., White M.G., Liu P.
Journal of Physical Chemistry C,
2016
32.

Karlberg G.S.
Physical Review B,
2006
33.

Lin X., Groß A.
Surface Science,
2012
34.

Feng Y.J., Bohnen K.P., Chan C.T.
Physical Review B,
2005
35.

Wynblatt P., Ku R.C.
Surface Science,
1977