Keywords
13C NMR
Chan–Lam reaction
copper catalysis
DFT
nitropyrazoles
pyrazoles
vinylboronic acids
vinylpyrazoles
Abstract
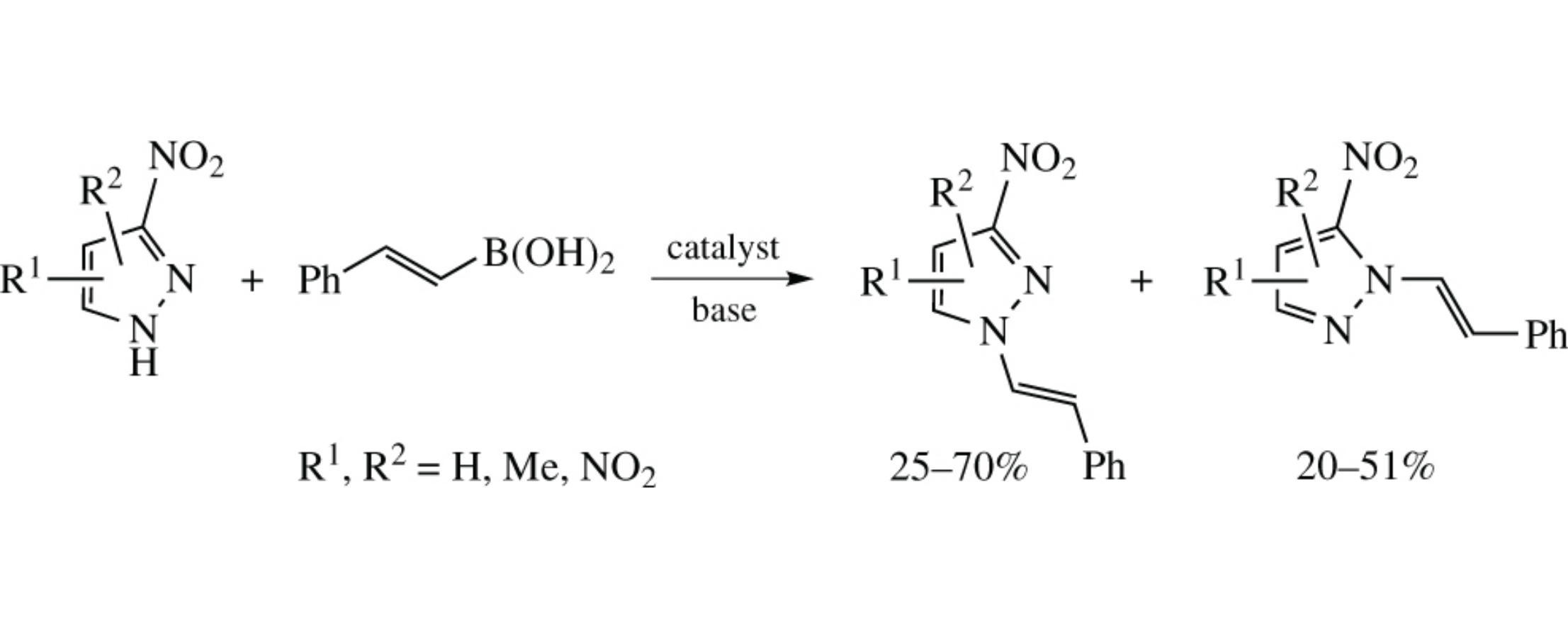
Regioselectivity of the Chan–Lam coupling of ambident nitropyrazoles with trans-styrylboronic acid depends on the base and catalyst nature and can vary the N(1)/N(2)-isomer ratio from ∼2 : 1 to ∼1 : 2. 2-Methyl-4-nitro- and 2,4-dinitroimidazoles are unreactive in this reaction. The structure of N(1)/N(2) isomers was elucidated by NOE measurements and by the comparison of experimental and DFT calculated 13C NMR chemical shifts.
References
1.
10.1016/j.mencom.2020.03.018_bib0005
Larina
Topics in Applied Chemistry. Nitroazoles: Synthesis, Structure and Application,
2009
2.

Ross W.J., Jamieson W.B., McCowen M.C.
Journal of Medicinal Chemistry,
1972
3.

Ross W.J., Jamieson W.B., McCowen M.C.
Journal of Medicinal Chemistry,
1973
4.

Ross W.J., Todd A.
Journal of Medicinal Chemistry,
1973
5.
10.1016/j.mencom.2020.03.018_sbref0015a
Preobrazhenskaya
1994
6.
10.1016/j.mencom.2020.03.018_sbref0015b
Clayton
Synthesis,
2005
7.

Clayton R., Ramsden C.A.
Journal of Heterocyclic Chemistry,
2004
8.

Obulesu O., Murugesh V., Harish B., Suresh S.
Journal of Organic Chemistry,
2018
9.

Grygorenko O.O., Nosik P.S.
Chemistry of Heterocyclic Compounds,
2019
10.

Ley S.V., Thomas A.W.
Angewandte Chemie - International Edition,
2003
11.

Beletskaya I.P., Cheprakov A.V.
Coordination Chemistry Reviews,
2004
12.

Beletskaya I.P., Cheprakov A.V.
Organometallics,
2012
13.

Fañanás-Mastral M.
Synthesis,
2017
14.

Taillefer M., Ouali A., Renard B., Spindler J.
Chemistry - A European Journal,
2006
15.

Chertkov V.A., Shestakova A.K., Davydov D.V.
Chemistry of Heterocyclic Compounds,
2011
16.

Davydov D.V., Oprunenko Y.F., Beletskaya I.P.
Tetrahedron Letters,
2017
17.
Davydov D.V., Chernyshev V.V., Rybakov V.B., Oprunenko Y.F., Beletskaya I.P.
Mendeleev Communications,
2018
18.
10.1016/j.mencom.2020.03.018_sbref0040a
Chan
Boronic Acids: Preparation and Application in Organic Synthesis and Medicine,
2005
19.

Qiao J., Lam P.
Synthesis,
2010
20.

Sanjeeva Rao K., Wu T.
Tetrahedron,
2012
21.

Munir I., Zahoor A.F., Rasool N., Naqvi S.A., Zia K.M., Ahmad R.
Molecular Diversity,
2018
22.

Kurpet M.K., Dąbrowska A., Jarosz M.M., Kajewska-Kania K., Kuźnik N., Suwiński J.W.
Beilstein Journal of Organic Chemistry,
2013
23.
![Di-μ-hydroxo-bis[(N,N,N′,N′-tetramethylethylenediamine)copper(II)] dichloride from X-ray powder data](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
Di-μ-hydroxo-bis[(N,N,N′,N′-tetramethylethylenediamine)copper(II)] dichloride from X-ray powder data
Albov D.V., Davydov D.V., Chernyshev V.V.
Acta Crystallographica Section E Structure Reports Online,
2004
24.
10.1016/j.mencom.2020.03.018_bib0055
Catalan
1987
25.

Zaitsev A.A., Dalinger I.L., Shevelev S.A.
Russian Chemical Reviews,
2009
26.

Newsoroff G., Sternhell S.
Australian Journal of Chemistry,
1968
27.
10.1016/j.mencom.2020.03.018_sbref0065b
Nies
J. Magn. Reson.,
1980
28.

Afonin A.V., Danovich D.K., Voronov V.K., Es'kova L.A., Baikalova L.V., Domina E.S.
Chemistry of Heterocyclic Compounds,
1990
29.
10.1016/j.mencom.2020.03.018_bib0075
Neese
Wiley Interdiscip. Rev.: Comput. Mol. Sci.,
2017
30.

Riveira M.J., Sarotti A.M.
Organic and Biomolecular Chemistry,
2018