Keywords
arylation
hydrazones
indole-3-carbaldehyde
quinazoline
trifluoroacetic acid
trifluoroacetylhydrazides
Abstract
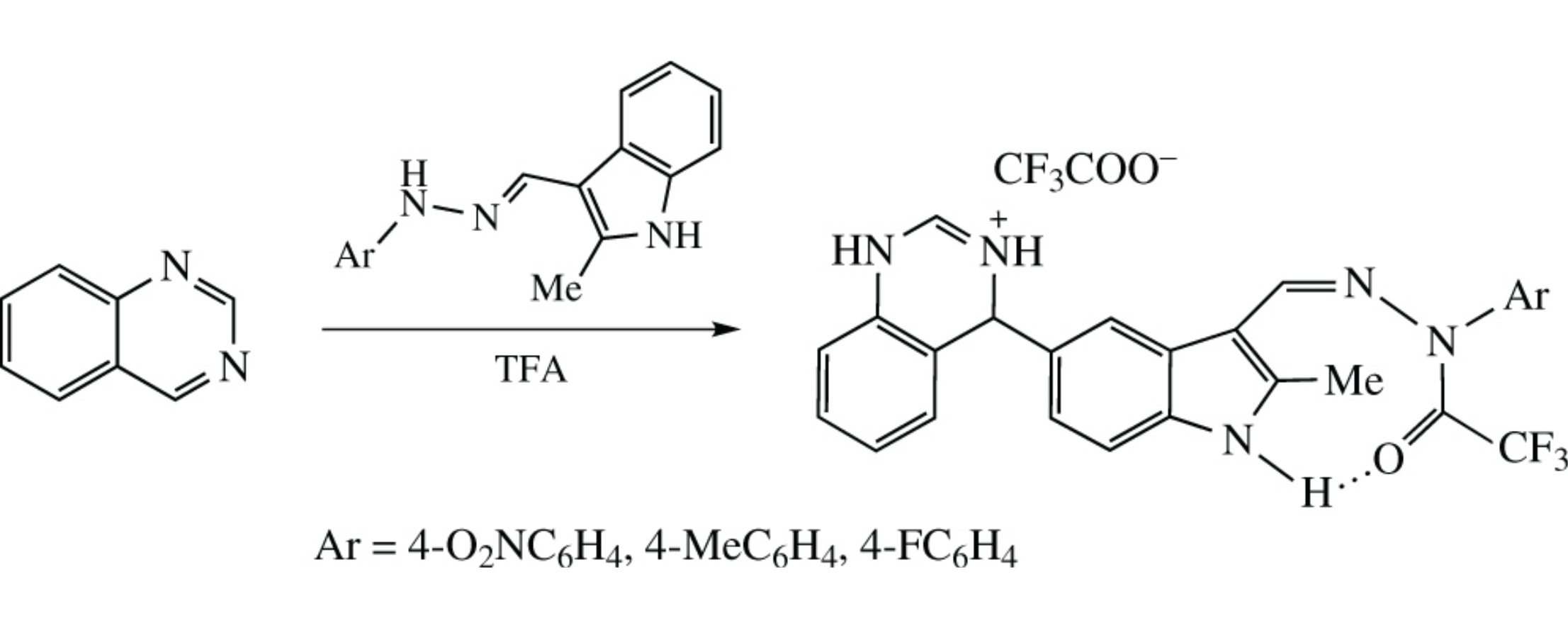
Addition of indole 3-carbaldehyde arylhydrazones to quinazoline in trifluoroacetic acid occurs at the C5-atom of the indole moiety affording quinazoline σ-adducts with indole and trifluoroacetylhydrazide moieties.
References
1.

D'yakonov A.L., Telezhenetskaya M.V.
Chemistry of Natural Compounds,
1997
2.
10.1016/j.mencom.2020.03.032_sbref0005b
Aniszewski
Alkaloids: Chemistry, Biology, Ecology, and Applications,
2015
3.
10.1016/j.mencom.2020.03.032_bib0010
Asif
Int. J. Med. Chem.,
2014
4.
Solyanik G.I.
Experimental Oncology,
2019
5.
10.1016/j.mencom.2020.03.032_bib0020
Tregubenko
Radiobiologiya,
1984
6.

Pilicheva T.L., Chupakhin O.N., Postovskii I.Y.
Chemistry of Heterocyclic Compounds,
1975
7.

Azev Y.A., Shorshnev S.V., Golomolzin B.V.
Tetrahedron Letters,
2009
8.
10.1016/j.mencom.2020.03.032_bib0035
Anastas
Green Chemistry: Theory and Practice,
1998
10.

Mąkosza M., Wojciechowski K.
Topics in Heterocyclic Chemistry,
2013
11.

Zabaleta N., Uria U., Reyes E., Carrillo L., Vicario J.L.
Chemical Communications,
2018
12.

Tunc˛ T., Tezcan H., Sarı M., Büyükgüngör O., Yaǵbasan R.
Acta Crystallographica Section C Crystal Structure Communications,
2003