Keywords
Acetylene
arylamines
ethynylation.
imines
pyrroles
superbases
vinylation
Abstract
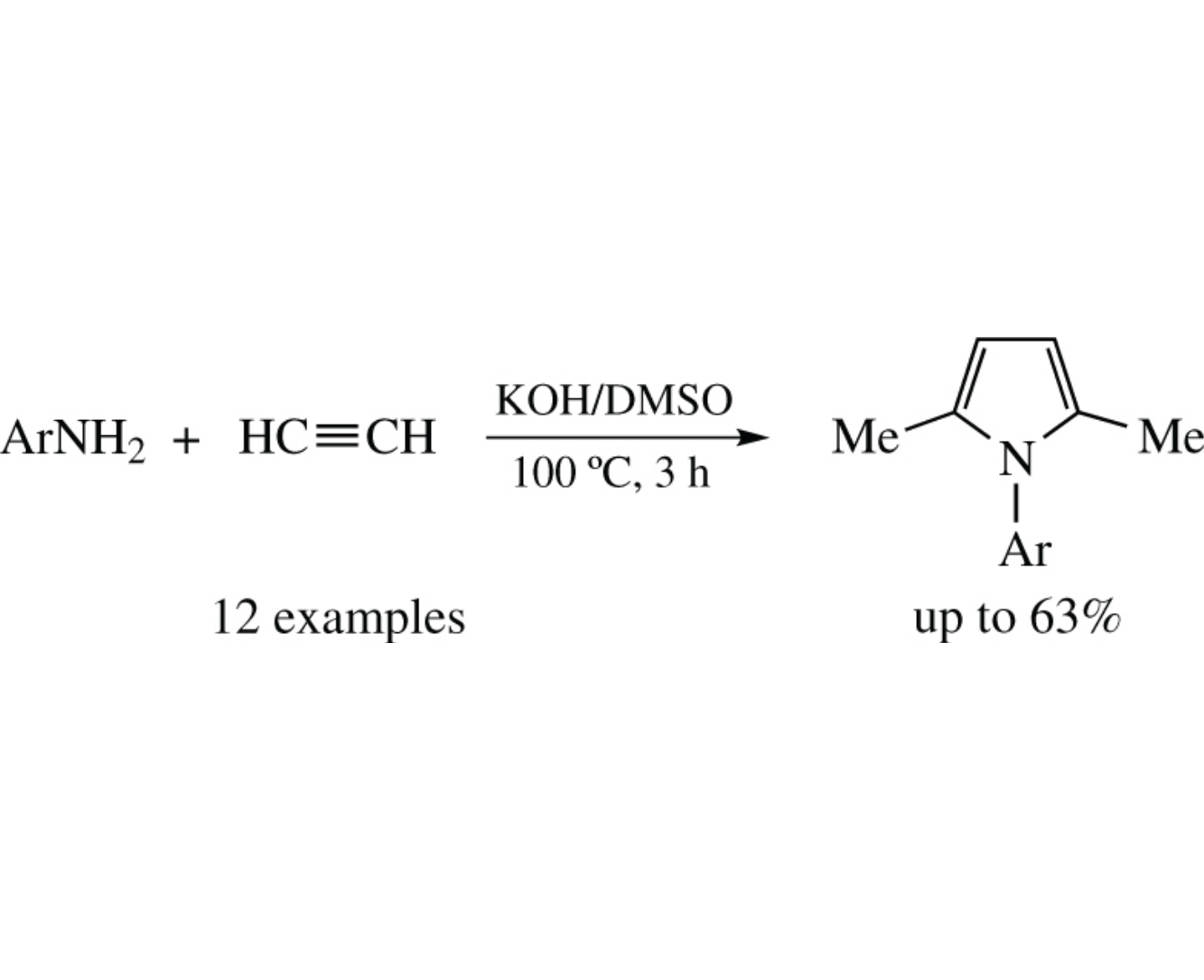
A new superbase-promoted reaction of acetylene involves self-organization of its three molecules with one molecule of arylamine in KOH/DMSO system to afford 1-aryl-2,5- dimethylpyrroles in up to 63% yields. The key step of this reaction cascade is assumed to be the nucleophilic addition of acetylene to the C=N bond of the intermediate aldimine (aza-Favorsky reaction).
References
1.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
2.

Clarke P., Ermanis K.
Current Organic Chemistry,
2013
3.
![Pot, atom and step economic synthesis: a diversity-oriented approach to construct 2-substituted pyrrolo[2,1-f][1,2,4]triazin-4(3H)-ones](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Xiang H., Chen Y., He Q., Xie Y., Yang C.
RSC Advances,
2013
4.

Jyoti Kalita S., Mecadon H., Deka D.C.
Tetrahedron Letters,
2015
5.
Vereshchagin A.N., Elinson M.N., Anisina Y.E., Ryzhkov F.V., Goloveshkin A.S., Bushmarinov I.S., Zlotin S.G., Egorov M.P.
Mendeleev Communications,
2015
6.

Rodygin K.S., Werner G., Kucherov F.A., Ananikov V.P.
Chemistry - An Asian Journal,
2016
7.

Voronin V.V., Ledovskaya M.S., Bogachenkov A.S., Rodygin K.S., Ananikov V.P.
Molecules,
2018
8.

Alabugin I.V., Gonzalez-Rodriguez E.
Accounts of Chemical Research,
2018
9.

Heravi M.M., Dehghani M., Zadsirjan V., Ghanbarian M.
Current Organic Synthesis,
2019
10.

Alyabyev S.B., Beletskaya I.P.
Russian Chemical Reviews,
2018
11.

Li G., Liu Q., Liu Z., Zhang Z.C., Li C., Wu W.
Angewandte Chemie - International Edition,
2010
12.

Schobert H.
Chemical Reviews,
2013
13.

Trotuş I., Zimmermann T., Schüth F.
Chemical Reviews,
2013
14.

Olmstead W.N., Margolin Z., Bordwell F.G.
Journal of Organic Chemistry,
1980
15.

Yuan Y., Thomé I., Kim S.H., Chen D., Beyer A., Bonnamour J., Zuidema E., Chang S., Bolm C.
Advanced Synthesis and Catalysis,
2010
16.

Trofimov B.A., Schmidt E.Y.
Accounts of Chemical Research,
2018
17.
![One-Pot Assembly of 7-Methylene-6,8-dioxabicyclo[3.2.1]octanes, Congeners of Frontalin, from Ketones and Acetylene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Trofimov B.A., Schmidt E.Y., Ushakov I.A., Mikhaleva A.I., Zorina N.V., Protsuk N.I., Senotrusova E.Y., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2009
18.

Schmidt E.Y., Trofimov B.A., Bidusenko I.A., Cherimichkina N.A., Ushakov I.A., Protzuk N.I., Gatilov Y.V.
Organic Letters,
2014
19.

Schmidt E.Y., Bidusenko I.A., Cherimichkina N.A., Ushakov I.A., Borodina T.N., Smirnov V.I., Trofimov B.A.
Chemistry - A European Journal,
2015
20.
![One-pot assembly of 4-methylene-3-oxa-1-azabicyclo[3.1.0]hexanes from alkyl aryl(hetaryl) ketoximes, acetylene, and aliphatic ketones: a new three-component reaction](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Trofimov B.A., Schmidt E.Y., Mikhaleva A.I., Ushakov I.A., Protsuk N.I., Senotrusova E.Y., Kazheva O.N., Aleksandrov G.G., Dyachenko O.A.
Tetrahedron Letters,
2009
21.

Bidusenko I.A., Schmidt E.Y., Ushakov I.A., Trofimov B.A.
European Journal of Organic Chemistry,
2018
22.
10.1016/j.mencom.2020.01.036_sbref0055a
Trofimov
Zh. Org. Khim.,
1979
23.
10.1016/j.mencom.2020.01.036_sbref0055b
Trofimov
Zh. Org. Khim.,
1976
24.

Schulte K.E., Reisch J., Walker H.
Chemische Berichte,
1965
25.

Zheng Q., Hua R.
Tetrahedron Letters,
2010
26.

Lee H., Kim B.H.
Tetrahedron,
2013