Keywords
cysteine-rich peptides
peptide toxins
recombinant production
refolding
thioredoxin
Abstract
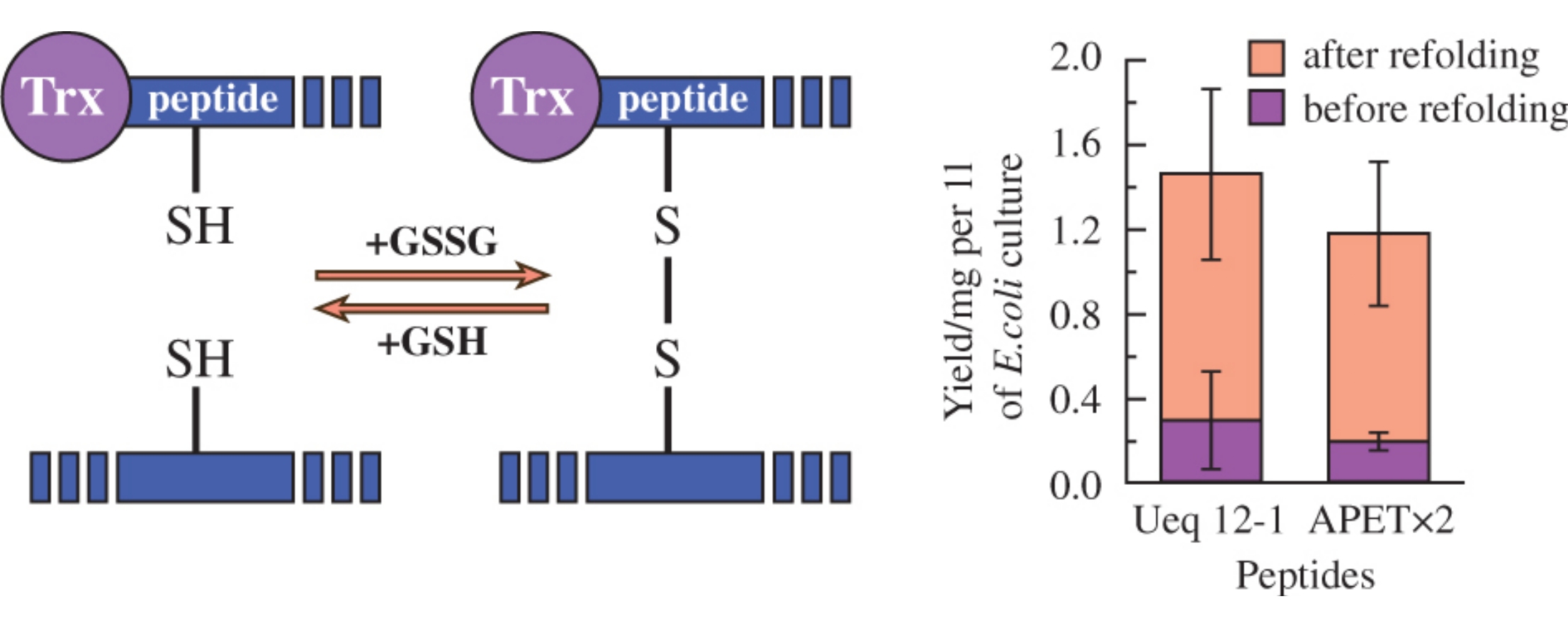
A protocol for refolding of thioredoxin-fused cysteine-rich peptides via addition of oxidized/reduced glutathione reagent directly to unfolded soluble fused protein has been developed. This procedure allows one to skip the steps of inclusion bodies purification, denaturation/disulfide reduction as well as lyophilization before oxidative folding, and thus to improve the yield of cysteine-rich peptides in their production using E. coli expression system.
References
1.

J. Prentis P., Pavasovic A., S. Norton R.
Toxins,
2018
2.

Tam J., Wang S., Wong K., Tan W.
Pharmaceuticals,
2015
3.

Slavokhotova A.A., Odintsova T.I., Rogozhin E.A., Musolyamov A.K., Andreev Y.A., Grishin E.V., Egorov T.A.
Biochimie,
2011
4.

Berkmen M.
Protein Expression and Purification,
2012
5.

Hatahet F., Boyd D., Beckwith J.
Biochimica et Biophysica Acta - Proteins and Proteomics,
2014
6.
10.1016/j.mencom.2020.03.028_bib0030
Rosano
Front. Microbiol.,
2014
7.

Stewart E.J.
EMBO Journal,
1998
8.

Yasukawa T., Kanei-Ishii C., Maekawa T., Fujimoto J., Yamamoto T., Ishii S.
Journal of Biological Chemistry,
1995
9.

Kudryashova K.S., Nekrasova O.V., Kuzmenkov A.I., Vassilevski A.A., Ignatova A.A., Korolkova Y.V., Grishin E.V., Kirpichnikov M.P., Feofanov A.V.
Analytical and Bioanalytical Chemistry,
2013
10.

Slavokhotova A.A., Naumann T.A., Price N.P., Rogozhin E.A., Andreev Y.A., Vassilevski A.A., Odintsova T.I.
FEBS Journal,
2014
11.

Logashina Y.A., Mosharova I.V., Korolkova Y.V., Shelukhina I.V., Dyachenko I.A., Palikov V.A., Palikova Y.A., Murashev A.N., Kozlov S.A., Stensvåg K., Andreev Y.A.
Journal of Biological Chemistry,
2017
12.

Osmakov D.I., Koshelev S.G., Andreev Y.A., Dyachenko I.A., Bondarenko D.A., Murashev A.N., Grishin E.V., Kozlov S.A.
Toxicon,
2016
13.

Piubelli L., Campa M., Temporini C., Binda E., Mangione F., Amicosante M., Terreni M., Marinelli F., Pollegioni L.
Microbial Cell Factories,
2013
14.

Berkut A.A., Peigneur S., Myshkin M.Y., Paramonov A.S., Lyukmanova E.N., Arseniev A.S., Grishin E.V., Tytgat J., Shenkarev Z.O., Vassilevski A.A.
Journal of Biological Chemistry,
2015
15.

Berkut A.A., Chugunov A.O., Mineev K.S., Peigneur S., Tabakmakher V.M., Krylov N.A., Oparin P.B., Lihonosova A.F., Novikova E.V., Arseniev A.S., Grishin E.V., Tytgat J., Efremov R.G., Vassilevski A.A.
Journal of Biological Chemistry,
2019
16.

Zhan J., Chen X., Wang C., Qiu J., Ma F., Wang K., Zheng S.
Biochemical and Biophysical Research Communications,
2003
17.

Vassilevski A.A., Sachkova M.Y., Ignatova A.A., Kozlov S.A., Feofanov A.V., Grishin E.V.
FEBS Journal,
2013
18.

Bae C., Kalia J., Song I., Yu J., Kim H.H., Swartz K.J., Kim J.I.
PLoS ONE,
2012
19.

Cassland P., Larsson S., Nilvebrant N., Jönsson L.J.
Journal of Biotechnology,
2004
20.

Lobstein J., Emrich C.A., Jeans C., Faulkner M., Riggs P., Berkmen M.
Microbial Cell Factories,
2012
21.

Jensen J.E., Durek T., Alewood P.F., Adams D.J., King G.F., Rash L.D.
Toxicon,
2009
22.

Anangi R., Chen C., Lin Y., Cheng Y., Cheng C., Chen Y., Chu Y., Chuang W.
Toxicon,
2010
23.

Anangi R., Rash L.D., Mobli M., King G.F.
Marine Drugs,
2012
24.

A new sea anemone peptide, APETx2, inhibits ASIC3, a major acid-sensitive channel in sensory neurons
Diochot S., Baron A., Rash L.D., Deval E., Escoubas P., Scarzello S., Salinas M., Lazdunski M.
EMBO Journal,
2004
25.

Chagot B., Escoubas P., Diochot S., Bernard C., Lazdunski M., Darbon H.
Protein Science,
2005
26.

Logashina Y.A., Solstad R.G., Mineev K.S., Korolkova Y.V., Mosharova I.V., Dyachenko I.A., Palikov V.A., Palikova Y.A., Murashev A.N., Arseniev A.S., Kozlov S.A., Stensvåg K., Haug T., Andreev Y.A.
Toxins,
2017
27.

Andreev Y.A., Kozlov S.A., Vassilevski A.A., Grishin E.V.
Analytical Biochemistry,
2010