Keywords
diazo compounds
diazonium salts
enones
pyrazoles
pyrazolo[5,1-c][1,2,4]triazines
trifluoromethyl group
Abstract
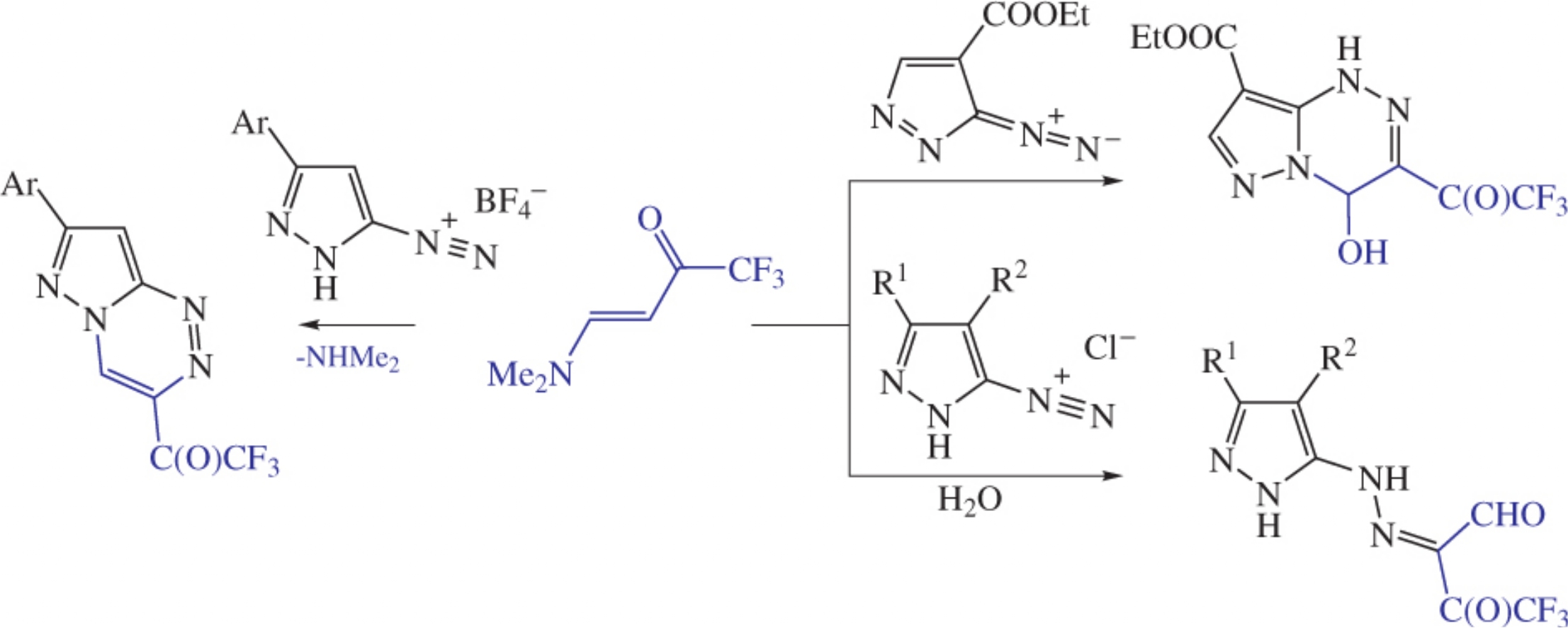
The reaction of 4-dimethylamino-1,1,1-trifluorobut-3-en-2one with 3-arylpyrazole-5-diazonium salt in MeCN affords 7-aryl-3-(trifluoroacetyl)pyrazolo[5,1-c][1,2,4]triazines. The transformation of relative 5-diazopyrazoles leads to 4-hydroxy-1,4-dihydropyrazolo[5,1-c][1,2,4]triazine derivatives, while the reaction in acidic aqueous solution results in 4,4,4-trifluoro-3-oxo-2-(pyrazol-5-ylhydrazono)butanals.
References
1.
10.1016/j.mencom.2020.03.016_sbref0005a
Lue
1996
2.

Elassar A.A., El-Khair A.A.
Tetrahedron,
2003
3.

Gaber H.M., Bagley M.C., Muhammad Z.A., Gomha S.M.
RSC Advances,
2017
4.

Al-Omran F., Khalik M.M., Abou-Elkhair A., Elnagdi M.H.
Synthesis,
1997
5.

Mach�?ek V., Ly?ka A., ?im?nek P., Weidlich T.
Magnetic Resonance in Chemistry,
2000
6.

Jiang H., Zhu S.
Journal of Fluorine Chemistry,
2008
7.

Alnajjar A., Abdelkhalik M.M., Al-Enezi A., Elnagdi M.H.
Molecules,
2008
8.
![Chemistry of 2-Arylhydrazonopropanals: Novel Synthesis of 1,6-Dihydropyridazines and 5-Heteroaryl Substituted Pyrazolo[1,5-a]Pyrimidines and Pyrazolo[3,4-b]Pyridines](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Abdel-Khalik M.M., Agamy S.M., Elnagdi M.H.
Synthesis,
2001
9.

Elnagdi M., Abdelhamid I., Nasra M.
Synlett,
2009
10.

Al-Shiekh M.A., El-Din A.M., Hafez E.A., Elnagdi M.H.
Journal of Chemical Research,
2004
11.

Al-Mousawi S., John E., Abdelkhalik M.M., Elnagdi M.H.
Journal of Heterocyclic Chemistry,
2003
12.
![1-Substituted 3-dimethylaminoprop-2-en-1-ones as building blocks in heterocyclic synthesis: New routes to 6-aroylpyridazin-3-ones, 4,6-diaroylpyridazin-3-imines and 3-aroylpyrazolo[5,1-c][1,2,4]triazines](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
13.
![2-(3-Arylhydrazono-3-Formyl-2-Oxopropyl)-1H-Isoindole-1,3(2H)-Dione in Heterocyclic Synthesis. Novel Derivatives of Pyridazin-6(1H)-One, Pyridazin-6(1H)-Imine, and Pyrazolo[5,1-c][1,2,4]Triazine Incorporating an N-(2-Oxoethyl)Phthalimide Moiety](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Al-Omran F., El-Khair A.A.
Journal of Chemical Research,
2006
14.
![Synthesis of some novel pyrazolo[1,5‐a]pyrimidine, 1,2,4‐triazolo[1,5‐a]pyrimidine, pyrido[2,3‐d]pyrimidine, pyrazolo[5,1‐c]‐1,2,4‐triazine and 1,2,4‐triazolo[5,1‐c]‐1,2,4‐triazine derivatives incorporating a thiazolo[3,2‐a]benzimidazole moiety](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Abdel-Aziz H.A., Hamdy N.A., Fakhr I.M., Farag A.M.
Journal of Heterocyclic Chemistry,
2008
15.
![An Efficient Single Step Synthesis of Pyridazine, Pyrazolo[5,1-c]-1,2,4-triazine, 1,2,4-Triazolo[5,1-c]-1,2,4-triazine and 1,2,4-Triazino[4,3-a]benzimidazole Derivatives](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
M. Farag A., R. Shaaban M., S. Saleh T.
Heterocycles,
2009
16.
![Convenient synthesis of some new pyrazolo[1,5‐a]pyrimidine, pyridine, thieno[2,3‐b]pyridine, and isoxazolo[3,4‐d]pyridazine derivatives containing benzofuran moiety](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Abdelhamid A.O.
Journal of Heterocyclic Chemistry,
2009
17.

A. Kheder N.
Heterocycles,
2009
18.

Hamama W.S., Berghot M.A., Baz E.A., Gouda M.A.
Archiv der Pharmazie,
2011
19.
![A new approach for the synthesis of some pyrazolo[5,1-c]triazines and pyrazolo[1,5-a]pyrimidines containing naphtofuran moiety](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Abdelhamid A.O., Shokry S.A., Tawfiek S.M.
Journal of Heterocyclic Chemistry,
2011
20.
Abdelhamid A.O., Fahmi A.A., Alsheflo A.A.
European Journal of Chemistry,
2012
21.
![A new convenient synthesis of 3-hetaryl-pyrazolo[5,1-c][1,2,4]triazines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Shawali A.S.
Journal of Advanced Research,
2012
22.
![Design and Synthesis of Some New Pyrazolo[1,5-a]pyrimidines, Pyrazolo[5,1-c]triazines, Pyrazolo[3,4-d]pyridazines, and Isoxazolo[3,4-d]pyridazines Containing the Pyrazole Moiety](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Abdelhamid A.O., Fahmi A.A., Halim K.N.
Synthetic Communications,
2013
23.
![Synthesis and reactivity of enaminone of naphtho[b]1,4-oxazine: One pot synthesis of novel isolated and heterocycle-fused derivatives with antimicrobial and antifungal activities](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
El Azab I.H., Khaled K.M.
Russian Journal of Bioorganic Chemistry,
2015
24.
![Synthesis of some new pyrazolo[1,5-a]pyrimidine, pyrazolo[5,1-c]triazine, 1,3,4-thiadiazole and pyridine derivatives containing 1,2,3-triazole moiety](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Abdelriheem N.A., Zaki Y.H., Abdelhamid A.O.
Chemistry Central Journal,
2017
25.

Sanad S.M., Mekky A.E.
Journal of Heterocyclic Chemistry,
2018
26.
![Synthesis, antibacterial activity and cytotoxicity of new fused pyrazolo[1,5-a]pyrimidine and pyrazolo[5,1-c][1,2,4]triazine derivatives from new 5-aminopyrazoles.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Al-Adiwish W.M., Tahir M.I., Siti-Noor-Adnalizawati A., Hashim S.F., Ibrahim N., Yaacob W.A.
European Journal of Medicinal Chemistry,
2013
27.
![Azolo[5,1-c]-1,2,4-triazines as a new class of antiviral compounds](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Rusinov V.L., Ulomskii E.N., Chupakhin O.N., Charushin V.N.
Russian Chemical Bulletin,
2008
28.
![Synthesis and Evaluation of Novel [1,2,4]Triazolo[5,1-c ][1,2,4]-triazines and Pyrazolo[5,1-c ][1,2,4]triazines as Potential Antidiabetic Agents](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Rusinov V.L., Sapozhnikova I.M., Bliznik A.M., Chupakhin O.N., Charushin V.N., Spasov A.A., Vassiliev P.M., Kuznetsova V.A., Rashchenko A.I., Babkov D.A.
Archiv der Pharmazie,
2017
29.

Cascioferro S., Parrino B., Spanò V., Carbone A., Montalbano A., Barraja P., Diana P., Cirrincione G.
European Journal of Medicinal Chemistry,
2017
30.

Druzhinin S.V., Balenkova E.S., Nenajdenko V.G.
Tetrahedron,
2007
31.
Nenajdenko V.G., Balenkova E.S.
Arkivoc,
2011
32.

Politanskaya L.V., Selivanova G.A., Panteleeva E.V., Tretyakov E.V., Platonov V.E., Nikul’shin P.V., Vinogradov A.S., Zonov Y.V., Karpov V.M., Mezhenkova T.V., Vasilyev A.V., Koldobskii A.B., Shilova O.S., Morozova S.M., Burgart Y.V., et. al.
Russian Chemical Reviews,
2019
33.
10.1016/j.mencom.2020.03.016_sbref0065a
Kirsch
Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications,
2013
34.
10.1016/j.mencom.2020.03.016_sbref0065b
2014
35.
10.1016/j.mencom.2020.03.016_sbref0065c
Fluorinated Heterocycles (ACS Symposium Series, Book 1003),
2009
36.
10.1016/j.mencom.2020.03.016_sbref0065d
Fluorinated Heterocyclic Compounds: Synthesis, Chemistry, and Applications,
2009
37.
10.1016/j.mencom.2020.03.016_sbref0065e
Reddy
Organofluorine Compounds in Biology and Medicine,
2015
38.
Alexeeva D.L., Sadchikova E.V., Volkova N.N., Efimov I.V., Jacobs J., Meervelt L.V., Dehaen W., Bakulev V.A.
Arkivoc,
2016
39.
![Synthesis and properties of 5-aryl-3-diazo-3H-pyrazoles and 3-aryl-1H-pyrazole-5-diazonium salts. Preparation and cytolytic activity studies of 2-arylpyrazolo-[5,1-c][1,2,4]benzotriazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Alekseeva D.L., Rakhimova V.Y., Minin A.S., Belousova A.V., Sadchikova E.V.
Chemistry of Heterocyclic Compounds,
2018
40.
Sadchikova E.V., Alexeeva D.L., Nenajdenko V.G.
Mendeleev Communications,
2019
41.

Sadchikova E.V., Mokrushin V.S.
Russian Chemical Bulletin,
2005
42.

Sanin A.V.
Synthesis,
1998
43.
10.1016/j.mencom.2020.03.016_bib0095
Spassova
Arzneim. Forsch.,
1977