Keywords
drug metabolites
MALDI MS
photocatalysis
photooxidation
titanium dioxide
Abstract
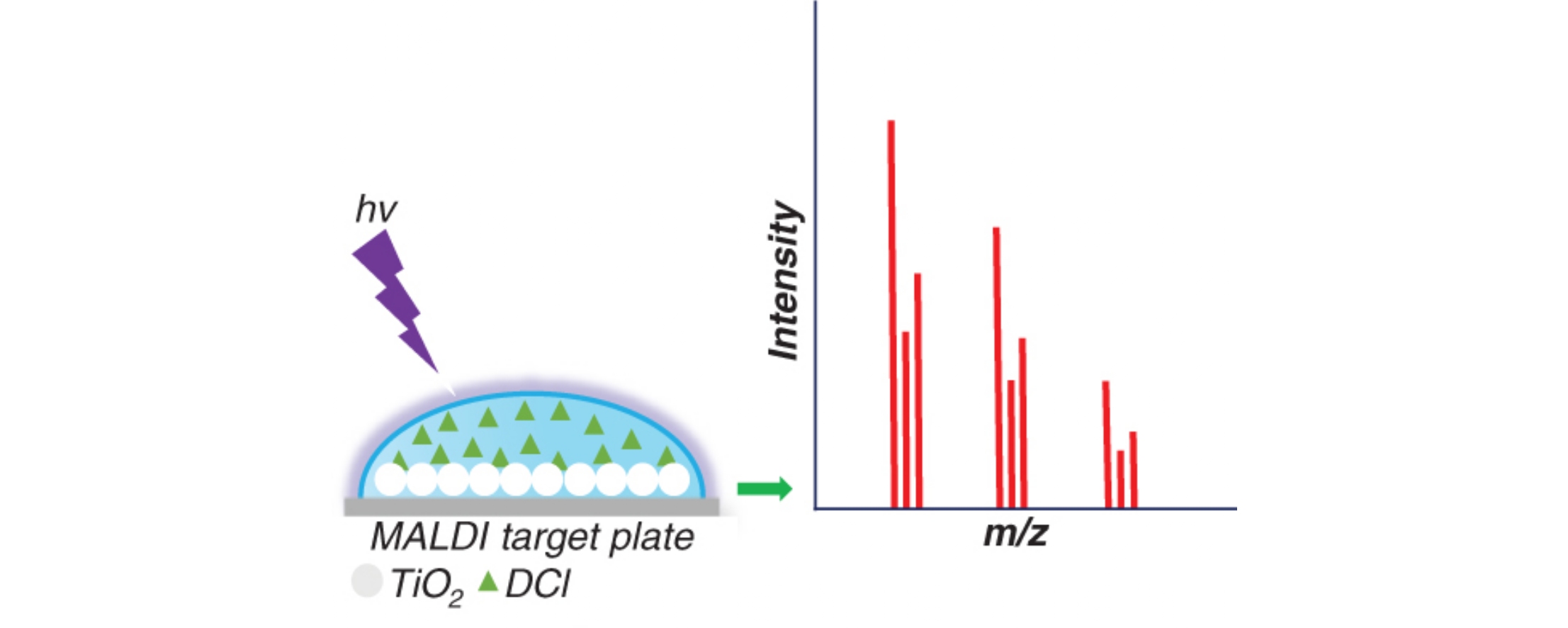
The UV-induced photocatalytic oxidation in the presence of TiO2 nanoparticles (UV/TiO2-PCO) is a more adequate approach than electrochemical oxidation to simulate the oxidative metabolism of diclofenac based on the comparative analysis of oxidation products using high-resolution tandem mass spectrometry. A simple and fast high-throughput technique is proposed for modeling the oxidative metabolism, which involves UV/TiO2-PCO performed directly on a MALDI target and subsequent analysis by matrix-assisted laser desorption/ionization mass spectrometry. The ranges and yields of diclofenac oxidation products obtained by the conventional bulk UV/TiO2-PCO and the proposed on-target version are in excellent agreement.
References
1.

Park B.K., Boobis A., Clarke S., Goldring C.E., Jones D., Kenna J.G., Lambert C., Laverty H.G., Naisbitt D.J., Nelson S., Nicoll-Griffith D.A., Obach R.S., Routledge P., Smith D.A., Tweedie D.J., et. al.
Nature Reviews Drug Discovery,
2011
2.

Lohmann W., Karst U.
Analytical and Bioanalytical Chemistry,
2007
3.
10.1016/j.mencom.2020.03.030_bib0015
Tong
Cent. Eur. J. Chem.,
2012
4.
Kovalev I.A., Petrov A.A., Ibragimova O.A., Shokod’ko A.V., Chernyavskii A.S., Goodilin E.A., Solntseva K.A., Tarasov A.B.
Mendeleev Communications,
2018
5.

Gainanova A.A., Kuz’micheva G.M., Vasil’eva I.G.
Russian Chemical Bulletin,
2018
6.

Gawlik M., Skibiński R., Trawiński J., Komsta Ł.
Acta Chromatographica,
2017
7.

Ruokolainen M., Gul T., Permentier H., Sikanen T., Kostiainen R., Kotiaho T.
European Journal of Pharmaceutical Sciences,
2016
8.

Gawlik M., Trawiński J., Skibiński R.
European Journal of Pharmaceutical Sciences,
2018
9.

Nissilä T., Sainiemi L., Karikko M., Kemell M., Ritala M., Franssila S., Kostiainen R., Ketola R.A.
Lab on a Chip,
2011
10.

Ruokolainen M., Miikkulainen V., Ritala M., Sikanen T., Kotiaho T., Kostiainen R.
Analytical Chemistry,
2017
11.

van Geenen F.A., Franssen M.C., Miikkulainen V., Ritala M., Zuilhof H., Kostiainen R., Nielen M.W.
Journal of the American Society for Mass Spectrometry,
2019
12.

Calza P., Sakkas V.A., Medana C., Baiocchi C., Dimou A., Pelizzetti E., Albanis T.
Applied Catalysis B: Environmental,
2006
13.

Tang W.
Current Drug Metabolism,
2003
14.

Stierlin H., Faigle J.W., Sallmann A., Kung W., Richter W.J., Kriemler H.-., Alt K.O., Winkler T.
Xenobiotica,
1979
15.
10.1002/(SICI)1096-9888(200003)35:3<417::AID-JMS952>3.0.CO;2-#
16.

Sonderegger H., Rameshan C., Lorenz H., Klauser F., Klerks M., Rainer M., Bakry R., Huck C.W., Bonn G.K.
Analytical and Bioanalytical Chemistry,
2011
17.

Calvano C.D., Monopoli A., Cataldi T.R., Palmisano F.
Analytical and Bioanalytical Chemistry,
2018
18.

Sunner J., Dratz E., Chen Y.
Analytical Chemistry,
1995