Keywords
2-imidazolines
aziridines
grinding
heterocyclization
nitriles
solvent-free reactions
Abstract
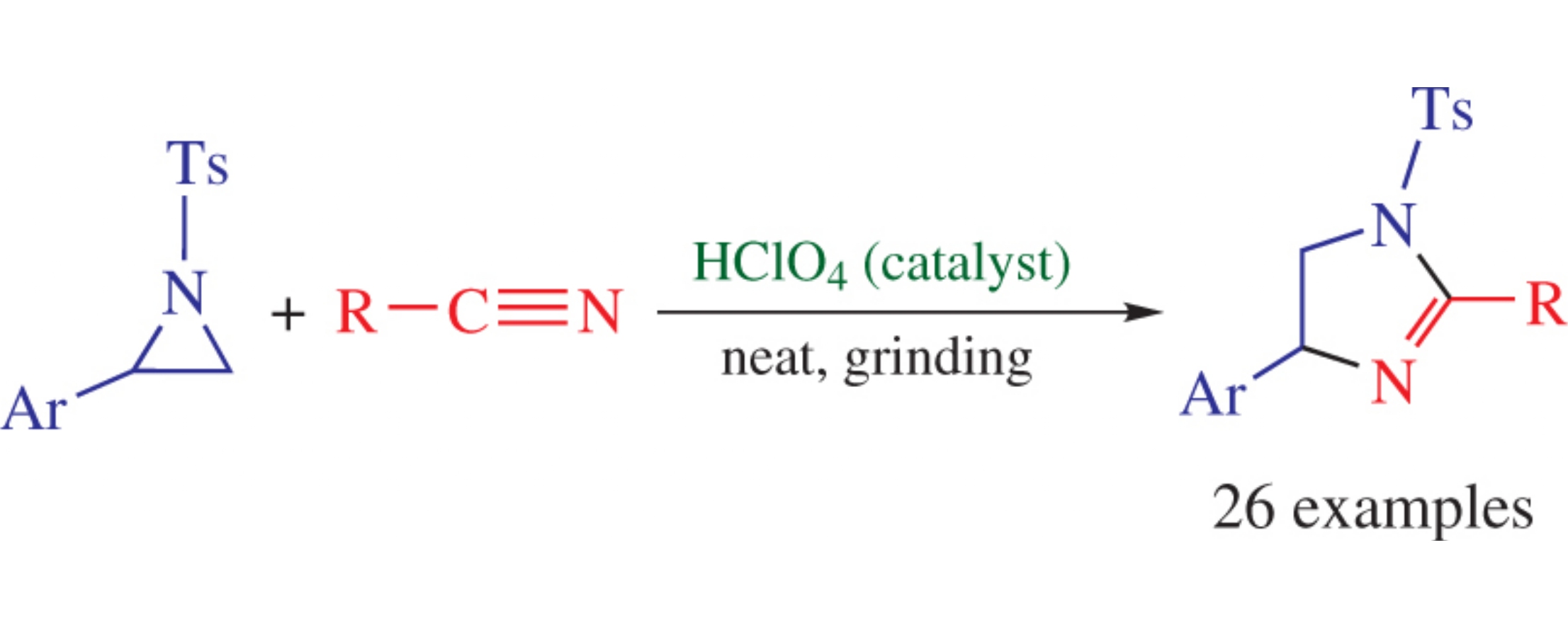
Solvent-free solid-state co-grinding of N-tosylaziridines and nitriles in the presence of perchloric acid as the catalyst affords 2-imidazolines in good yields.
References
1.

James S.L., Adams C.J., Bolm C., Braga D., Collier P., Friščić T., Grepioni F., Harris K.D., Hyett G., Jones W., Krebs A., Mack J., Maini L., Orpen A.G., Parkin I.P., et. al.
Chemical Society Reviews,
2012
2.

Friščić T.
Chemical Society Reviews,
2012
3.
10.1016/j.mencom.2020.03.019_bib0010
Grimmett
1996
4.

Peddibhotla S., Jayakumar S., Tepe J.J.
Organic Letters,
2002
5.

Rondu F., Le Bihan G., Wang X., Lamouri A., Touboul E., Dive G., Bellahsene T., Pfeiffer B., Renard P., Guardiola-Lemaitre B., Manechez D., Penicaud L., Ktorza A., Godfroid J.
Journal of Medicinal Chemistry,
1997
6.

Chern J., Liaw Y., Chen C., Rong J., Huang C., Chan C., H.-J. Wang A.
Heterocycles,
1993
7.

Puntener K., Hellman M.D., Kuester E., Hegedus L.S.
Journal of Organic Chemistry,
2000
8.

Dalko P.I., Langlois Y.
Chemical Communications,
1998
9.

Morimoto T., Tachibana K., Achiwa K.
Synlett,
1997
10.

Liu H., Du D.
Advanced Synthesis and Catalysis,
2009
11.

Zhou L., Zhou J., Tan C.K., Chen J., Yeung Y.
Organic Letters,
2011
12.

Chen J., Yu W., Wei Y., Li T., Xu P.
Journal of Organic Chemistry,
2016
13.

Neef G., Eder U., Sauer G.
Journal of Organic Chemistry,
1981
14.

Rousselet G., Capdevielle P., Maumy M.
Tetrahedron Letters,
1993
15.

Gagnon J.L., Walters T.R., Zajac W.W., Buzby J.H.
Journal of Organic Chemistry,
1993
16.
![BF3·Et2O-Catalyzed Formal [3 + 2] Reaction of Aziridinofullerenes with Carbonyl Compounds](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yang H., Xing M., Zhu Y., Sun X., Cheng J., Miao C., Li F.
Journal of Organic Chemistry,
2014
17.

Mohammadpoor-Baltork I., Mirkhani V., Moghadam M., Tangestaninejad S., Zolfigol M.A., Abdollahi-Alibeik M., Khosropour A.R., Kargar H., Hojati S.F.
Catalysis Communications,
2008
18.

Bai G., Xu K., Chen G., Yang Y., Li T.
Synthesis,
2011
19.

Hsueh N., Clarkson G.J., Shipman M.
Organic Letters,
2015
20.
10.1016/j.mencom.2020.03.019_sbref0045l
Ishihara
Synthesis,
2007
21.

Gandhi S., Bisai A., Prasad B.A., Singh V.K.
Journal of Organic Chemistry,
2007
22.
![A New and Efficient Procedure for Bi(OTf)3‐Promoted [3+2] Cycloaddition of N‐Tosylaziridines to Yield Imidazolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Li X., Yang X., Chang H., Li Y., Ni B., Wei W.
European Journal of Organic Chemistry,
2011
23.

Ghorai M.K., Ghosh K., Das K.
Tetrahedron Letters,
2006
24.

Li R., Jiang H., Liu W., Gu P., Li X.
Chinese Chemical Letters,
2014
25.
![Silylmethyl-Substituted Aziridine and Azetidine as Masked 1,3- and 1,4-Dipoles for Formal [3 + 2] and [4 + 2] Cycloaddition Reactions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yadav V.K., Sriramurthy V.
Journal of the American Chemical Society,
2005
26.

Concellón J.M., Riego E., Suárez J.R., García-Granda S., Díaz M.R.
Organic Letters,
2004
27.

Chatterjee R., Samanta S., Mukherjee A., Santra S., Zyryanov G.V., Majee A.
Tetrahedron Letters,
2019
28.

Chakraborty Ghosal N., De A., Mahato S., Santra S., V. Zyryanov G., Majee A.
ChemistrySelect,
2018
29.

Samanta S., Chatterjee R., Mahato S., Hajra A., Santra S., Zyryanov G.V., Majee A.
Synthetic Communications,
2018
30.

Ghosal N., Mahato S., Chatterjee R., Santra S., Zyryanov G., Majee A.
SynOpen,
2017
31.

Ghosal N.C., Santra S., Zyryanov G.V., Hajra A., Majee A.
Tetrahedron Letters,
2016
32.

Chakraborty Ghosal N., Santra S., Das S., Hajra A., Zyryanov G.V., Majee A.
Green Chemistry,
2016
33.

Thakur V.V., Sudalai A.
Tetrahedron Letters,
2003