Keywords
activation energy
chain
cylinders
flame
palladium
penetration
platinum
termination.
Abstract
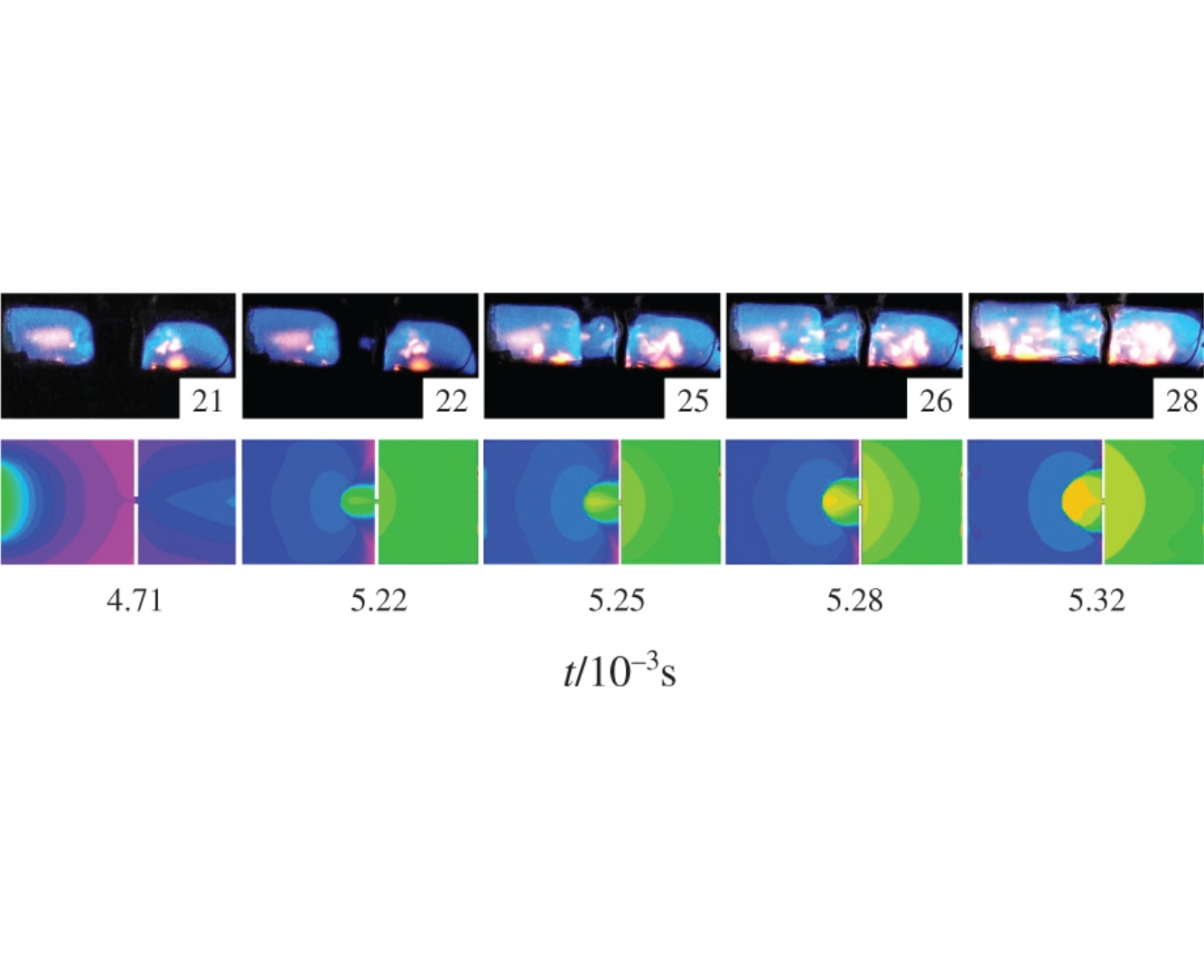
The value of effective activation energy of the dark reaction on Pd wire for 2H2+O2 reaction at 80 Torr has been estimated as 4.1±1kcal mol–1 that is characteristic of a surface process. It was demonstrated that at pressures up to 200 Torr, the rate of chain termination determines the critical diameter value in the flame penetration through Pt and Pd cylinders. The efficiency of Pd surface in the chain termination reaction was found much greater than that of Pt.
References
1.

Burns P.C., Ewing R.C., Navrotsky A.
Science,
2012
2.

Ten Hoeve J.E., Jacobson M.Z.
Energy and Environmental Science,
2012
3.

Sawarn T.K., Banerjee S., Kumar S.
Journal of Nuclear Materials,
2016
4.

Kelm S., Schoppe L., Dornseiffer J., Hofmann D., Reinecke E., Leistner F., Jühe S.
Nuclear Engineering and Design,
2009
5.

Wilberforce T., Alaswad A., Palumbo A., Dassisti M., Olabi A.G.
International Journal of Hydrogen Energy,
2016
6.

Woo C.H., Benziger J.B.
Chemical Engineering Science,
2007
7.

Dodds P.E., Staffell I., Hawkes A.D., Li F., Grünewald P., McDowall W., Ekins P.
International Journal of Hydrogen Energy,
2015
8.

Fernández A., Arzac G.M., Vogt U.F., Hosoglu F., Borgschulte A., Jiménez de Haro M.C., Montes O., Züttel A.
Applied Catalysis B: Environmental,
2016
9.

Petrov A.W., Ferri D., Tarik M., Kröcher O., van Bokhoven J.A.
Topics in Catalysis,
2016
10.
Rubtsov N.M., Chernysh V.I., Tsvetkov G.I., Troshin K.Y., Shamshin I.O., Kalinin A.P.
Mendeleev Communications,
2018
11.
Rubtsov N.M., Chernysh V.I., Tsvetkov G.I., Troshin K.Y., Shamshin I.O.
Mendeleev Communications,
2019
12.

Rubtsov N.M., Vinogradov A.N., Kalinin A.P., Rodionov A.I., Rodionov I.D., Troshin K.Y., Tsvetkov G.I., Chernysh V.I.
Russian Journal of Physical Chemistry B,
2019
13.
Rubtsov N.M., Chernysh V.I., Tsvetkov G.I., Troshin K.Y.
Mendeleev Communications,
2017
14.
10.1016/j.mencom.2020.01.041_bib0070
Repinski
Vvedenie v khimicheskuyu fiziku poverkhnosti tverdykh tel (Introduction into Chemical Physics of the Surface of Solids),
1993
15.
10.1016/j.mencom.2020.01.041_bib0075
Lewis
Combustion, Flames and Explosions of Gases,
1987
16.
10.1016/j.mencom.2020.01.041_bib0080
Semenov
O nekotorykh problemakh khimicheskoi kinetiki I reaktsionnoi sposobnosti (On Some Problems of Chemical Kinetics and Reactivity),
1958
17.
10.1016/j.mencom.2020.01.041_bib0085
Majda
Equations for Low Mach Number Combustion, Center for Pure and Applied Mathematics,
1982
18.
10.1016/j.mencom.2020.01.041_bib0090
Backstrom
Simple Fields of Physics by Finite Element Analysis,
2005