Keywords
carbon monoxide surrogates
formic acid esters
reduction
reductive amination
rhodium.
Abstract
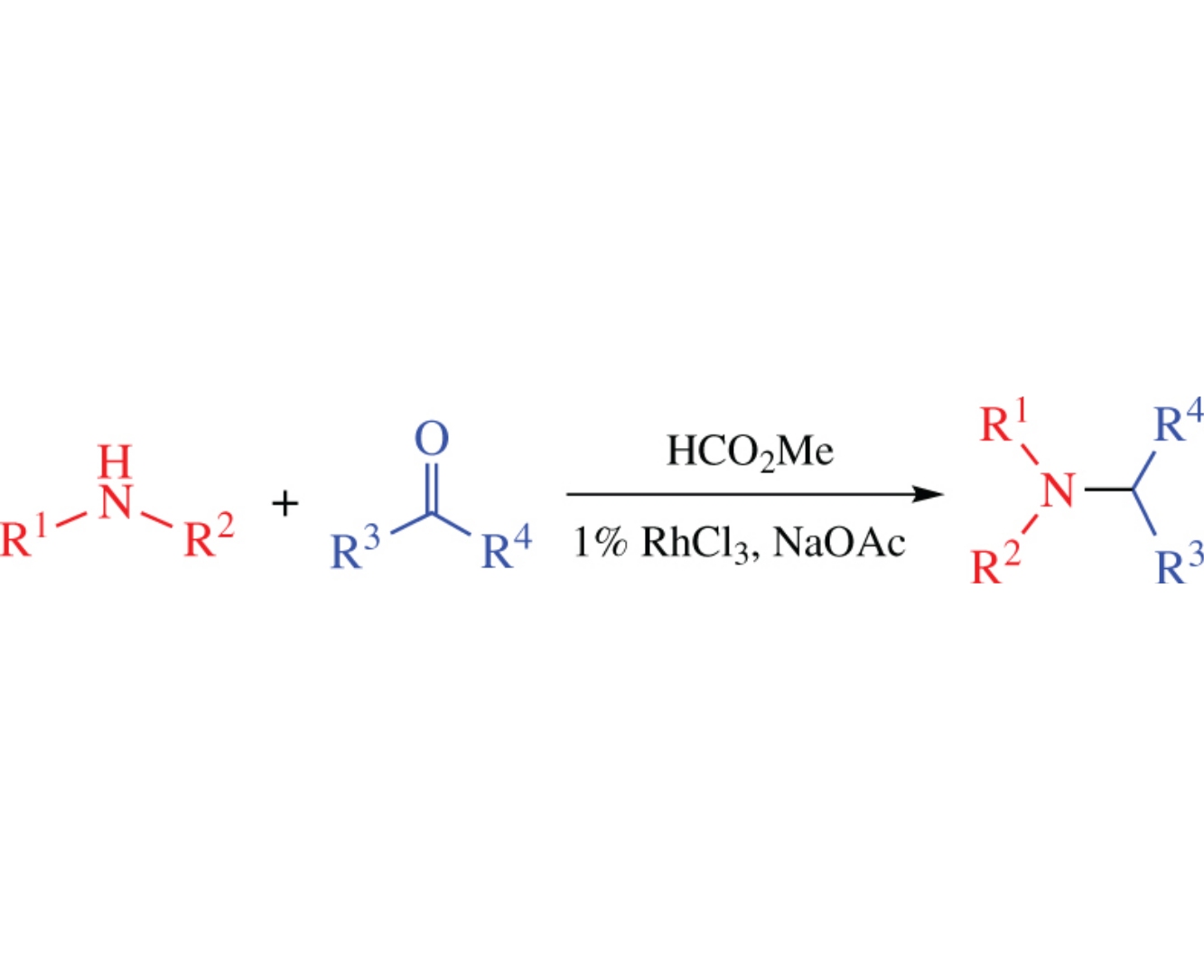
Alkyl formates in the presence of basic additives can serve as a reagent in the direct reductive amination of carbonyl compounds. The developed procedure can be applied to various aldehydes and ketones with electron donating and electron withdrawing groups.
References
1.
Vereshchagin A.N., Karpenko K.A., Elinson M.N., Dorofeeva E.O., Goloveshkin A.S., Egorov M.P.
Mendeleev Communications,
2018
2.
Trainov K.P., Salikov R.F., Luponosov Y.N., Savchenko P.S., Mannanov A.L., Ponomarenko S.A., Platonov D.N., Tomilov Y.V.
Mendeleev Communications,
2019
3.
Baranov V.V., Barsegyan Y.A., Kolotyrkina N.G., Kravchenko A.N.
Mendeleev Communications,
2019
4.

Gusak K.N., Ignatovich Z.V., Koroleva E.V.
Russian Chemical Reviews,
2015
5.

Abdel-Magid A.F., Carson K.G., Harris B.D., Maryanoff C.A., Shah R.D.
Journal of Organic Chemistry,
1996
6.

Nugent T., El-Shazly M.
Advanced Synthesis and Catalysis,
2010
7.

Tripathi R., Verma S., Pandey J., Tiwari V.
Current Organic Chemistry,
2008
8.

Podyacheva E., Afanasyev O.I., Tsygankov A.A., Makarova M., Chusov D.
Synthesis,
2019
9.

Chusov D., List B.
Angewandte Chemie - International Edition,
2014
10.

Shvydkiy N.V., Trifonova E.A., Shved A.M., Nelyubina Y.V., Chusov D., Perekalin D.S., Kudinov A.R.
Organometallics,
2016
11.
Tsygankov A.A., Makarova M., Chusov D.
Mendeleev Communications,
2018
12.

13.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Perekalin D.S., Shvydkiy N.V., Maleev V.I., Kudinov A.R., Chusov D.
ACS Catalysis,
2016
14.

Formenti D., Ferretti F., Ragaini F.
ChemCatChem,
2017
15.

Gockel S.N., Hull K.L.
Organic Letters,
2015
16.

Hermange P., Lindhardt A.T., Taaning R.H., Bjerglund K., Lupp D., Skrydstrup T.
Journal of the American Chemical Society,
2011
17.

Jana N., Zhou F., Driver T.G.
Journal of the American Chemical Society,
2015
18.

Afanasyev O.I., Usanov D.L., Chusov D.
Organic and Biomolecular Chemistry,
2017
19.

Afanasyev O.I., Zarochintsev A., Petrushina T., Cherkasova A., Denisov G., Cherkashchenko I., Chusova O., Jinho O., Man-Seog C., Usanov D.L., Semenov S.E., Chusov D.
European Journal of Organic Chemistry,
2018
20.

Li H., Neumann H., Beller M., Wu X.
Angewandte Chemie - International Edition,
2014
21.

Katafuchi Y., Fujihara T., Iwai T., Terao J., Tsuji Y.
Advanced Synthesis and Catalysis,
2011
22.

Chavan S.P., Bhanage B.M.
European Journal of Organic Chemistry,
2015
23.

Chang W., Li J., Ren W., Shi Y.
Organic and Biomolecular Chemistry,
2016
24.

Konishi H., Nagase H., Manabe K.
Chemical Communications,
2015
25.

Ueda T., Konishi H., Manabe K.
Organic Letters,
2012
26.

Fujihara T., Hosoki T., Katafuchi Y., Iwai T., Terao J., Tsuji Y.
Chemical Communications,
2012
27.

Aavula S.K., Chikkulapalli A., Hanumanthappa N., Jyothi I., Vinod Kumar C.H., Manjunatha S.G.
Tetrahedron Letters,
2013