Keywords
oxazoles
oxazolo[3,2-a]pyridinium
ring opening
tautomerism
X-ray diffraction
Abstract
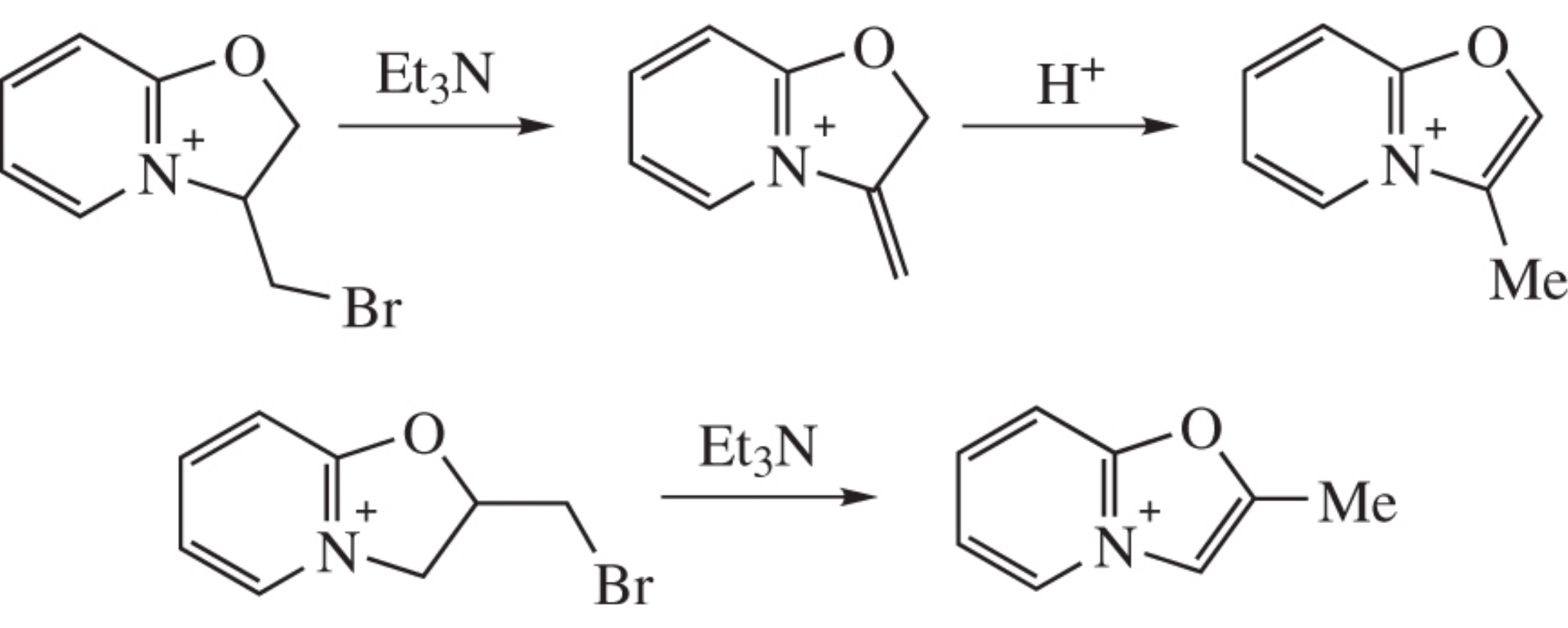
3-(Bromomethyl)dihydrooxazolo[3,2-a]pyridinium bromide under the action of base gives stable non-aromatic methylidene structure which can be converted into aromatic oxazolopyridinium cation only under the action of a superacid. On the contrary, 2-bromomethyl isomer under the same conditions affords only aromatic cation. The structure of the products was confirmed by X-ray diffraction.
References
1.

Ram R.N., Charles I.
Chemical Communications,
1999
2.

Nagashima E., Suzuki K., Sekiya M.
Tetrahedron Letters,
1981
3.

NAGASHIMA E., SUZUKI K., SEKIYA M.
Chemical and Pharmaceutical Bulletin,
2011
4.

Tidwell J.H., Senn D.R., Buchwald S.L.
Journal of the American Chemical Society,
1991
5.

Tidwell J.H., Buchwald S.L.
Journal of the American Chemical Society,
1994
6.

Younes S.
European Journal of Medicinal Chemistry,
2000
7.

Yang X., Liu W., Li L., Wei W., Li C.
Chemistry - A European Journal,
2016
8.
![Tricyclic Quinoxalinediones: 5,6-Dihydro-1H-pyrrolo[1,2,3-de]quinoxaline-2,3-diones and 6,7-Dihydro-1H,5H-pyrido[1,2,3-de]quinoxaline-2,3-diones as Potent Antagonists for the Glycine Binding Site of the NMDA Receptor](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Nagata R., Tanno N., Kodo T., Ae N., Yamaguchi H., Nishimura T., Antoku F., Tatsuno T., Kato T.
Journal of Medicinal Chemistry,
1994
9.

Chen W., Yang X., Li Y., Yang L., Wang X., Zhang G., Zhang H.
Organic and Biomolecular Chemistry,
2011
10.

Tidwell J.H., Peat A.J., Buchwald S.L.
Journal of Organic Chemistry,
1994
11.

Dichiarante V., Fagnoni M., Mella M., Albini A.
Chemistry - A European Journal,
2006
12.

Fotadar U., Becu C., Borremans F.A., Anteunis M.J.
Tetrahedron,
1978
13.
![Halocyclization of 2-(2-{4-[allylamino(thioxo)methyl]piperazin-1-yl}ethyl)-1H-benzo[de]isoquinoline-1,3(2H)-dione](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Zborovskii Y.L., Orysyk V.V., Staninets V.I., Bon’ V.V.
Russian Journal of Organic Chemistry,
2011
14.
![An alternate route to the synthesis of imidazo[1,2-a]quinolines using I2-NaI reagent](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Upadhyay S., Chandra A., Singh S., Singh R.M.
Journal of Heterocyclic Chemistry,
2011
15.
![Synthesis of new imidazo[1,2-a]pyrazolo[4,3-e]pyrimidin-4(6H)-one derivatives by iodocyclization of 6-alkenyl(alkynyl)-aminopyrazolo[3,4-d]pyrimidin-4(5H)-ones](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Bentya A.V., Vas’kevich R.I., Turov A.V., Rusanov E.B., Vovk M.V., Staninets V.I.
Russian Journal of Organic Chemistry,
2011
16.
![Iodocyclization of 6-allylamino-4,5-dihydropyrazolo[3,4-d]pyrimidines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Vas’kevich R.I., Bentya A.V., Turov A.V., Rusanov E.B., Staninets V.I., Vovk M.V.
Russian Journal of Organic Chemistry,
2012
17.
![Simple Synthesis of Imidazo[1,2-a]pyrazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Slepukhin P.A., Kim D.G., Rusinov G.L., Charushin V.N., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2002
18.
10.1016/j.mencom.2020.03.033_bib0090
Kim
Izv. Chelyabinskogo Nauchnogo Tsentra,
2004
19.
![4-Hydroxy-2-quinolones. 118. Synthesis, structure, and chemical properties of 2-bromomethyl-5-oxo-1,2-dihydro-5H-oxazolo-[3,2-a]quinoline-4-carboxylic acid and its ethyl ester](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ukrainets I.V., Sidorenko L.V., Gorokhova O.V., Shishkina S.V., Turov A.V.
Chemistry of Heterocyclic Compounds,
2007
20.
![Synthesis and Properties of Thiazolo- and Oxazolo-[3,2-a]Quinolinium Systems and Their Hydrogenated Derivatives (Review)*](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kim D.G., Vershinina E.A.
Chemistry of Heterocyclic Compounds,
2014
21.
![4-Hydroxy-2-quinolones 123. Amidation of 2-bromomethyl-5-oxo-1,2-dihydro-5H-oxazolo[3,2-a]-quinoline-4-carboxylic acid](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ukrainets I.V., Bereznyakova N.L., Turov A.V., Shishkina S.V.
Chemistry of Heterocyclic Compounds,
2007
22.
10.1016/j.mencom.2020.03.033_bib0110
Babaev
Incorporation of Heterocycles into Combinatorial Chemistry,
2017
23.
![Oxazolo[3,2-a]pyridinium and oxazolo[3,2-a]pyrimidinium salts in organic synthesis](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Babaev E.V., Alifanov V.L., Efimov A.V.
Russian Chemical Bulletin,
2008
24.

Okul’ E.M., Rybakov V.B., Babaev E.V.
Chemistry of Heterocyclic Compounds,
2017
25.

Kalita E.V., Kim D.G., Eltsov O.S., Shtukina T.S., Mukhametgaleeva I.V.
Chemistry of Heterocyclic Compounds,
2019
26.
![Dakin-West Trick in the Design of Novel 2-Alkyl(aralkyl) Derivatives of Oxazolo[3,2-a]pyridines](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Kazhkenov Z., Bush A., Babaev E.
Molecules,
2005
27.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
28.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
29.

Walker N., Stuart D.
Acta Crystallographica Section A Foundations of Crystallography,
1983