Abstract
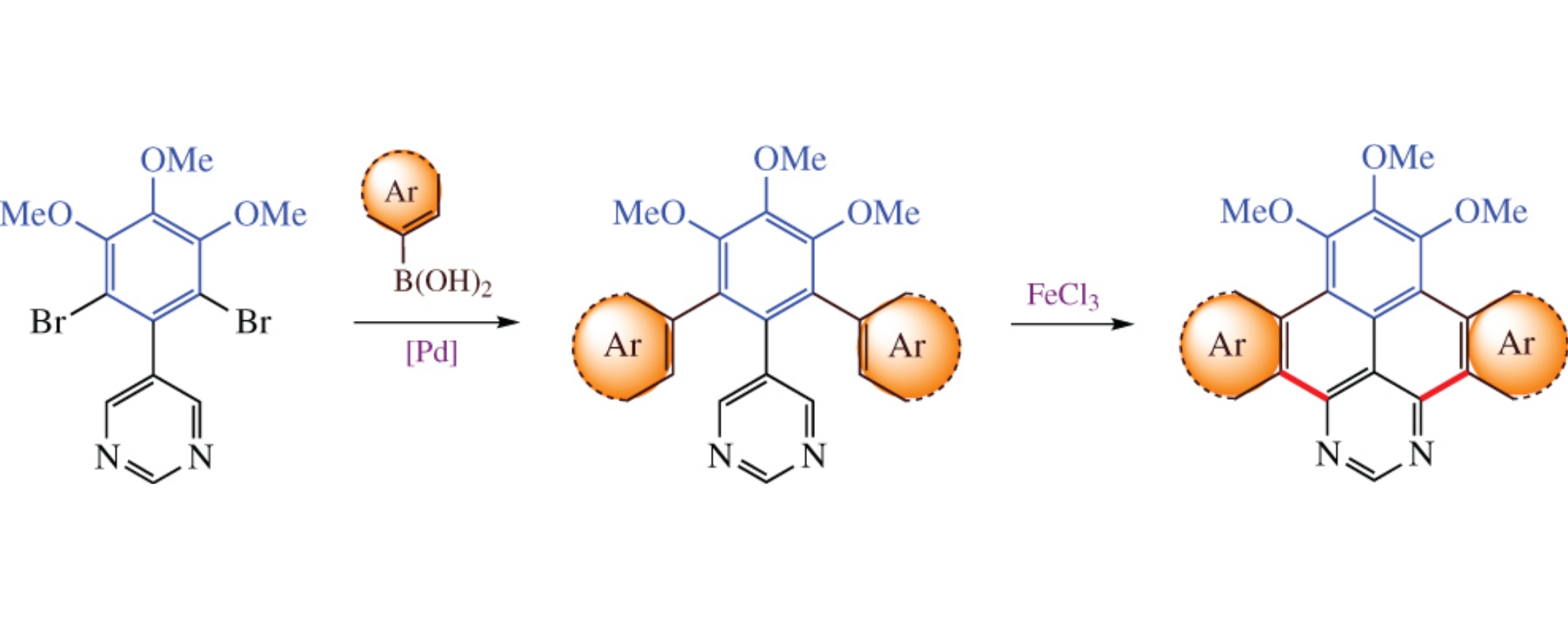
Di(het)areno[e,l][1,3]diazapyrene core was constructed by FeCl3-mediated intramolecular oxidative cyclodehydrogenation of 5-[2,6-di(het)arylphenyl]pyrimidine precursors, which in turn were obtained by the Suzuki cross-coupling of 5-(2,6-dibromophenyl)pyrimidine derivative with the corresponding (het)arylboronic acids. Molecular orbital calculations as well as redox and photophysical measurements show that the fused products are promising for organic electronic application.
References
1.

Figueira-Duarte T.M., Müllen K.
Chemical Reviews,
2011
2.

1,3,6,8-Tetraazapyrenes: Synthesis, Solid-State Structures, and Properties as Redox-Active Materials
Geib S., Martens S.C., Zschieschang U., Lombeck F., Wadepohl H., Klauk H., Gade L.H.
Journal of Organic Chemistry,
2012
3.

Martens S.C., Zschieschang U., Wadepohl H., Klauk H., Gade L.H.
Chemistry - A European Journal,
2012
4.

Stępień M., Gońka E., Żyła M., Sprutta N.
Chemical Reviews,
2016
5.

Wang X., Yao X., Narita A., Müllen K.
Accounts of Chemical Research,
2019
6.

Aksenov A.V., Shcherbakov S.V., Lobach I.V., Aksenova I.V., Rubin M.
European Journal of Organic Chemistry,
2017
7.

Verbitskiy E.V., Cheprakova E.M., Makarova N.I., Dorogan I.V., Metelitsa A.V., Minkin V.I., Slepukhin P.A., Svalova T.S., Ivanova A.V., Kozitsina A.N., Rusinov G.L., Chupakhin O.N., Charushin V.N.
European Journal of Organic Chemistry,
2016
8.
10.1016/j.mencom.2020.03.003_bib0040
Grzybowski
Angew. Chem., Int. Ed.,
2019
9.
Verbitskiy E.V., Rusinov G.L., Charushin V.N.
Arkivoc,
2017
10.

Verbitskiy E., Rusinov G., Chupakhin O., Charushin V.
Synthesis,
2017
11.

Verbitskiy E.V., Eltsov O.S., Zhilina E.F., Pakhomov I.M., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Tetrahedron,
2019
12.
K. Rurack, in Standardization and Quality Assurance in Fluorescence Measurements I. Techniques (Springer Series on Fluorescence, ed. M. Hof, vol. 5), ed. U. Resch-Genger, Springer, Berlin, 2008, pp. 101-145.
13.
10.1016/j.mencom.2020.03.003_bib0065
Ghalib
Aust. J. Basic Appl. Sci.,
2014
14.

Takimiya K., Shinamura S., Osaka I., Miyazaki E.
Advanced Materials,
2011