Keywords
3,7-diazabicyclo[3.3.1]nonane
AMPA receptor
NAM
negative allosteric modulator
patch clamp
Abstract
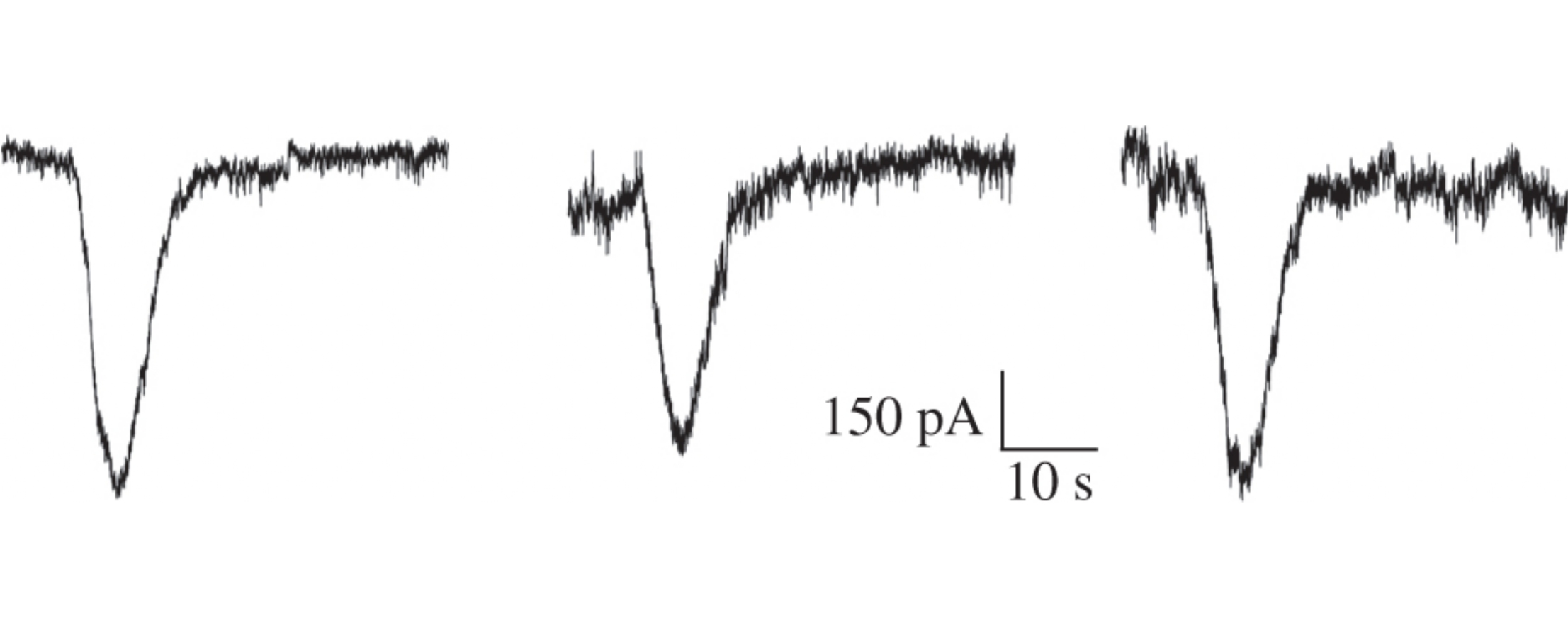
Synthesis of 6-[4-methoxy-3-(pyrrolidin-1-ylmethyl)benzyl]-1,11-dimethyl-3,6,9-triazatricyclo[7.3.1.13,11]tetradecane-4,8,12-trione has been carried out and optimized. In vitro studies using electrophysiological patch clamp technique have revealed a negative effect of this compound on kainateinduced currents in Purkinje neurons in a wide range of concentrations from 10−11 to 10−6m.
References
1.

Traynelis S.F., Wollmuth L.P., McBain C.J., Menniti F.S., Vance K.M., Ogden K.K., Hansen K.B., Yuan H., Myers S.J., Dingledine R.
Pharmacological Reviews,
2010
2.

Robbins T.W., Murphy E.R.
Trends in Pharmacological Sciences,
2006
3.

Brogi S., Campiani G., Brindisi M., Butini S.
ACS Medicinal Chemistry Letters,
2019
4.

Lynch G.
Current Opinion in Pharmacology,
2004
5.

Twomey E.C., Yelshanskaya M.V., Grassucci R.A., Frank J., Sobolevsky A.I.
Nature,
2017
6.
10.1016/j.mencom.2020.03.008_bib0030
O’Neill
IDrugs,
2007
7.

Reuillon T., E. Ward S., Beswick P.
Current Topics in Medicinal Chemistry,
2016
8.

Ring-fused thiadiazines as core structures for the development of potent AMPA receptor potentiators.
Pirotte B., Francotte P., Goffin E., Fraikin P., Danober L., Lesur B., Botez I., Caignard D.-., Lestage P., de Tullio P.
Current Medicinal Chemistry,
2010
9.

Drapier T., Geubelle P., Bouckaert C., Nielsen L., Laulumaa S., Goffin E., Dilly S., Francotte P., Hanson J., Pochet L., Kastrup J.S., Pirotte B.
Journal of Medicinal Chemistry,
2018
10.

Lauterborn J.C., Palmer L.C., Jia Y., Pham D.T., Hou B., Wang W., Trieu B.H., Cox C.D., Kantorovich S., Gall C.M., Lynch G.
Journal of Neuroscience,
2016
11.

Yelshanskaya M., Singh A., Sampson J., Narangoda C., Kurnikova M., Sobolevsky A.
Neuron,
2016
12.

Adler L.A., Kroon R.A., Stein M., Shahid M., Tarazi F.I., Szegedi A., Schipper J., Cazorla P.
Biological Psychiatry,
2012
13.
10.1016/j.mencom.2020.03.008_bib0065
Lee
2016
14.

Pirotte B., Francotte P., Goffin E., de Tullio P.
Expert Opinion on Therapeutic Patents,
2013
15.
Radchenko E.V., Karlov D.S., Lavrov M.I., Palyulin V.A.
Mendeleev Communications,
2017
16.

Lavrov M.I., Grigor’ev V.V., Bachurin S.O., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2015
17.
Lavrov M.I., Karlov D.S., Palyulin V.A., Grigoriev V.V., Zamoyski V.L., Brkich G.E., Pyatigorskaya N.V., Zapolskiy M.E.
Mendeleev Communications,
2018
18.

Nazarova A.A., Sedenkova K.N., Karlov D.S., Lavrov M.I., Grishin Y.K., Kuznetsova T.S., Zamoyski V.L., Grigoriev V.V., Averina E.B., Palyulin V.A.
MedChemComm,
2019
19.
![Novel Positive Allosteric Modulators of AMPA Receptors Based on 3,7-Diazabicyclo[3.3.1]nonane Scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Lavrov M.I., Karlov D.S., Voronina T.A., Grigoriev V.V., Ustyugov A.A., Bachurin S.O., Palyulin V.A.
Molecular Neurobiology,
2019
20.

Kuznetsov A.I., Basargin E.B., Moskovkin A.S., Ba M.K., Miroshnichenko I.V., Botnikov M.Y., Unkovskii B.V.
Chemistry of Heterocyclic Compounds,
1985
21.
![Synthesis of 3,7-diacyl-1,5-dimethyl-3,7-diazabicyclo[3.3.1]nonane derivatives as promising lipid bilayer modifiers](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Veremeeva P.N., Grishina I.V., Zaborova O.V., Averin A.D., Palyulin V.A.
Tetrahedron,
2019
22.

Perlovich G.L., Proshin A.N., Volkova T.V., Kurkov S.V., Grigoriev V.V., Petrova L.N., Bachurin S.O.
Journal of Medicinal Chemistry,
2009
23.

Ptak C.P., Ahmed A.H., Oswald R.E.
Biochemistry,
2009