Keywords
aggregation
albumin
fluorescence
IR spectroscopy
porphyrin
UV-VIS spectroscopy
Abstract
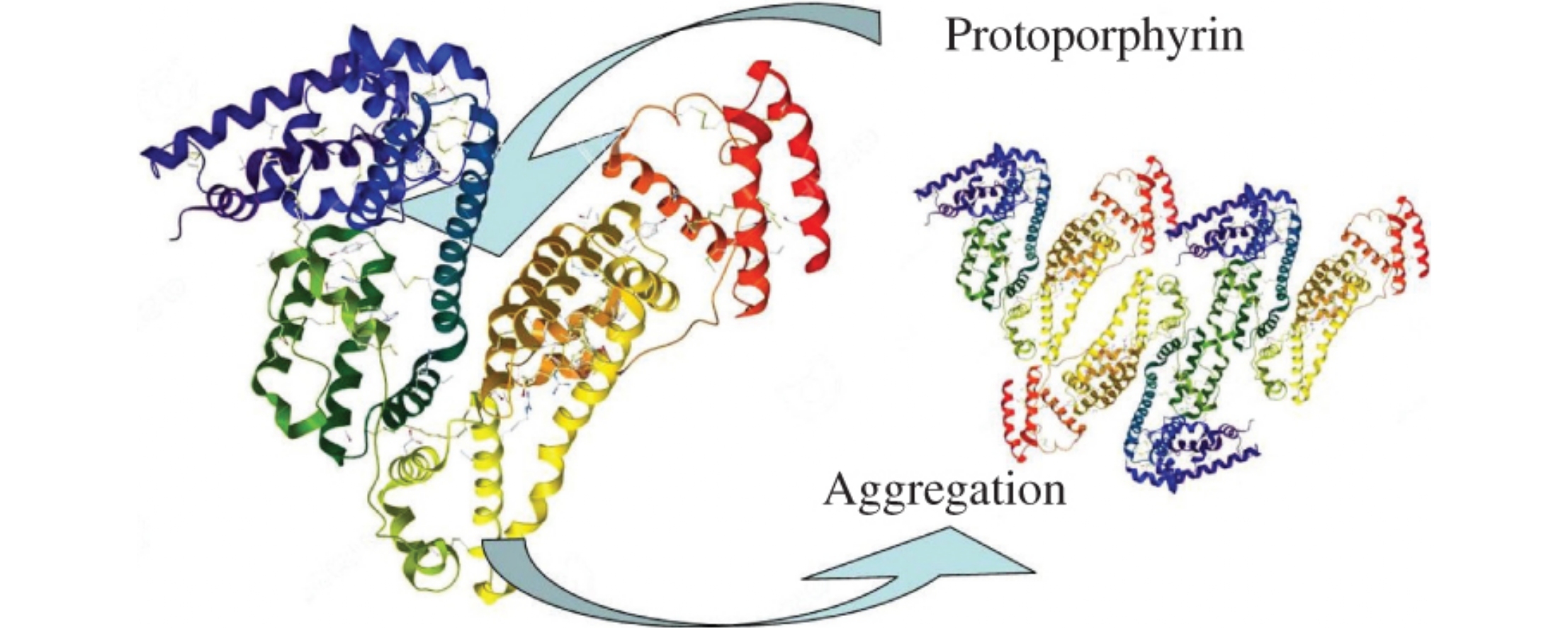
Protoporphyrin upon its binding with serum albumin changes its secondary structure due to the conversion of part of α helices into β-folding. This process results in the association of albumin globules in vitro.
References
1.

Makarska-Bialokoz M.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2018
2.
Chiabrando D., Mercurio S., Tolosano E.
Haematologica,
2014
3.

Chung J., Chen C., Paw B.H.
Current Opinion in Hematology,
2012
4.

Balwani M., Desnick R.J.
Hematology / the Education Program of the American Society of Hematology. American Society of Hematology. Education Program,
2012
5.
10.1016/j.mencom.2020.03.027_bib0025
Miller
Biochem. Mol. Biol. Int.,
1994
6.

Schaer D.J., Buehler P.W., Alayash A.I., Belcher J.D., Vercellotti G.M.
Blood,
2013
7.

Schmitt T.H., Frezzatti W.A., Schreier S.
Archives of Biochemistry and Biophysics,
1993
8.

Jeney V., Balla J., Yachie A., Varga Z., Vercellotti G.M., Eaton J.W., Balla G.
Blood,
2002
9.

KRISHNAMURTHY P., XIE T., SCHUETZ J.
Pharmacology and Therapeutics,
2007
10.

Lebedeva N.S., Yurina E.S., Gubarev Y.A., Syrbu S.A.
Journal of Molecular Liquids,
2019
11.
![The interaction of 5,10,15,20-tetrakis [4- (2,3,4,6-tetra-O-acetyl-β-D-galactopyranosyl) phenyl] porphine with biopolymers](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Lebedeva N.S., Yurina E.S., Guseinov S.S., Gubarev Y.A., Syrbu S.A.
Dyes and Pigments,
2019
12.
Lebedeva N.S., Yurina E.S., Gubarev Y.A., Syrbu S.A., Pechnikova N.L.
Mendeleev Communications,
2018
13.
10.1016/j.mencom.2020.03.027_bib0065
Siddiqi
Front. Biosci., Elite Ed.,
2017
14.

Lebedeva N.S., Gubarev Y.A., Vyugin A.I., Koifman O.I.
Journal of Luminescence,
2015
15.
![Greatly enhanced binding of a cationic porphyrin towards bovine serum albumin by cucurbit[8]uril](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Lei W., Jiang G., Zhou Q., Zhang B., Wang X.
Physical Chemistry Chemical Physics,
2010
16.

Jisha V.S., Arun K.T., Hariharan M., Ramaiah D.
Journal of the American Chemical Society,
2006
17.

Hu D., Sheng Z., Gao G., Siu F., Liu C., Wan Q., Gong P., Zheng H., Ma Y., Cai L.
Biomaterials,
2016
18.

Fanali G., di Masi A., Trezza V., Marino M., Fasano M., Ascenzi P.
Molecular Aspects of Medicine,
2012
19.

Zunszain P.A., Ghuman J., Komatsu T., Tsuchida E., Curry S.
BMC Structural Biology,
2003
20.

Wardell M., Wang Z., Ho J.X., Robert J., Ruker F., Ruble J., Carter D.C.
Biochemical and Biophysical Research Communications,
2002
21.

Pacheco M.E., Bruzzone L.
Journal of Luminescence,
2013
22.
![Study on the binding of 2,3-diazabicyclo[2.2.2]oct-2-ene with bovine serum albumin by fluorescence spectroscopy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Anbazhagan V., Renganathan R.
Journal of Luminescence,
2008
23.

Hu Y., Liu Y., Wang J., Xiao X., Qu S.
Journal of Pharmaceutical and Biomedical Analysis,
2004
24.

Jackson M., Mantsch H.H.
Critical Reviews in Biochemistry and Molecular Biology,
1995
25.
10.1016/j.mencom.2020.03.027_bib0125
Cooper
Physical Methods to Characterize Pharmaceutical Proteins (Pharmaceutical Biotechnology, vol. 7),
1995
26.

Prestrelski S.J., Arakawa T., Carpenter J.F.
Archives of Biochemistry and Biophysics,
1993
27.

Boucher L.J., Katz J.J.
Journal of the American Chemical Society,
1967
28.