Keywords
chromeno[2,3-b]pyridines
chromones
heterocyclization
nucleophilic reactions
pyrano[3,2-c]chromenes
pyrano[3′,4′:5,6]pyrano[2,3-b]chromenes
triacetic acid lactone
Abstract
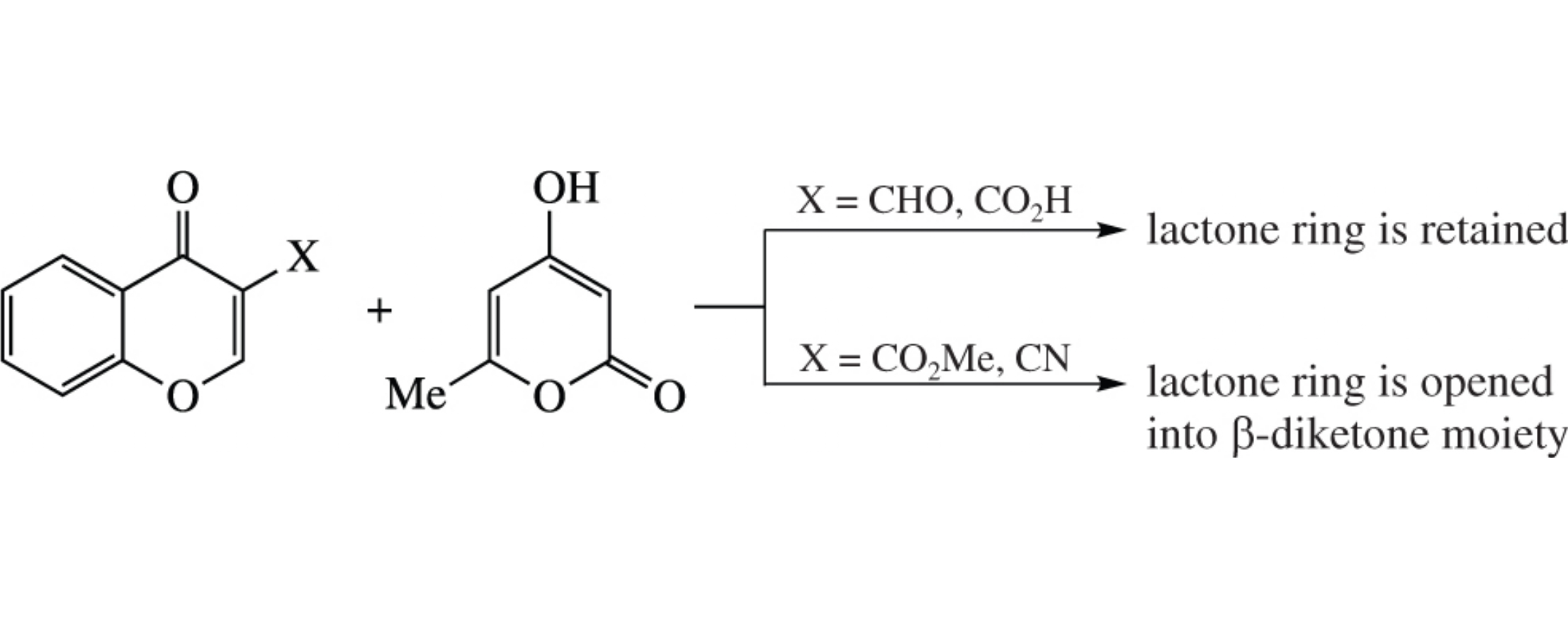
Reaction of 3-formylchromones and triacetic acid lactone proceeds with retention of the chromone skeleton and cyclization into pyrano[3′,4′:5,6]pyrano[2,3-b]chromene system. The crystal structure of the reaction product from unsubstituted 3-formylchromone has been determined from X-ray data. Other chromone analogues bearing CO2H, CO2Me and CN substituents react with triacetic acid lactone affording an o-hydroxychalcone heteroanalogue, pyrano[3,2-c]chromene and chromeno[2,3-b]pyridine derivatives, respectively.
References
1.

Keri R.S., Budagumpi S., Pai R.K., Balakrishna R.G.
European Journal of Medicinal Chemistry,
2014
2.

Gaspar A., Matos M.J., Garrido J., Uriarte E., Borges F.
Chemical Reviews,
2014
3.

Horton D.A., Bourne G.T., Smythe M.L.
Chemical Reviews,
2003
4.

Kornev M.Y., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2016
5.

A. Ibrahim M., M. El-Gohary N., Said S.
Heterocycles,
2015
6.
Ghosh C.K., Chakraborty A.
Arkivoc,
2015
7.
Kornev M.Y., Moshkin V.S., Eltsov O.S., Sosnovskikh V.Y.
Mendeleev Communications,
2016
8.
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
Mendeleev Communications,
2019
9.

Yu J., Landberg J., Shavarebi F., Bilanchone V., Okerlund A., Wanninayake U., Zhao L., Kraus G., Sandmeyer S.
Biotechnology and Bioengineering,
2018
10.

Song L., Yao H., Zhu L., Tong R.
Organic Letters,
2012
11.

Bio-based triacetic acid lactone in the synthesis of azaheterocyclesviaa ring-opening transformation
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
New Journal of Chemistry,
2018
12.
Obydennov D.L., El-Tantawy A.I., Kornev M.Y., Sosnovskikh V.Y.
Mendeleev Communications,
2019
13.
10.1016/j.mencom.2020.03.035_bib0035
Uddin
Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem.,
1995
14.

Siddiqui Z.N., Musthafa T.N., Praveen S.
Medicinal Chemistry Research,
2012
15.
10.1016/j.mencom.2020.03.035_sbref0040b
Asad
Acta Pol. Pharm. – Drug Res.,
2013
16.

Kumbhar D., Patil R., Patil D., Patravale A., Chandam D., Jadhav S., Deshmukh M.
Synthetic Communications,
2015
17.

Bari A., Ali S.S., Kadi A., Hashmi I.A., Ng S.W.
Chemistry of Heterocyclic Compounds,
2014
18.
![A One-Pot Condensation of Pyrones and Enals. Synthesis of 1H,7H-5a,6,8,9-Tetrahydro-1-oxopyrano[4,3-b][1]benzopyrans](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hua D.H., Chen Y., Sin H., Maroto M.J., Robinson P.D., Newell S.W., Perchellet E.M., Ladesich J.B., Freeman J.A., Perchellet J., Chiang P.K.
Journal of Organic Chemistry,
1997
19.
![Sequential 1,2-Addition−Electrocyclic Ring Closures Involving Acyclic α,β-Unsaturated Iminiums: A Formal [3 + 3] Cycloaddition Strategy to Unique Pyranyl Spirocycles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hsung R.P., Shen H.C., Douglas C.J., Morgan C.D., Degen S.J., Yao L.J.
Journal of Organic Chemistry,
1999
20.
![Metal-Free Brønsted Acid Catalyzed Formal [3 + 3] Annulation. Straightforward Synthesis of Dihydro-2H-Chromenones, Pyranones, and Tetrahydroquinolinones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Moreau J., Hubert C., Batany J., Toupet L., Roisnel T., Hurvois J., Renaud J.
Journal of Organic Chemistry,
2009
21.

Kornev M.Y., Moshkin V.S., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2015
22.
10.1016/j.mencom.2020.03.035_bib0065
Siddiqui
Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem.,
2006
23.
10.1016/j.mencom.2020.03.035_sbref0070a
Siddiqui
Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem.,
2006
24.

Siddiqui Z., Praveen S., Farooq F.
Chemical Papers,
2010