Keywords
fleximers
heterocyclic aromatic compounds
iodination
Pyrrole
silyl protection
Suzuki–Miyaura cross-coupling
Abstract
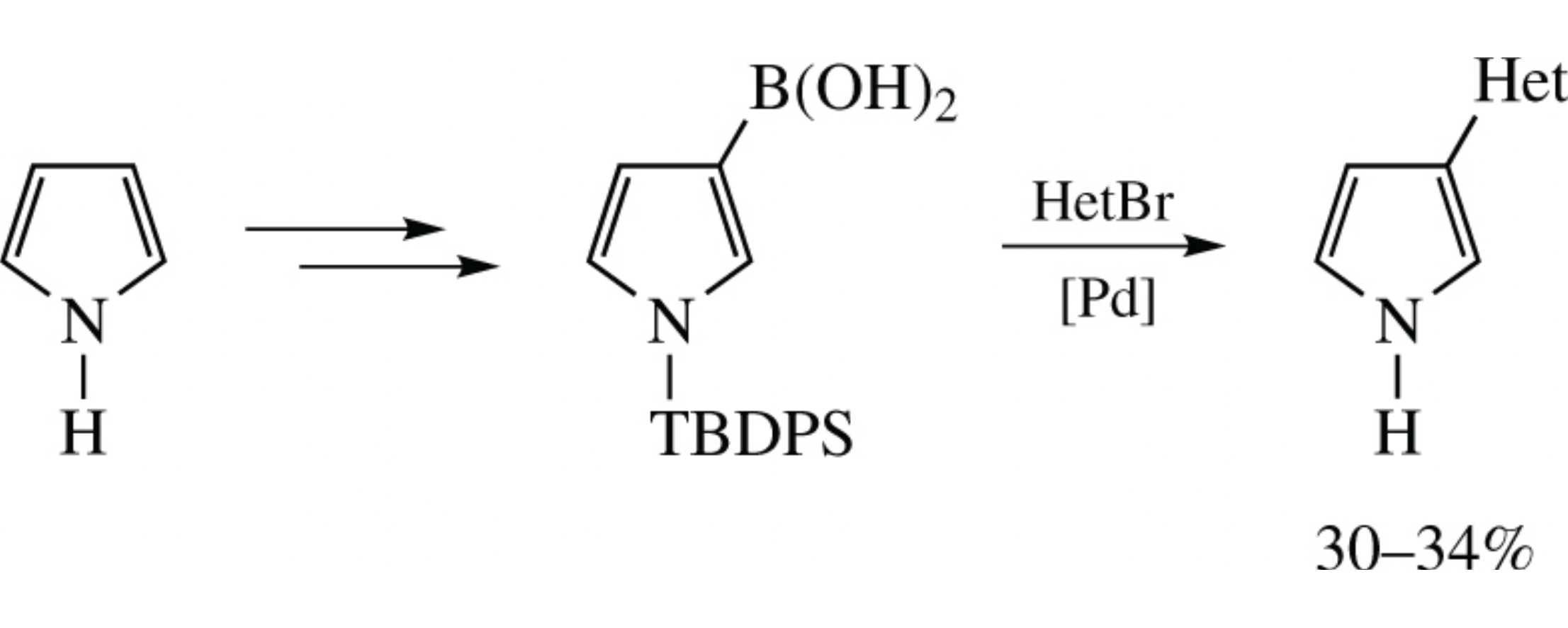
1-[tert-Butyl(diphenyl)silyl]pyrrol-3-ylboronic acid was obtained from pyrrole in three steps. Its Suzuki–Miyaura cross-coupling with functionalized pyridinyl and pyrimidinyl bromides afforded new promising 3-hetaryl-1H-pyrroles.
References
1.
10.1016/j.mencom.2020.03.034_bib0005
Asif
Int. J. Bioorg. Chem.,
2017
2.
10.1016/j.mencom.2020.03.034_bib0010
Selvam
Res. Pharm.,
2012
3.

Chiacchio M.A., Iannazzo D., Romeo R., Giofrè S.V., Legnani L.
Current Medicinal Chemistry,
2018
4.

Sharma S., Mehndiratta S., Kumar S., Singh J., Bedi P., Nepali K.
Recent Patents on Anti-Cancer Drug Discovery,
2015
5.

Abu El Asrar R., Margamuljana L., Abramov M., Bande O., Agnello S., Jang M., Herdewijn P.
ChemBioChem,
2017
6.

Babkov D.A., Geisman A.N., Khandazhinskaya A.L., Novikov M.S.
Russian Chemical Reviews,
2016
7.

Fateev I.V., Kharitonova M.I., Antonov K.V., Konstantinova I.D., Stepanenko V.N., Esipov R.S., Seela F., Temburnikar K.W., Seley-Radtke K.L., Stepchenko V.A., Sokolov Y.A., Miroshnikov A.I., Mikhailopulo I.A.
Chemistry - A European Journal,
2015
8.

Kharitonova M.I., Denisova A.O., Andronova V.L., Kayushin A.L., Konstantinova I.D., Kotovskaya S.K., Galegov G.A., Charushin V.N., Miroshnikov A.I.
Bioorganic and Medicinal Chemistry Letters,
2017
9.

Ku T., Lopresti N., Shirley M., Mori M., Marchant J., Heng X., Botta M., Summers M.F., Seley-Radtke K.L.
Bioorganic and Medicinal Chemistry,
2019
10.

Vichier-Guerre S., Dugué L., Bonhomme F., Pochet S.
Organic and Biomolecular Chemistry,
2016
11.

Vichier-Guerre S., Dugué L., Bonhomme F., Pochet S.
Organic and Biomolecular Chemistry,
2017
12.

Polak M., Seley K.L., Plavec J.
Journal of the American Chemical Society,
2004
13.

Seley K.L., Zhang L., Hagos A., Quirk S.
Journal of Organic Chemistry,
2002
14.

Seley K.L., Salim S., Zhang L., O'Daniel P.I.
Journal of Organic Chemistry,
2005
15.

Peters H., Ku T., Seley-Radtke K.
Current Medicinal Chemistry,
2015
16.

Yates M.K., Raje M.R., Chatterjee P., Spiropoulou C.F., Bavari S., Flint M., Soloveva V., Seley-Radtke K.L.
Bioorganic and Medicinal Chemistry Letters,
2017
17.

Nicolaou K.C., Bulger P.G., Sarlah D.
Angewandte Chemie - International Edition,
2005
18.

Légaré M., Courtemanche M., Rochette É., Fontaine F.
Science,
2015
19.

Billingsley K.L., Anderson K.W., Buchwald S.L.
Angewandte Chemie - International Edition,
2006
20.

Nadeau J.M., Swager T.M.
Tetrahedron,
2004
21.

Cui K., Gao M., Zhao H., Zhang D., Yan H., Huang H.
Molecules,
2019
22.

Vichier-Guerre S., Dugué L., Pochet S.
Tetrahedron Letters,
2014
23.

Schoenfeld R.C., Bourdet D.L., Brameld K.A., Chin E., de Vicente J., Fung A., Harris S.F., Lee E.K., Le Pogam S., Leveque V., Li J., Lui A.S., Najera I., Rajyaguru S., Sangi M., et. al.
Journal of Medicinal Chemistry,
2013
24.

Takase M., Inabe A., Sugawara Y., Fujita W., Nishinaga T., Nomura K.
Organic Letters,
2013
25.

Lee C., Chung Y.J.
Tetrahedron Letters,
2000
26.

Bray B.L., Mathies P.H., Naef R., Solas D.R., Tidwell T.T., Artis D.R., Muchowski J.M.
Journal of Organic Chemistry,
1990
27.

Jolicoeur B., Chapman E.E., Thompson A., Lubell W.D.
Tetrahedron,
2006
28.

Zhao S., Zhang Y., Zhou H., Xi S., Zou B., Bao G., Wang L., Wang J., Zeng T., Gong P., Zhai X.
European Journal of Medicinal Chemistry,
2016
29.

Zhang Y., Sun X., Fan N., Zhao J., Tu J., Chen X., Liu J., Wang X.
MedChemComm,
2015
30.

Crouch R.D., Blobaum A.L., Felts A.S., Conn P.J., Lindsley C.W.
Drug Metabolism and Disposition,
2017
31.
![Synthesis of 3,4‐diaminopyridine and imidazo[4,5‐c]pyridines by nitration of 4‐acylaminopyridines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bakke J.M., Riha J.
Journal of Heterocyclic Chemistry,
1999