Keywords
anti-stress effect
cytotoxicity
oxidative stress.
substituted ureas
synthetic cytokinins
Abstract
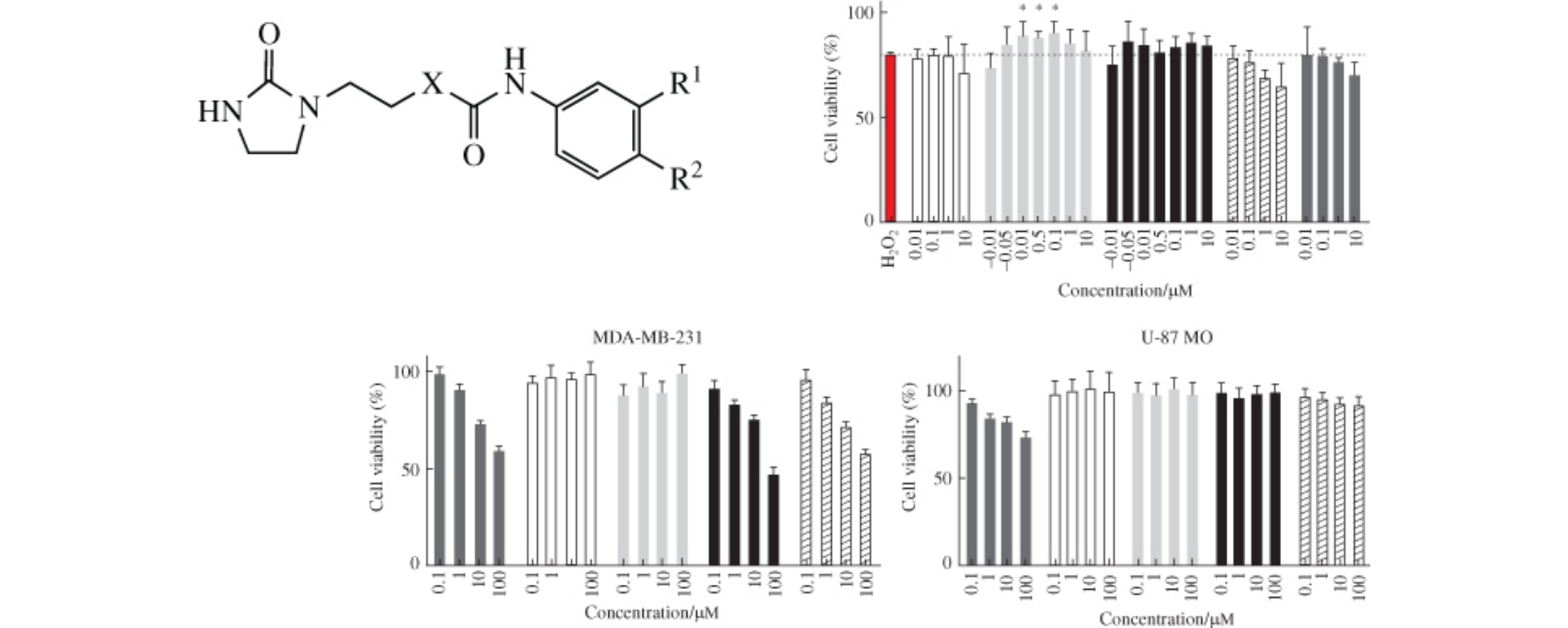
A series of aryl-substituted ureas and carbamates containing chlorinated aromatic and modified imidazolidinone moieties were synthesized. These compounds were found to be cytotoxic to breast cancer cell line MDA-MB-231, glioblastoma U-87 MG and neuroblastoma SH-SY5Y, but not to melanoma A-375.
References
1.

Laezza C., Caruso M.G., Gentile T., Notarnicola M., Malfitano A.M., Di Matola T., Messa C., Gazzerro P., Bifulco M.
International Journal of Cancer,
2009
2.
10.1016/j.mencom.2020.03.007_bib0010
Kovalenko
Butlerovskie Soobshcheniya,
2017
3.

Zalabák D., Pospíšilová H., Šmehilová M., Mrízová K., Frébort I., Galuszka P.
Biotechnology Advances,
2013
4.

Savelieva E.M., Oslovsky V.E., Karlov D.S., Kurochkin N.N., Getman I.A., Lomin S.N., Sidorov G.V., Mikhailov S.N., Osolodkin D.I., Romanov G.A.
Phytochemistry,
2018
5.

Natural and synthetic cytokinins and their applications in biotechnology, agrochemistry and medicine
Oshchepkov M.S., Kalistratova A.V., Savelieva E.M., Romanov G.A., Bystrova N.A., Kochetkov K.A.
Russian Chemical Reviews,
2020
6.

Voller J., Zatloukal M., Lenobel R., Doležal K., Béreš T., Kryštof V., Spíchal L., Niemann P., Džubák P., Hajdúch M., Strnad M.
Phytochemistry,
2010
7.

Othman E.M., Naseem M., Awad E., Dandekar T., Stopper H.
PLoS ONE,
2016
8.

Ciaglia E., Abate M., Laezza C., Pisanti S., Vitale M., Seneca V., Torelli G., Franceschelli S., Catapano G., Gazzerro P., Bifulco M.
International Journal of Cancer,
2016
9.

Ciaglia E., Laezza C., Abate M., Pisanti S., Ranieri R., D'alessandro A., Picardi P., Gazzerro P., Bifulco M.
International Journal of Cancer,
2017
10.

Voller J., Béres T., Zatloukal M., Kaminski P.A., Niemann P., Doležal K., Džubák P., Hajdúch M., Strnad M.
Phytochemistry,
2017
11.

Kittakoop P.
Studies in Natural Products Chemistry,
2015
12.
![Studies on the mechanisms of ozone tolerance: Cytokinin-like activity of N-[2-(2-oxo-1-imidazolidinyl)ethyl]-N'-phenylurea, a compound protecting against ozone injury](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
13.

Singh S., Agrawal S.B.
Water, Air, and Soil Pollution,
2010
14.
J. S. Kerr, G. A. Boswell, US Patent 5001141A, 1991.
15.

Anselmi C., Ettorre A., Andreassi M., Centini M., Neri P., Di Stefano A.
Biochemical Pharmacology,
2002
16.
Spindola D.G., Hinsberger A., Antunes V.M., Michelin L.F., Bincoletto C., Oliveira C.R.
Brazilian Journal of Pharmaceutical Sciences,
2018
17.

Wang Y., Tang Z., Xue R., Singh G.K., Liu W., Lv Y., Yang L.
Molecular and Cellular Biochemistry,
2011
18.

Jelluma N., Yang X., Stokoe D., Evan G.I., Dansen T.B., Haas-Kogan D.A.
Molecular Cancer Research,
2006
19.

Akimov M.G., Ashba A.M., Fomina-Ageeva E.V., Gretskaya N.M., Myasoedov N.F., Bezuglov V.V.
Doklady Biochemistry and Biophysics,
2019
20.
P. Lardenois, J. Frost, P. Pasau, P. George, M.C. Renones, R. Bartsch, W.-T. Li, P. Magat, R. Dupont, US Patent WO 009309, 1997.