Keywords
A431 cells
chlorin e6
glycoconjugate
photodynamic therapy
water soluble
Abstract
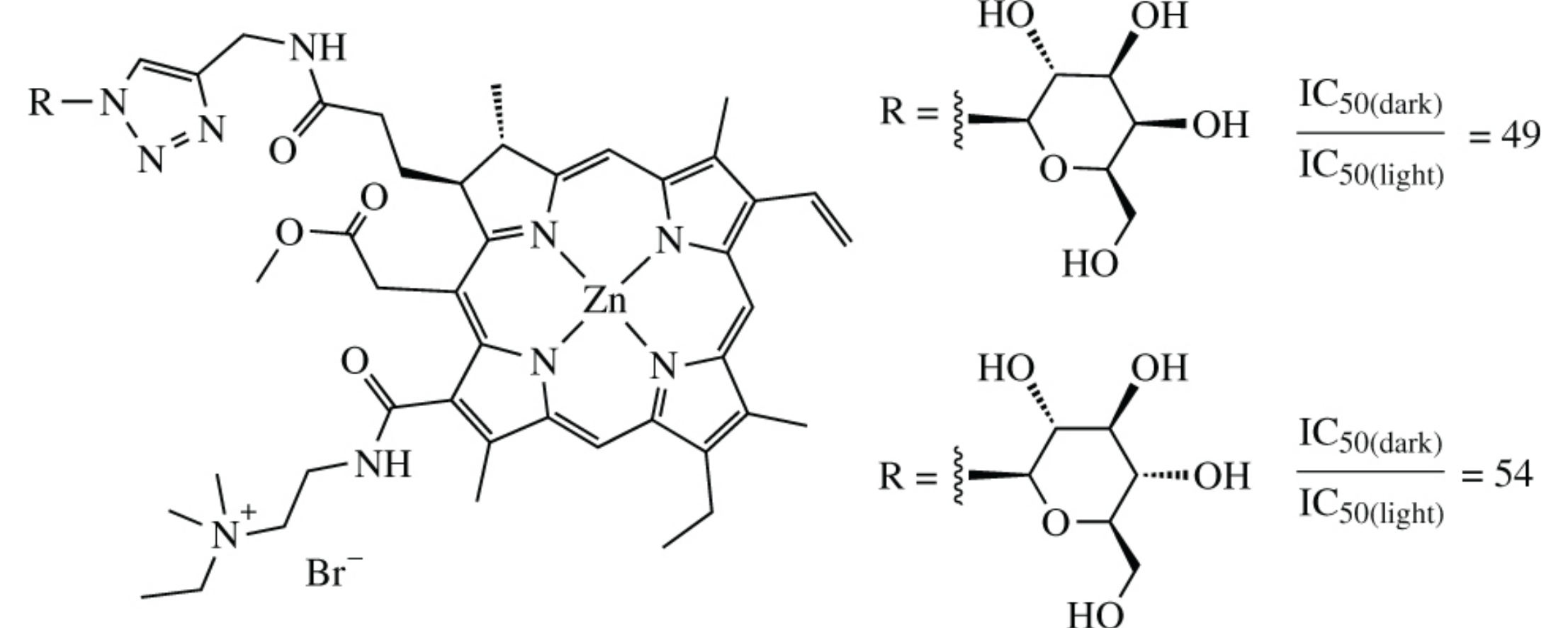
New water soluble conjugates of chlorin e6 derivative–zinc complex with β-d-galactose or β-d-glucose have been synthesized as potential agents for the photodynamic therapy of cancer. They exhibit photoinduced cytotoxicity at low micromolar concentrations with IC50(dark)/IC50(light) ratio of ∼50.
References
1.

QIANG Y., ZHANG X., LI J., HUANG Z.
Chinese Medical Journal,
2019
2.

Brown S.B., Brown E.A., Walker I.
The Lancet Oncology,
2004
3.

Li Y., Wang J., Zhang X., Guo W., Li F., Yu M., Kong X., Wu W., Hong Z.
Organic and Biomolecular Chemistry,
2015
4.

Liu L., Wang R., Wang C., Wang J., Chen L., Cheng J.
Biomaterials Science,
2018
5.

Otvagin V.F., Nyuchev A.V., Kuzmina N.S., Grishin I.D., Gavryushin A.E., Romanenko Y.V., Koifman O.I., Belykh D.V., Peskova N.N., Shilyagina N.Y., Balalaeva I.V., Fedorov A.Y.
European Journal of Medicinal Chemistry,
2018
6.

Fedorov A., Nyuchev A., Otvagin V., Gavryushin A., Romanenko Y., Koifman O., Belykh D., Schmalz H.
Synthesis,
2015
7.

Otvagin V.F., Kuzmina N.S., Krylova L.V., Volovetsky A.B., Nyuchev A.V., Gavryushin A.E., Meshkov I.N., Gorbunova Y.G., Romanenko Y.V., Koifman O.I., Balalaeva I.V., Fedorov A.Y.
Journal of Medicinal Chemistry,
2019
8.

Cancer cell–selective in vivo near infrared photoimmunotherapy targeting specific membrane molecules
Mitsunaga M., Ogawa M., Kosaka N., Rosenblum L.T., Choyke P.L., Kobayashi H.
Nature Medicine,
2011
9.

Mal'shakova M.V., Pylina Y.I., Belykh D.V.
Bioorganic and Medicinal Chemistry Letters,
2019
10.
Grin M.A., Lonin I.S., Makarov A.I., Lakhina A.A., Toukach F.V., Kachala V.V., Orlova A.V., Mironov A.F.
Mendeleev Communications,
2008
11.

Moret F., Gobbo M., Reddi E.
Photochemical and Photobiological Sciences,
2015
12.
Gradova M.A., Ostashevskaya I.I., Gradov O.V., Lobanov A.V., Lebedeva V.S., Mironov A.F.
Mendeleev Communications,
2018
13.

Mironov A.F., Zhdanova K.A., Bragina N.A.
Russian Chemical Reviews,
2018
14.

Singh S., Aggarwal A., Bhupathiraju N.V., Arianna G., Tiwari K., Drain C.M.
Chemical Reviews,
2015
15.

Hasan S.S., Ashraf G.M., Banu N.
Cancer Letters,
2007
16.

17.

Wu M., Li H., Liu R., Gao X., Zhang M., Liu P., Fu Z., Yang J., Zhang-Negrerie D., Gao Q.
European Journal of Medicinal Chemistry,
2016
18.

Tekade R.K., Sun X.
Drug Discovery Today,
2017
19.

Tamiaki H., Takeuchi S., Tsudzuki S., Miyatake T., Tanikaga R.
Tetrahedron,
1998
20.

Zenkevich E., Sagun E., Knyukshto V., Shulga A., Mironov A., Efremova O., Bonnett R., Songca S.P., Kassem M.
Journal of Photochemistry and Photobiology B: Biology,
1996
21.

Dąbrowski J.M., Pucelik B., Regiel-Futyra A., Brindell M., Mazuryk O., Kyzioł A., Stochel G., Macyk W., Arnaut L.G.
Coordination Chemistry Reviews,
2016
22.

Titov D.V., Gening M.L., Tsvetkov Y.E., Nifantiev N.E.
Russian Chemical Reviews,
2014
23.

Lin Y., Tungpradit R., Sinchaikul S., An F., Liu D., Phutrakul S., Chen S.
Journal of Medicinal Chemistry,
2008
24.

Liang H., Chen C., Chen S., Kulkarni A.R., Chiu Y., Chen M., Sung H.
Biomaterials,
2006
25.

Abellán Flos M., García Moreno M.I., Ortiz Mellet C., García Fernández J.M., Nierengarten J., Vincent S.P.
Chemistry - A European Journal,
2016
26.

Salunke S.B., Babu N.S., Chen C.
Chemical Communications,
2011
27.

Ameen M.A., Karsten S., Fenger R., Liebscher J.
Tetrahedron Letters,
2010
28.

Hein J.E., Fokin V.V.
Chemical Society Reviews,
2010