Keywords
5,6,7,8-tetrahydronaphthols
cyclohexane
ionic hydrogenation
naphthyl ethers
organofluorine compounds
polyfluoropyridines
Abstract
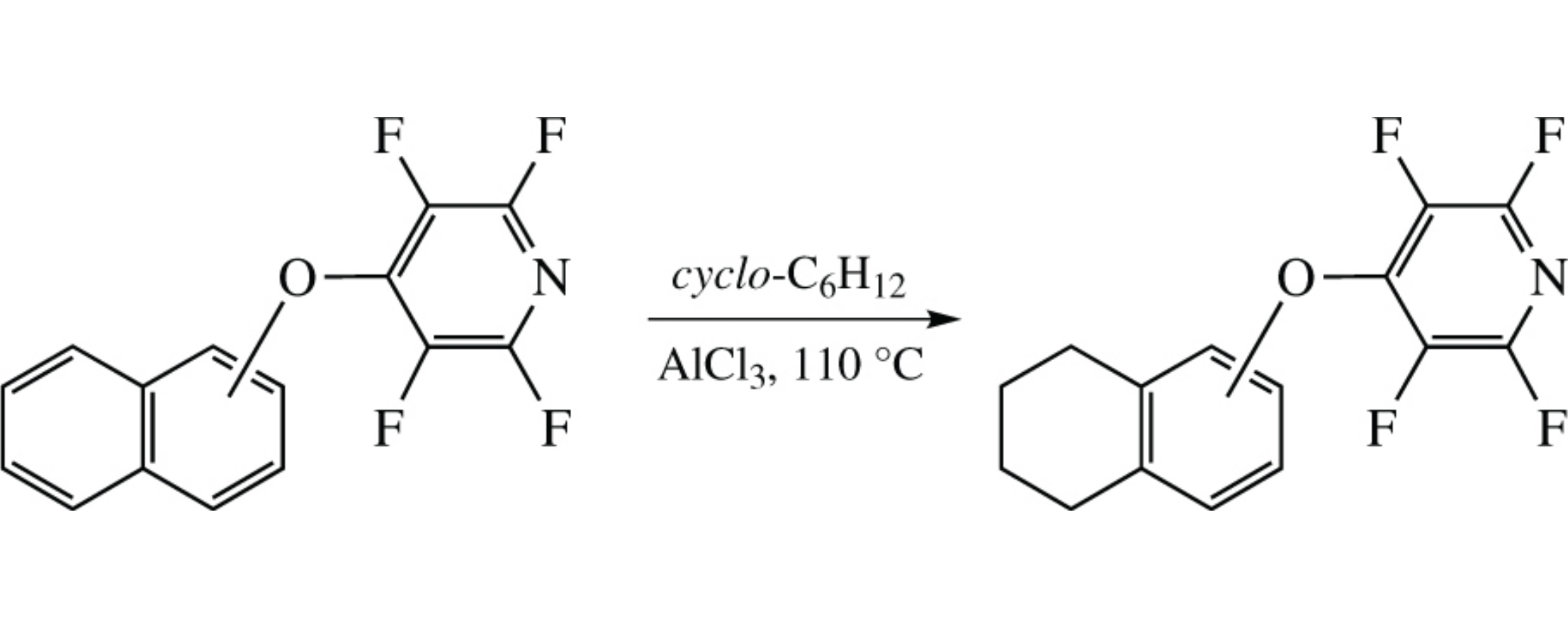
O-Tetrafluoropyridin-4-yl-protected naphthols undergo regioselective reduction with cyclohexane in the presence of aluminium chloride to afford the corresponding 5,6,7,8-tetrahydronaphthyl ethers.
References
1.
10.1016/j.mencom.2020.03.020_sbref0005a
Koltunov
Russ. J. Org. Chem.,
1997
2.
10.1016/j.mencom.2020.03.020_sbref0005b
Koltunov
Russ. J. Org. Chem.,
1998
3.
Zhu Z., Koltunov K.Y.
Mendeleev Communications,
2016
4.
5.
10.1016/j.mencom.2020.03.020_sbref0005e
Olah
Superelectrophiles and Their Chemistry,
2008
6.

Brittain W.D., Cobb S.L.
Organic and Biomolecular Chemistry,
2019
7.

Jarman M., McCague R.
Journal of the Chemical Society Chemical Communications,
1984
8.

Chambers R.D., Hoskin P.R., Sandford G., Yufit D.S., Howard J.A.
Journal of the Chemical Society Perkin Transactions 1,
2001
9.

Chambers R.D.
Journal of Fluorine Chemistry,
2010
10.

Litvak V.V., Mainagashev I.Y., Bukhanets O.G.
Russian Chemical Bulletin,
2007
11.

Koltunov K.Y., Prakash G.K., Rasul G., Olah G.A.
European Journal of Organic Chemistry,
2006
12.
10.1016/j.mencom.2020.03.020_sbref0020a
Olah
Superacid Chemistry,
2009
13.

Smith G.P., Dworkin A.S., Pagni R.M., Zingg S.P.
Journal of the American Chemical Society,
1989
14.

Smith G.P., Dworkin A.S., Pagni R.M., Zingg S.P.
Journal of the American Chemical Society,
1989
15.

Mechanism of Isobutylene Polymerization: Quantum Chemical Insight into AlCl3/H2O-Catalyzed Reactions
Vo M.N., Basdogan Y., Derksen B.S., Proust N., Cox G.A., Kowall C., Keith J.A., Johnson J.K.
ACS Catalysis,
2018
16.

Zhu Z., Ostashevskaya L.A., Koltunov K.Y.
Tetrahedron Letters,
2015
17.

Tsukinoki T., Kanda T., Liu G., Tsuzuki H., Tashiro M.
Tetrahedron Letters,
2000
18.

He Y., Tang J., Luo M., Zeng X.
Organic Letters,
2018