Keywords
aluminum trichloride
benzene
benzophenone
one-pot synthesis
Schiff bases
tetrachloromethane
Abstract
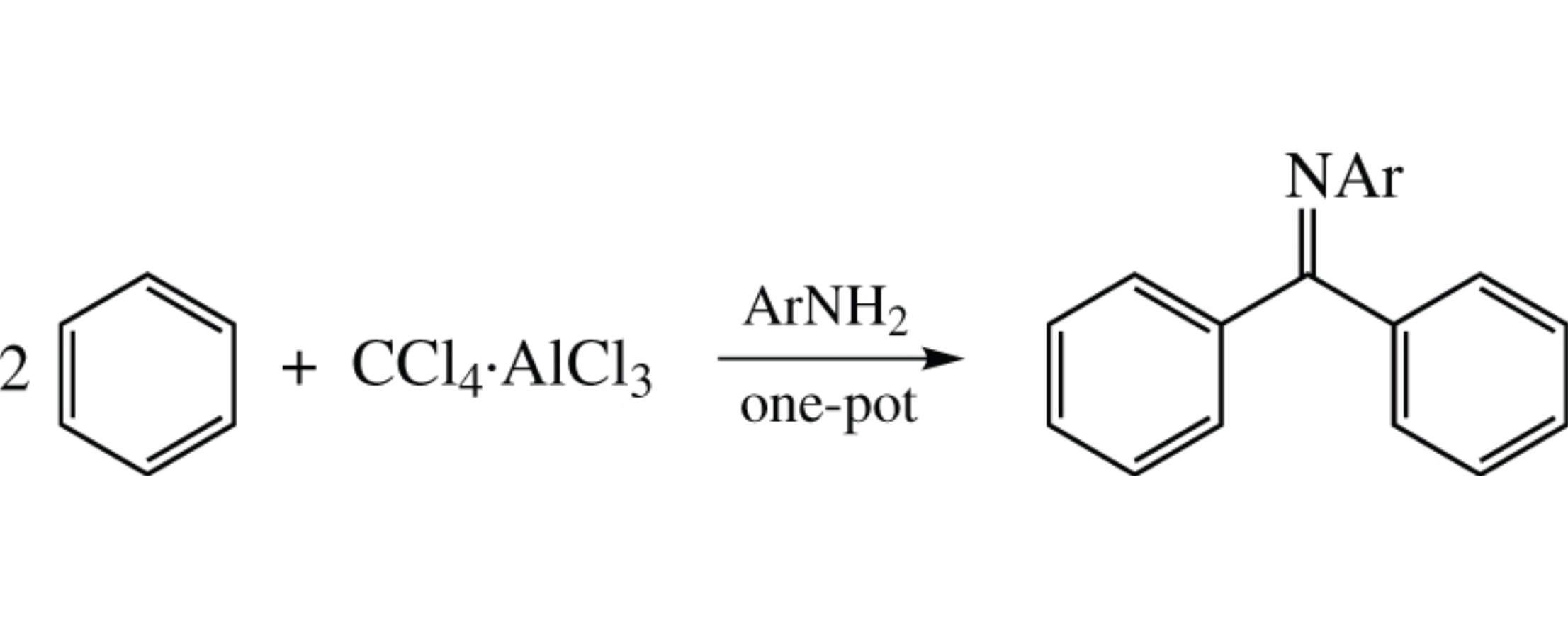
A simple and efficient one-pot synthesis of benzophenone Schiff bases from benzene, CCl4 and aromatic amines was developed based on the the reaction of benzene with CCl4·AlCl3 complex. This method affords Ph2CCl2 as well as the products of its subsequent reaction with aromatic amines, benzophenone Schiff bases, selectively and in good yields.
References
1.
10.1016/j.mencom.2020.03.037_bib0005
Khosa
Pak. J. Pharm. Sci.,
2015
2.
10.1016/j.mencom.2020.03.037_sbref0010a
Makioka
Bull. Soc. Chim. Fr.,
1997
3.

Imaoka T., Kawana Y., Tsuji M., Yamamoto K.
Chemistry - A European Journal,
2010
4.
J. Wang, Z. Gao, X. Zhang, H Liu, J. Ma, J. Chen, X. Li, C. Li, J. Hu, X. Cai and J. Ma, CN Patent 105085730A, 2015. (in Chinese).
5.

Wang R., Ma M., Gong X., Fan X., Walsh P.J.
Organic Letters,
2018
6.

van As D.J., Connell T.U., Brzozowski M., Scully A.D., Polyzos A.
Organic Letters,
2018
7.

Bidusenko I.A., Schmidt E.Y., Ushakov I.A., Trofimov B.A.
European Journal of Organic Chemistry,
2018
8.

Liu Y., Yang Q., Hao D., Zhang W.
Australian Journal of Chemistry,
2012
9.

Wolfe J.P., Singer R.A., Yang B.H., Buchwald S.L.
Journal of the American Chemical Society,
1999
10.
![Nickel-Catalyzed C−N Cross-Coupling of Primary Imines with Subsequent In Situ [2+2] Cycloaddition or Alkylation](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Power D.J., Jones K.D., Kampmann S.S., Flematti G.R., Stewart S.G.
Asian Journal of Organic Chemistry,
2017
11.

Strekowski L., Cegla M.T., Harden D.B., Kong S.B.
Journal of Organic Chemistry,
1989
12.

Liu S., Yu Y., Liebeskind L.S.
Organic Letters,
2007
13.
Bazhin D.N., Kudyakova Y.S., Slepukhin P.A., Burgart Y.V., Malysheva N.N., Kozitsina A.N., Ivanova A.V., Bogomyakov A.S., Saloutin V.I.
Mendeleev Communications,
2018
14.
10.1016/j.mencom.2020.03.037_bib0020
Schiff
Ann. Chem. Pharm.,
1864
15.

Gomberg M.
Berichte der deutschen chemischen Gesellschaft,
1900
16.

Boeseken I.
Recueil des Travaux Chimiques des Pays-Bas et de la Belgique,
1905
17.

Gomberg M., Jickling R.L.
Journal of the American Chemical Society,
1915
18.

Gomberg M.
Journal of the American Chemical Society,
1913
19.

Wallace C.H., Willard J.E.
Journal of the American Chemical Society,
1950
20.

Belen"kii L.I., Chuvylkin N.D., Suslov I.A.
Russian Chemical Bulletin,
2001
21.

22.

Raabe D., Hörhold H.-.
Journal für praktische Chemie,
1987
23.
10.1016/j.mencom.2020.03.037_sbref0040c
Prakash
Three Carbon–Heteroatom Bonds: Thio-, Seleno-, and Tellurocarboxylic Acids and Derivatives; Imidic Acids and Derivatives; Ortho Acid Derivatives (Science of Synthesis, category 3. Compounds with Four and Three Carbon–Heteroatom Bonds, vol. 22),,
2005
24.
10.1016/j.mencom.2020.03.037_sbref0045a
Friedel
Ann. Chim. Phys.,
1887
25.
10.1016/j.mencom.2020.03.037_sbref0045b
Friedel
Bull. Soc. Chim. Fr.,
1887
26.

Zarkadis A.K., Neumann W.P., Uzick W.
Chemische Berichte,
1985
27.

Brand K., Vogt W.
Journal für praktische Chemie,
1924
28.

Andrews L.J., Kaeding W.W.
Journal of the American Chemical Society,
1951
29.

30.

Lee C., Lee S., Min B., Kim D., Jun C.
Organic Letters,
2018
31.

Baran J.R., Lagow R.J.
Journal of the American Chemical Society,
1990
32.

Schmidt M., Ungvári J., Glöde J., Dobner B., Langner A.
Bioorganic and Medicinal Chemistry,
2007
33.

Kawata S., Ashizawa S., Hirama M.
Journal of the American Chemical Society,
1997
34.

Alig L., Alsenz J., Andjelkovic M., Bendels S., Bénardeau A., Bleicher K., Bourson A., David-Pierson P., Guba W., Hildbrand S., Kube D., Lübbers T., Mayweg A.V., Narquizian R., Neidhart W., et. al.
Journal of Medicinal Chemistry,
2008
35.

Gattermann L.
Berichte der deutschen chemischen Gesellschaft,
1895
36.
10.1016/j.mencom.2020.03.037_sbref0065b
Stanforth
2005
37.

38.

Olah G.A., Rasul G., Heiliger L., Prakash G.K.
Journal of the American Chemical Society,
1996
39.

Vancik H., Percac K., Sunko D.E.
Journal of the American Chemical Society,
1990
40.

Akhrem I., Orlinkov A., Vol'pin M.
Journal of the Chemical Society Chemical Communications,
1993
41.

Akhrem I., Orlinkov A.
Chemical Reviews,
2007
42.
![Regioselective additions of grignard and lithium reagents to 2-[(benzylidene)aminoi]benzonitrile and 2-[(diphenylmethylene)amino]benzonitrile](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Strekowski L., Cegla M.T., Harden D.B., Mokrosz J.L., Mokrosz M.J.
Tetrahedron Letters,
1988
43.

Knorr R.
Chemische Berichte,
1980
44.

Okubo M., Ueda S.
Bulletin of the Chemical Society of Japan,
1980
45.

\={O}kubo M., Ueda S.
Bulletin of the Chemical Society of Japan,
1979
46.
pKa Table, http://evans.rc.fas.harvard.edu/pdf/evans_pKa_table.pdf.
47.

Butcher K.J., Hurst J.
Tetrahedron Letters,
2009
48.

Jasien P.G.
The Journal of Physical Chemistry,
1992