Keywords
adsorption isotherm
adsorption kinetics
diffusion mechanism
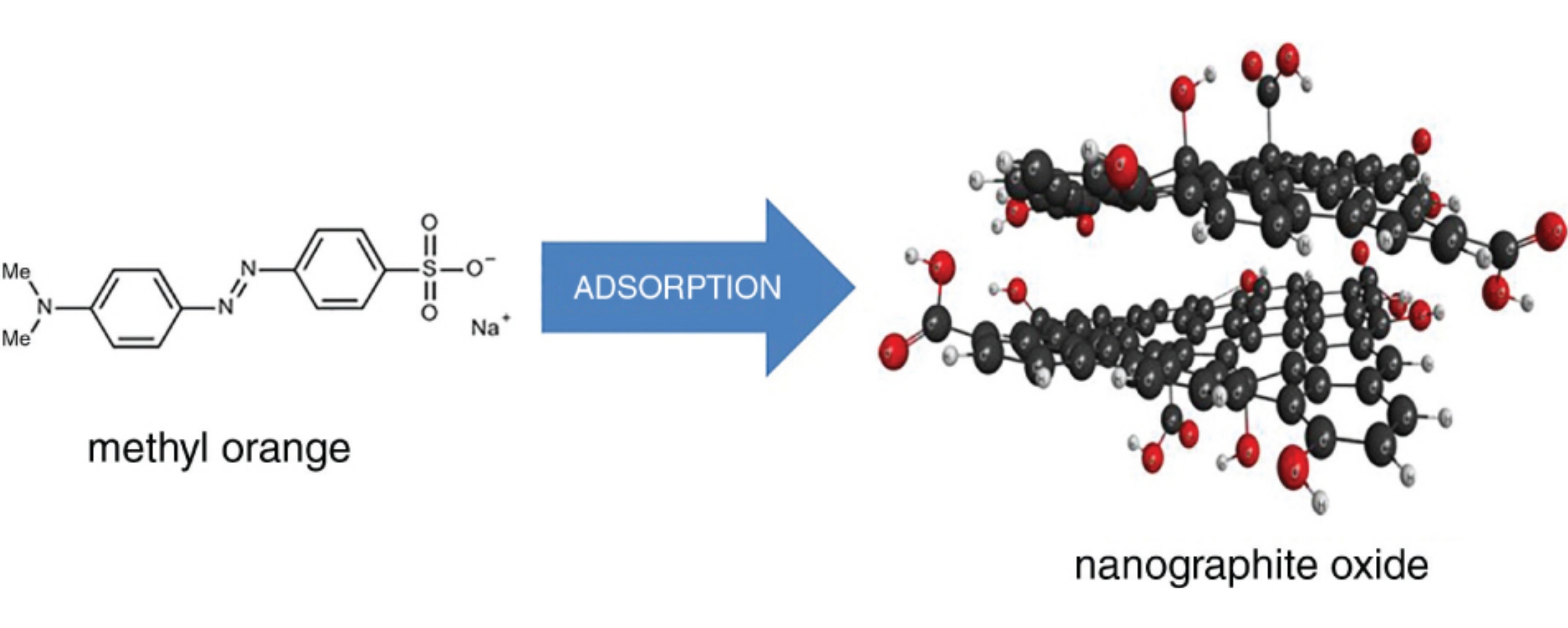
methyl orange
nanographite oxide
thermally expanded graphite
Abstract
Nanographite oxide was synthesized from thermally expanded graphite in an environmentally friendly electrochemical manner, and its adsorption properties were examined in a batch system. Three two-parameter isotherm models (Langmuir, Freundlich, and Dubinin–Radushkevich) were applied for the experimental data processing, and the adsorption kinetics was estimated according to pseudo-first and pseudo-second order simplified kinetic models. The acquired results revealed that obtaned nanographite oxide can be employed as an inexpensive and efficient adsorbent for removal of acidic dyes from aqueous solutions.
References
1.
10.1016/j.mencom.2020.03.014_bib0005
Kant
Nat. Sci.,
2012
2.

Chung K.
Environmental Carcinogenesis and Ecotoxicology Reviews,
2016
3.
10.1016/j.mencom.2020.03.014_bib0015
Hassaan
Am. J. Environ. Sci. Eng.,
2017
4.

Ahmad A., Mohd-Setapar S.H., Chuong C.S., Khatoon A., Wani W.A., Kumar R., Rafatullah M.
RSC Advances,
2015
5.
10.1016/j.mencom.2020.03.014_bib0025
Vital
J. Biorem. Biodegrad.,
2016
6.

Singh N.B., Nagpal G., Agrawal S., Rachna
Environmental Technology and Innovation,
2018
7.
10.1016/j.mencom.2020.03.014_bib0035
Rao
Int. J. Eng. Sci. Technol.,
2010
8.

Sheet I., Kabbani A., Holail H.
Energy Procedia,
2014
9.
10.1016/j.mencom.2020.03.014_bib0045
Hassan
Environmental Aspects of Textile Dyeing,
2007
10.
Li W., Lin X., Yu M., Mubeen I., Buekens A., Li X.
Aerosol and Air Quality Research,
2016
11.

Li C., Dong Y., Yang J., Li Y., Huang C.
Journal of Molecular Liquids,
2014
12.

Zhang B., Li F., Wu T., Sun D., Li Y.
Colloids and Surfaces A: Physicochemical and Engineering Aspects,
2015
13.
Obraztsova E.Y., Degtyarev A.A., Rukhov A.V., Bakunin E.S.
Vestnik Tambovskogo gosudarstvennogo tehnicheskogo universiteta,
2019
14.

Hummers W.S., Offeman R.E.
Journal of the American Chemical Society,
1958
15.
10.1016/j.mencom.2020.03.014_bib0075
Khan
Handbook of Materials Characterization,
2018
16.

Pal J., Deb M.K., Deshmukh D.K., Verma D.
Applied Water Science,
2013
17.
10.1016/j.mencom.2020.03.014_bib0085
Nandhini
Int. J. Trend Sci. Res. Dev.,
2017
18.
10.1016/j.mencom.2020.03.014_bib0090
Freundlich
J. Chem. Phys.,
1906
19.

Langmuir I.
Journal of the American Chemical Society,
1918
20.
10.1016/j.mencom.2020.03.014_bib0100
Dubinin
Dokl. Akad. Nauk SSSR,
1947
21.
10.1016/j.mencom.2020.03.014_bib0105
Lagergren
K. Sven. Vetenskapsakad. Handl.,
1898
22.

Ho Y.S., McKay G.
Process Biochemistry,
1999
23.

Boyd G.E., Adamson A.W., Myers L.S.
Journal of the American Chemical Society,
1947
24.

Weber W.J., Morris J.C.
Journal of the Sanitary Engineering Division,
2021
25.
10.1016/j.mencom.2020.03.014_bib0125
Samarghandi
Iran. J. Environ. Health Sci. Eng.,
2009