Keywords
aryl bromides
arylation
cinnamyl group.
cross-coupling
dihydrofuran-2(3H)-ones
Heck reaction
tri(o-tolyl)phosphine
Abstract
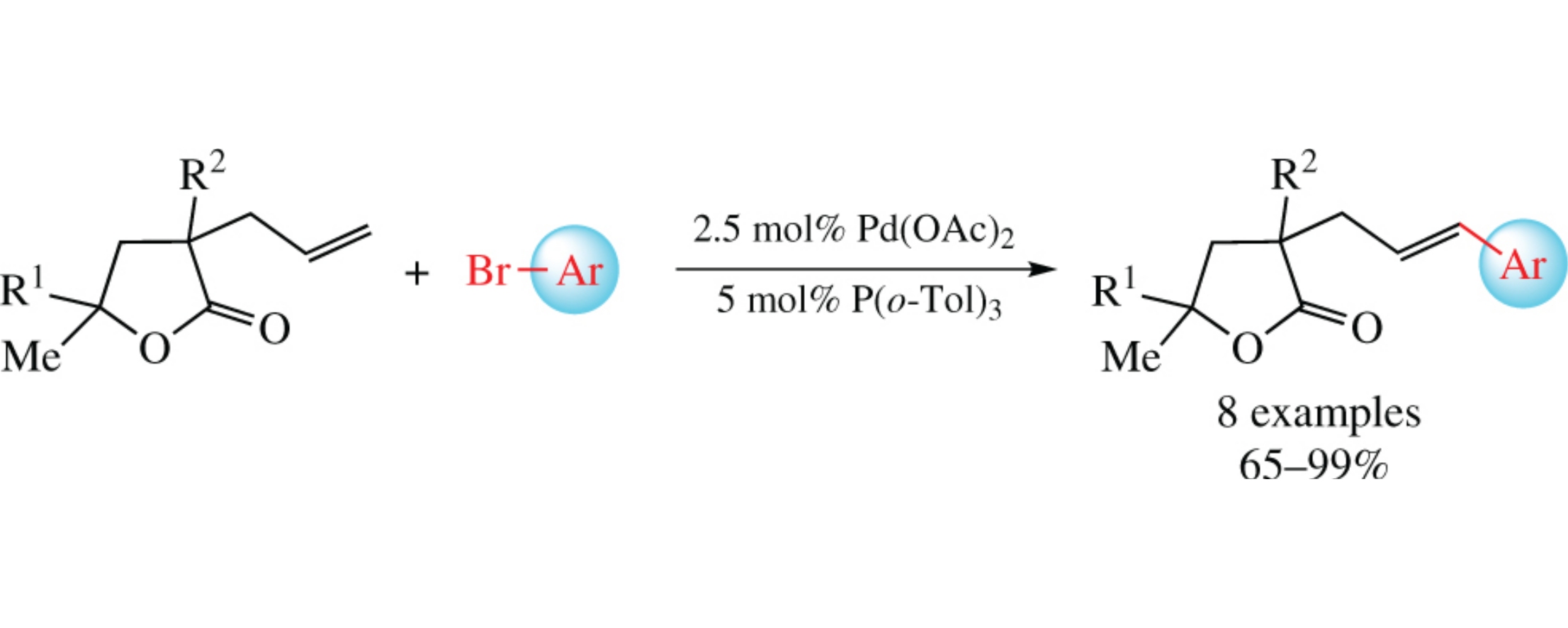
The Heck cross-coupling of 3-allyldihydrofuran-2(3H)-ones with aryl bromides [Pd(OAc)3, P(o-Tol)3] afforded the corresponding 3-cinnamyl derivatives. The structure of the products was confirmed by NMR spectroscopy, mass spectrometry and X-ray diffraction.
References
1.

Whitcombe N.J., Hii K.K., Gibson S.E.
Tetrahedron,
2001
2.
10.1016/j.mencom.2020.03.036_bib0010
Anctil
Metal-Catalized Cross-Coupling Reactions and More,
2014
3.

Murata T., Nakano M., Miyase T., Yoshizaki F.
Chemical and Pharmaceutical Bulletin,
2014
4.

Zhao M., Cheng S., Yuan W., Dong J., Huang K., Sun Z., Yan P.
Chemical and Pharmaceutical Bulletin,
2015
5.

Hu C., Xiong J., Li J., Gao L., Wang W., Cheng K., Yang G., Li J., Hu J.
European Journal of Organic Chemistry,
2016
6.

Fomekong Fotsop D., Roussi F., Le Callonec C., Bousserouel H., Litaudon M., Guéritte F.
Tetrahedron,
2008
7.

Arai T., Toda Y., Kato K., Miyamoto K., Hasegawa T., Yamada K., Ueda J., Hasegawa K., Inoue T., Shigemori H.
Tetrahedron,
2013
8.

Cheng Y., Lo I., Shyur L., Yang C., Hsu Y., Su J., Lu M., Chiou S., Lan C., Wu Y., Chang F.
Tetrahedron,
2015
9.

Yokoe H., Noboru K., Manabe Y., Yoshida M., Shibata H., Shishido K.
Chemical and Pharmaceutical Bulletin,
2012
10.

Jian Y., Wu Y.
Organic and Biomolecular Chemistry,
2010
11.

Francuz J., Kovačević I., Popsavin M., Benedeković G., Zelenović B.S., Kojić V., Jakimov D., Aleksić L., Bogdanović G., Srdić-Rajić T., Lončar E., Rodić M.V., Divjaković V., Popsavin V.
European Journal of Medicinal Chemistry,
2017
12.

Li C., Han J.
Synlett,
2015
13.

Casey M., McCarthy R.
Synlett,
2011
14.

Markovič M., Ďuranová M., Koóš P., Szolcsányi P., Gracza T.
Tetrahedron,
2013
15.

Lloyd M.G., D'Acunto M., Taylor R.J., Unsworth W.P.
Tetrahedron,
2015
16.

Grabarczyk M., Wińska K., Mączka W., Żarowska B., Maciejewska G., Dancewicz K., Gabryś B., Anioł M.
Tetrahedron,
2016
17.

Petrosyan A., Ghochikyan T.V., Ejaz S.A., Mardiyan Z.Z., Khan S.U., Grigoryan T., Gevorgyan A., Samvelyan M.A., Galstyan A.S., Parpart S., Rahman Q., Iqbal J., Langer P.
ChemistrySelect,
2017
18.

Ghochikyan T.V., Samvelyan M.A., Galstyan A.S., Gevorgyan A., Vardanyan G., Grigoryan T., Langer P.
Russian Journal of Organic Chemistry,
2017
19.

Kantam M.L., Srinivas P., Yadav J., Likhar P.R., Bhargava S.
Journal of Organic Chemistry,
2009
20.
![1,1′-Methylene-3,3′-bis[(N-(tert-butyl)imidazol-2-ylidene] and Its Effect in Palladium-Catalyzed C–C Coupling](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Joshaghani M., Nadri S., Rafiee E., Jamali S.
Synlett,
2015
21.

Yu L., Huang Y., Wei Z., Ding Y., Su C., Xu Q.
Journal of Organic Chemistry,
2015
22.

Lee D., Taher A., Hossain S., Jin M.
Organic Letters,
2011
23.

Xu H., Zhao Y., Zhou X.
Journal of Organic Chemistry,
2011
24.

Wang G., Shang R., Cheng W., Fu Y.
Journal of the American Chemical Society,
2017