Keywords
2,4-dihydro-3H-pyrazol-3-one
aldehydes
electrocatalysis
kojic acid
multicomponent process
tandem reaction
Abstract
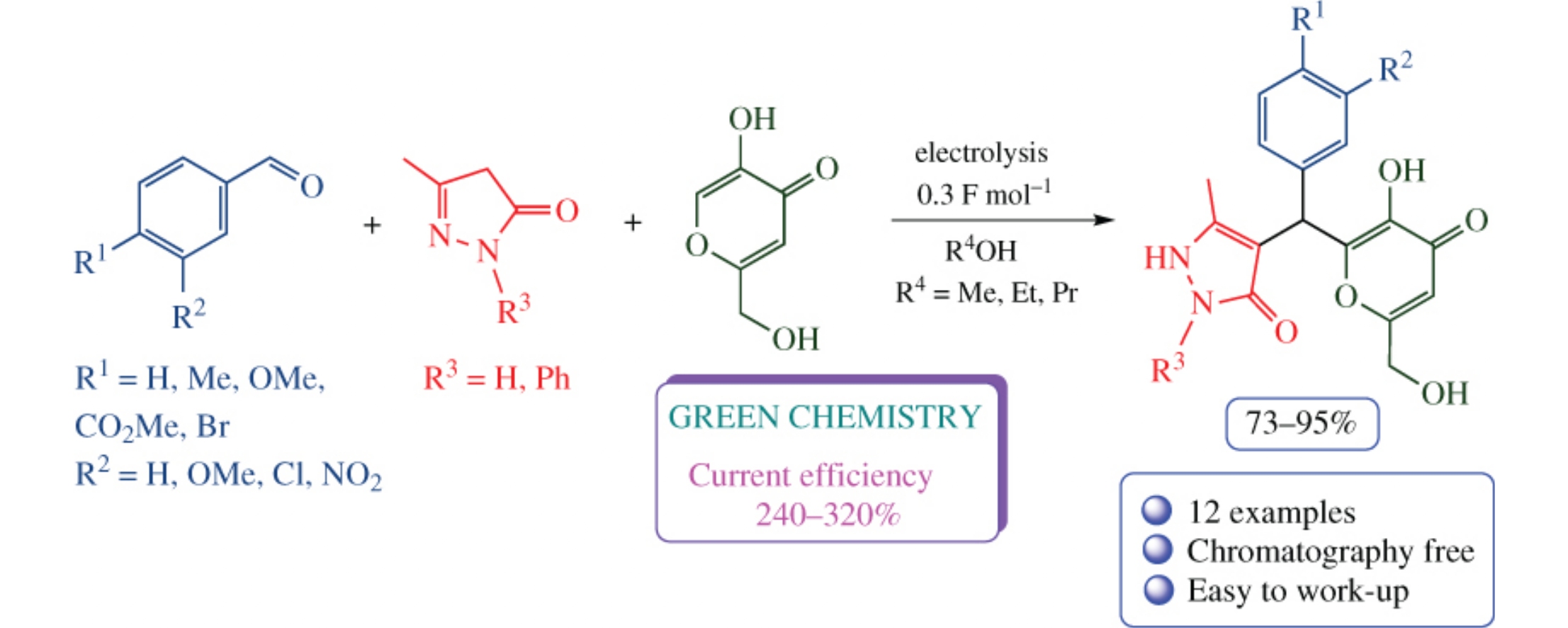
Electrocatalytic transformation of aldehydes, 2,4-dihydro-3H-pyrazol-3-ones and kojic acid in alcohols occurs in an undivided cell in the presence of sodium halides, selectively affording substituted 4-{[3-hydroxy-6-(hydroxymethyl)-4-oxo-4H-pyran-2-yl](aryl)methyl}-5-methyl-1,2-dihydro-3Hpyrazol-3-ones in 73–95% yields and with 240–320% current efficiency. The multicomponent process provides a facile and efficient way to the new type of systems with heterocyclic moieties separated by aryl-substituted C-spacer. The product structure has been confirmed by X-ray diffraction data
References
1.
10.1016/j.mencom.2020.03.031_sbref0005a
Organic Electrochemistry: Revised and Expanded,
2015
2.

Yan M., Kawamata Y., Baran P.S.
Angewandte Chemie - International Edition,
2017
3.
Nikishin G.I., Elinson M.N., Makhova I.V.
1988
4.

Elinson M.N., Feducovich S.K., Lizunova T.L., Nikishin G.I.
Tetrahedron,
2000
5.

Elinson M., Dorofeev A., Miloserdov F., Ilovaisky A., Feducovich S., Belyakov P., Nikishin G.
Advanced Synthesis and Catalysis,
2008
6.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Krymov S.K., Fakhrutdinov A.N., Egorov M.P.
Mendeleev Communications,
2019
7.

Catalysis of Cascade and Multicomponent Reactions of Carbonyl Compounds and CH Acids by Electricity
Elinson M.N., Vereshchagin A.N., Ryzhkov F.V.
Chemical Record,
2016
8.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Goloveshkin A.S., Ushakov I.E., Egorov M.P.
Mendeleev Communications,
2018
9.
![Multicomponent transformation of salicylaldehydes, 2-aminoprop-1-ene-1,1,3-tricarbonitrile, and pyrazolin-5-ones into substituted 2,4-diamino-5-(5-hydroxy-3-methyl-1H-pyrazol-4-yl)-5H-chromeno[2,3-b]pyridine-3-carbonitriles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Goloveshkin A.S., Ushakov I.E., Egorov M.P.
Russian Chemical Bulletin,
2018
10.
10.1016/j.mencom.2020.03.031_bib0025
Arora
Int. J. Pharm. Sci. Res.,
2012
11.

Taylor A.P., Robinson R.P., Fobian Y.M., Blakemore D.C., Jones L.H., Fadeyi O.
Organic and Biomolecular Chemistry,
2016
12.
10.1016/j.mencom.2020.03.031_bib0035
Wiley
Pyrazolones, Pyrazolidones, and Derivatives,
1964
13.
10.1016/j.mencom.2020.03.031_bib0040
Elguero
1996
14.

15.

Wise L.D., Butler D.E., DeWald H.A., Lustgarten D.M., Pattison I.C., Schweiss D.N., Coughenour L.L., Downs D.A., Heffner T.G., Pugsley T.A.
Journal of Medicinal Chemistry,
1987
16.

Bentley R.
Natural Product Reports,
2006
17.

Parvez S., Kang M., Chung H., Cho C., Hong M., Shin M., Bae H.
Phytotherapy Research,
2006
18.
10.1016/j.mencom.2020.03.031_bib0065
Shafiquzzaman
New and Future Developments in Microbial Biotechnology and Bioengineering. Pennicillium System Properties and Applications,
2018
19.
10.1016/j.mencom.2020.03.031_bib0070
Aytemir
Arzneim. Forsch.,
2010
20.

Rho H.S., Ahn S.M., Yoo D.S., Kim M.K., Cho D.H., Cho J.Y.
Bioorganic and Medicinal Chemistry Letters,
2010
21.

Tanaka R., Tsujii H., Yamada T., Kajimoto T., Amano F., Hasegawa J., Hamashima Y., Node M., Katoh K., Takebe Y.
Bioorganic and Medicinal Chemistry,
2009
22.

Burdock G.A., Soni M.G., Carabin I.G.
Regulatory Toxicology and Pharmacology,
2001
23.

Karakaya G., Türe A., Ercan A., Öncül S., Aytemir M.D.
Bioorganic Chemistry,
2019
24.
S. Ansorge, U. Bank, K. Nordhoff, M. Taeger, F. Striggow, US Patent 2007/0037752 A1, 2007.
25.

Elinson M.N., Dorofeeva E.O., Vereshchagin A.N., Nasybullin R.F., Egorov M.P.
Catalysis Science and Technology,
2015
26.

Elinson M.N., Merkulova V.M., Ilovaisky A.I., Barba F., Batanero B.
Electrochimica Acta,
2011
27.

Patai S., Israeli Y.
Journal of the Chemical Society (Resumed),
1960