Keywords
arylboronic acids
diarylborinic acids
homogeneous catalysis
Hydrogen storage
hydrogenation
organoboron compounds
organofluorine compounds
triarylboranes
Abstract
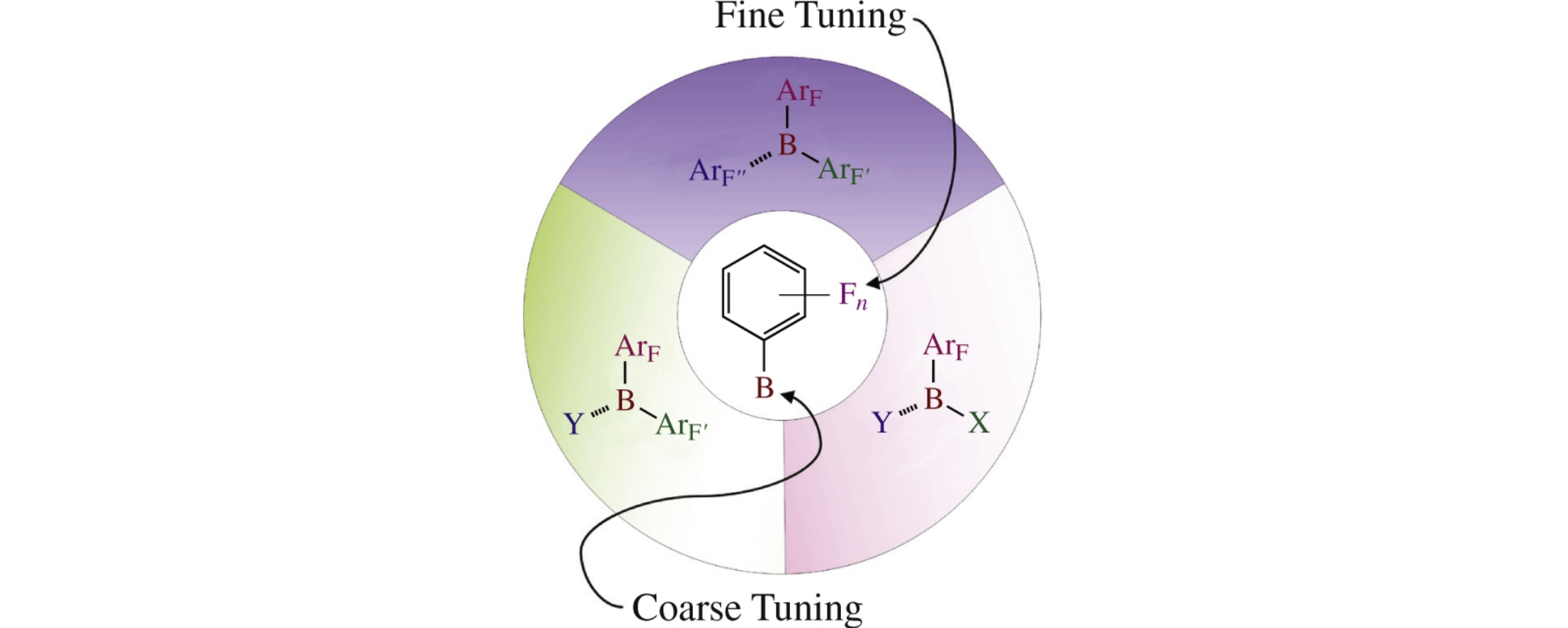
The review analyzes actual data on the use of polyfluorinated triarylboranes, diarylborinic and arylboronic acids as catalysts for organic reactions, e.g., hydrogenation of unsaturated bonds and alkylation of aromatic compounds.
References
1.
10.1016/j.mencom.2020.05.002_bib0005
Stephan
Frustrated Lewis Pairs I: Uncovering and Understanding,
2013
2.

Liu L., Lukose B., Jaque P., Ensing B.
Green Energy and Environment,
2019
3.

Sumerin V., Schulz F., Atsumi M., Wang C., Nieger M., Leskelä M., Repo T., Pyykkö P., Rieger B.
Journal of the American Chemical Society,
2008
4.

Adonin N.Y., Bardin V.V.
Russian Chemical Reviews,
2010
5.

Welch G.C., Stephan D.W.
Journal of the American Chemical Society,
2007
6.
10.1016/j.mencom.2020.05.002_bib0030
Boronic Acids: Preparation and Applications in Organic Synthesis, Medicine and Materials,
2011
7.

8.

Massey A.G., Park A.J.
Journal of Organometallic Chemistry,
1964
9.

Frohn H.-., Franke H., Fritzen P., Bardin V.V.
Journal of Organometallic Chemistry,
2000
10.

Frohn H.-., Adonin N.Y., Bardin V.V., Starichenko V.F.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2002
11.

Süsse L., Hermeke J., Oestreich M.
Journal of the American Chemical Society,
2016
12.

Gyömöre Á., Bakos M., Földes T., Pápai I., Domján A., Soós T.
ACS Catalysis,
2015
13.

Sumerin V., Chernichenko K., Nieger M., Leskelä M., Rieger B., Repo T.
Advanced Synthesis and Catalysis,
2011
14.

Mewald M., Fröhlich R., Oestreich M.
Chemistry - A European Journal,
2011
15.

Ullrich M., Lough A.J., Stephan D.W.
Journal of the American Chemical Society,
2008
16.

Ashley A.E., Herrington T.J., Wildgoose G.G., Zaher H., Thompson A.L., Rees N.H., Krämer T., O’Hare D.
Journal of the American Chemical Society,
2011
17.

Politanskaya L.V., Selivanova G.A., Panteleeva E.V., Tretyakov E.V., Platonov V.E., Nikul’shin P.V., Vinogradov A.S., Zonov Y.V., Karpov V.M., Mezhenkova T.V., Vasilyev A.V., Koldobskii A.B., Shilova O.S., Morozova S.M., Burgart Y.V., et. al.
Russian Chemical Reviews,
2019
18.

Patrick E.A., Piers W.E.
Chemical Communications,
2020
19.

Li X., Tian J., Liu N., Tu X., Zeng N., Wang X.
Angewandte Chemie - International Edition,
2019
20.

Meng W., Feng X., Du H.
Accounts of Chemical Research,
2017
21.

Khan I., Reed-Berendt B.G., Melen R.L., Morrill L.C.
Angewandte Chemie - International Edition,
2018
22.

Rosorius C., Möricke J., Wibbeling B., McQuilken A.C., Warren T.H., Daniliuc C.G., Kehr G., Erker G.
Chemistry - A European Journal,
2015
23.

Ghattas G., Chen D., Pan F., Klankermayer J.
Dalton Transactions,
2012
24.

Chen D., Leich V., Pan F., Klankermayer J.
Chemistry - A European Journal,
2012
25.

Jiang C., Blacque O., Fox T., Berke H.
Dalton Transactions,
2011
26.

Chen D., Wang Y., Klankermayer J.
Angewandte Chemie - International Edition,
2010
27.

Du H., Ren X., Han C., Feng X.
Synlett,
2017
28.

Li S., Meng W., Du H.
Organic Letters,
2017
29.

Liu Y., Du H.
Journal of the American Chemical Society,
2013
30.

MASSEY A.G., PARK A.J.
1986
31.

Beckett M.A., Strickland G.C., Holland J.R., Sukumar Varma K.
Polymer,
1996
32.

Beckett M.A., Brassington D.S., Coles S.J., Hursthouse M.B.
Inorganic Chemistry Communication,
2000
33.

Mayer U., Gutmann V., Gerger W.
Monatshefte fur Chemie,
1975
34.

Gutmann V.
Coordination Chemistry Reviews,
1976
35.

Morgan M.M., Marwitz A.J., Piers W.E., Parvez M.
Organometallics,
2012
36.

Greb L., Daniliuc C., Bergander K., Paradies J.
Angewandte Chemie - International Edition,
2013
37.

Frohn H.-., Adonin N.Y., Bardin V.V., Starichenko V.F.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2002
38.

Zarzeczańska D., Adamczyk‐Woźniak A., Kulpa A., Ossowski T., Sporzyński A.
European Journal of Inorganic Chemistry,
2017
39.

Gozdalik J.T., Adamczyk-Woźniak A., Sporzyński A.
Pure and Applied Chemistry,
2017
40.

Cox P.A., Reid M., Leach A.G., Campbell A.D., King E.J., Lloyd-Jones G.C.
Journal of the American Chemical Society,
2017
41.

Diemoz K.M., Franz A.K.
Journal of Organic Chemistry,
2018
42.

Beringhelli T., D'Alfonso G., Donghi D., Maggioni D., Mercandelli P., Sironi A.
Organometallics,
2003
43.

Beringhelli T., D'Alfonso G., Donghi D., Maggioni D., Mercandelli P., Sironi A.
Organometallics,
2004
44.

Beringhelli T., D'Alfonso G., Donghi D., Maggioni D., Mercandelli P., Sironi A.
Organometallics,
2007
45.

Donghi D., Maggioni D., Beringhelli T., D'Alfonso G., Mercandelli P., Sironi A.
European Journal of Inorganic Chemistry,
2008
46.

Britovsek G.J., Ugolotti J., Hunt P., White A.J.
Chemical Communications,
2006
47.

Donghi D., Maggioni D., Beringhelli T., D'Alfonso G.
European Journal of Inorganic Chemistry,
2008
48.

Maggioni D., Beringhelli T., D' Alfonso G., Malatesta M.C., Mercandelli P., Donghi D.
Zeitschrift fur Physikalische Chemie,
2013
49.

Welch G.C., Juan R.R., Masuda J.D., Stephan D.W.
Science,
2006
50.

Ullrich M., Lough A.J., Stephan D.W.
Organometallics,
2010
51.

Zhang Z., Du H.
Angewandte Chemie - International Edition,
2014
52.

Dorkó É., Kótai B., Földes T., Gyömöre Á., Pápai I., Soós T.
Journal of Organometallic Chemistry,
2017
53.

Sitte N.A., Bursch M., Grimme S., Paradies J.
Journal of the American Chemical Society,
2018
54.

Scott D.J., Fuchter M.J., Ashley A.E.
Angewandte Chemie - International Edition,
2014
55.

Scott D.J., Fuchter M.J., Ashley A.E.
Journal of the American Chemical Society,
2014
56.

Zhu X., Du H.
Organic Letters,
2015
57.

Letsinger R.L., Dandegaonker S., Vullo W.J., Morrison J.D.
Journal of the American Chemical Society,
1963
58.

Ishihara K., Ohara S., Yamamoto H.
Journal of Organic Chemistry,
1996
59.

Estopiñá‐Durán S., Donnelly L.J., Mclean E.B., Hockin B.M., Slawin A.M., Taylor J.E.
Chemistry - A European Journal,
2019
60.

Manhas S., Taylor M.S.
Carbohydrate Research,
2018
61.

Liu S., Yang Y., Liu X., Ferdousi F.K., Batsanov A.S., Whiting A.
European Journal of Organic Chemistry,
2013
62.
10.1016/j.mencom.2020.05.002_bib0310
Khaskin
Zh. Prikl. Khim.,
1960
63.

Ulrich H., Tucker B., Richter R.
Journal of Organic Chemistry,
1978
64.

El Dine T.M., Rouden J., Blanchet J.
Chemical Communications,
2015
65.

Arkhipenko S., Sabatini M.T., Batsanov A.S., Karaluka V., Sheppard T.D., Rzepa H.S., Whiting A.
Chemical Science,
2018
66.

Verdelet T., Ward R.M., Hall D.G.
European Journal of Organic Chemistry,
2017
67.

Tale R.H., Patil K.M., Dapurkar S.E.
Tetrahedron Letters,
2003
68.

Ishihara K., Kurihara H., Yamamoto H.
Journal of Organic Chemistry,
1997
69.

Zheng H., Lejkowski M., Hall D.G.
Chemical Science,
2011
70.

Zheng H., Lejkowski M., Hall D.G.
Tetrahedron Letters,
2013
71.

Zheng H., Hall D.G.
Tetrahedron Letters,
2010
72.
![Mild arylboronic acid catalyzed selective [4 + 3] cycloadditions: access to cyclohepta[b]benzofurans and cyclohepta[b]indoles](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Cao K., Bian H., Zheng W.
Organic and Biomolecular Chemistry,
2015
73.

Zheng W., Tang W., Cao K., Meng S.
Synthesis,
2017
74.

Dimakos V., Singh T., Taylor M.S.
Organic and Biomolecular Chemistry,
2016
75.

Liu J., Yao H., Wang C.
ACS Catalysis,
2018
76.

Yao H., Liu J., Wang C.
Organic and Biomolecular Chemistry,
2019
77.

Tatina M.B., Mengxin X., Peilin R., Judeh Z.M.
Beilstein Journal of Organic Chemistry,
2019
78.

Tatina M.B., Moussa Z., Xia M., Judeh Z.M.
Chemical Communications,
2019
79.

McCubbin J.A., Hosseini H., Krokhin O.V.
Journal of Organic Chemistry,
2010
80.

Ricardo C.L., Mo X., McCubbin J.A., Hall D.G.
Chemistry - A European Journal,
2015
81.

McCubbin J.A., Krokhin O.V.
Tetrahedron Letters,
2010
82.

Ang H.T., Rygus J.P., Hall D.G.
Organic and Biomolecular Chemistry,
2019
83.

Wolf E., Richmond E., Moran J.
Chemical Science,
2015
84.

McCubbin J., Nassar C., Krokhin O.
Synthesis,
2011
85.

Zheng H., Ghanbari S., Nakamura S., Hall D.G.
Angewandte Chemie - International Edition,
2012