Keywords
diamines
nickel complexes
oxidation
panchromatic absorption
porphyrins
pyrazine-fused dimers
Abstract
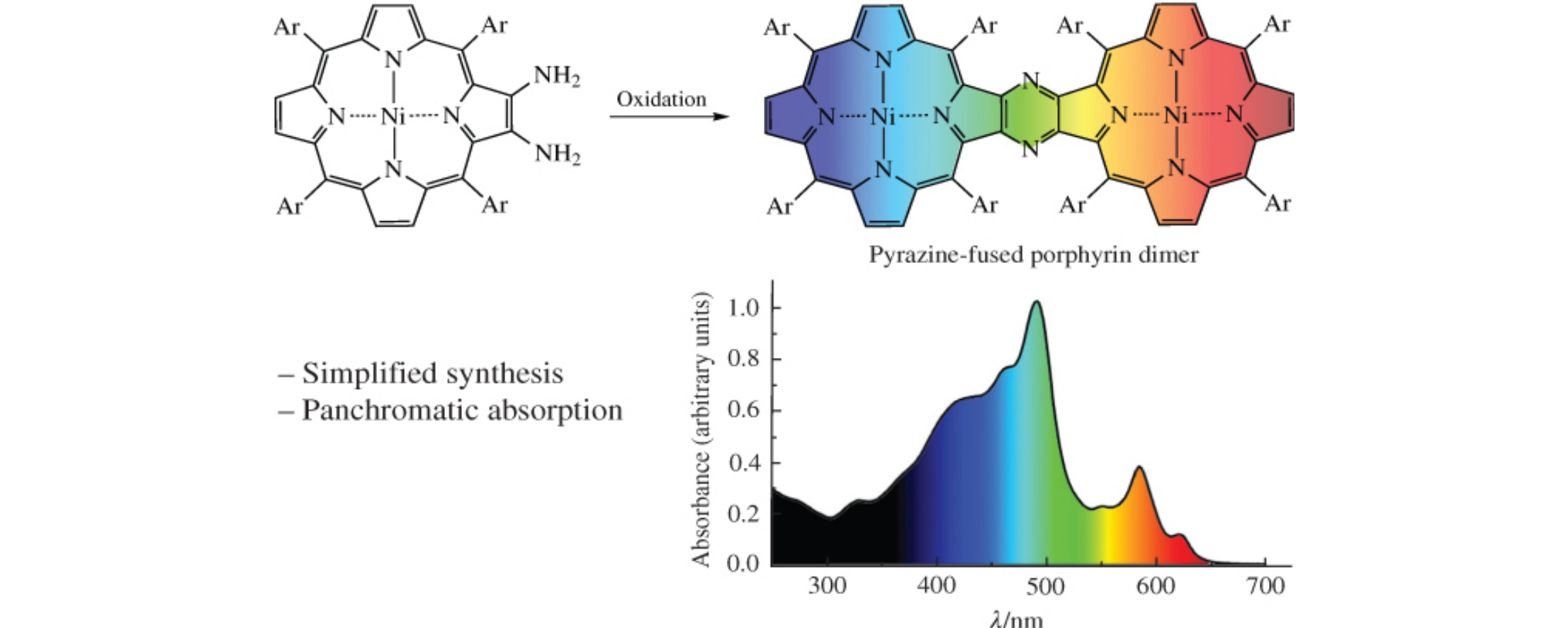
New pyrazine-fused porphyrin dimer was unexpectedly produced by mild oxidation of [2,3-diamino-5,10,15,20-tetrakis(4-butoxyphenyl)porphyrinato]nickel(ii) in 40% yield. The obtained porphyrin dimer demonstrates bathochromic shift of absorption bands compared to the reported analogues, along with panchromatic absorption in the 250–650nm range, which is promising for optical, photovoltaic and medical applications.
References
1.

Arslanov V.V., Kalinina M.A., Ermakova E.V., Raitman O.A., Gorbunova Y.G., Aksyutin O.E., Ishkov A.G., Grachev V.A., Tsivadze A.Y.
Russian Chemical Reviews,
2019
2.

Fang Y., Wang L., Xu W., Ou Z., Chen M., Cong L., Shan W., Ke X., Kadish K.M.
Inorganic Chemistry,
2019
3.

Kumar S., Jiang X., Shan W., Jinadasa R.G., Kadish K.M., Wang H.
Chemical Communications,
2018
4.

Hu Y., Yellappa S., Thomas M.B., Jinadasa R.G., Matus A., Shulman M., D'Souza F., Wang H.
Chemistry - An Asian Journal,
2017
5.

Kobayashi N., Numao M., Kondo R., Nakajima S., Osa T.
Inorganic Chemistry,
1991
6.

Vicente M.G., Cancilla M.T., Lebrilla C.B., Smith K.M.
Chemical Communications,
1998
7.
![Synthesis of a gable bis-porphyrin linked with a bicyclo[2.2.2]octadiene ring and its conversion into a conjugated planar bis-porphyrin](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ito S., Nakamoto K., Uno H., Murashima T., Ono N.
Chemical Communications,
2001
8.

Uno H., Nakamoto K., Kuroki K., Fujimoto A., Ono N.
Chemistry - A European Journal,
2007
9.

Uoyama H., Kim K., Kuroki K., Shin J., Nagata T., Okujima T., Yamada H., Ono N., Kim D., Uno H.
Chemistry - A European Journal,
2010
10.

Crossley M.J., Burn P.L., Langford S.J., Pyke S.M., Stark A.G.
Journal of the Chemical Society Chemical Communications,
1991
11.

Crossley M.J., Burn P.L., Chew S.S., Cuttance F.B., Newsom I.A.
Journal of the Chemical Society Chemical Communications,
1991
12.

Ou Z., Khoury T., Fang Y., Zhu W., Sintic P.J., Crossley M.J., Kadish K.M.
Inorganic Chemistry,
2013
13.

Ou Z., Zhu W., Sintic P.J., Fang Y., Crossley M.J., Kadish K.M.
Journal of Porphyrins and Phthalocyanines,
2012
14.

Crossley M.J., Burn P.L.
Journal of the Chemical Society Chemical Communications,
1987
15.

Crossley M.J., Govenlock L.J., Prashar J.K.
Journal of the Chemical Society Chemical Communications,
1995
16.

Akita M., Hiroto S., Shinokubo H.
Angewandte Chemie - International Edition,
2012
17.

Mandoj F., Nardis S., Pudi R., Lvova L., Fronczek F.R., Smith K.M., Prodi L., Genovese D., Paolesse R.
Dyes and Pigments,
2013
18.

Bruhn T., Witterauf F., Götz D.C., Grimmer C.T., Würtemberger M., Radius U., Bringmann G.
Chemistry - A European Journal,
2014
19.

Ito S., Hiroto S., Lee S., Son M., Hisaki I., Yoshida T., Kim D., Kobayashi N., Shinokubo H.
Journal of the American Chemical Society,
2014
20.

Ito S., Hiroto S., Ousaka N., Yashima E., Shinokubo H.
Chemistry - An Asian Journal,
2016
21.

Takiguchi A., Wakita M., Hiroto S., Shinokubo H.
Chemistry Letters,
2019
22.

Birin K.P., Poddubnaya A.I., Abdulaeva I.A., Gorbunova Y.G., Tsivadze A.Y.
Dyes and Pigments,
2018
23.

Abdulaeva I.A., Birin K.P., Michalak J., Romieu A., Stern C., Bessmertnykh-Lemeune A., Guilard R., Gorbunova Y.G., Tsivadze A.Y.
New Journal of Chemistry,
2016
24.

Abdulaeva I.A., Birin K.P., Gorbunova Y.G., Tsivadze A.Y., Bessmertnykh-Lemeune A.
Journal of Porphyrins and Phthalocyanines,
2018