Keywords
acenaphthenes
aluminum hydrides
diimine ligands
imines
redox-active ligands
sterically hindered ligands
Abstract
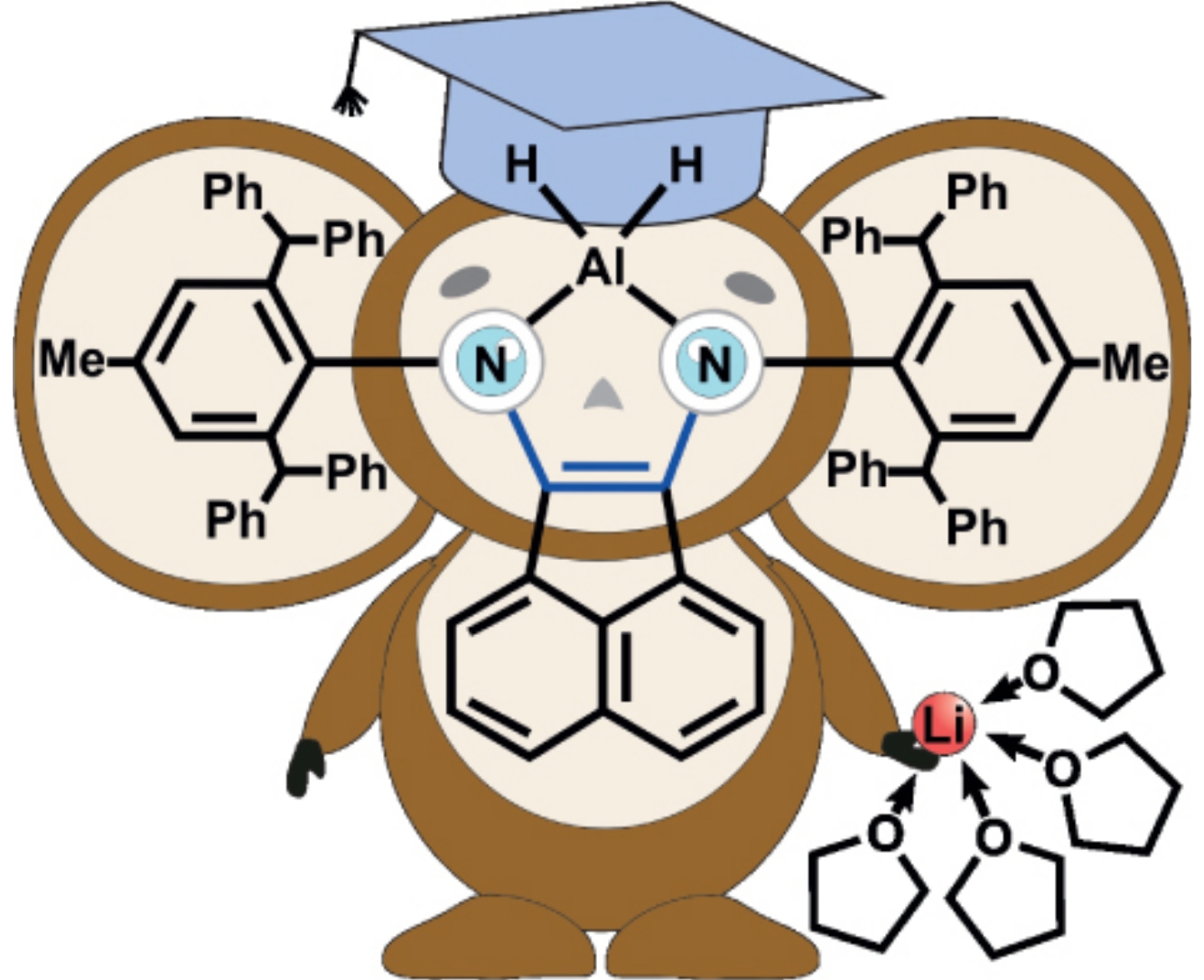
Treatment of a bulky dbhmp-bian ligand (dbhmp-bian==1,2-bis[(2,6-dibenzhydryl-4-methylphenyl)imino]acenaphthene) by 1 equiv. of LiAlH4 or mixture of 3/4 equiv. LiAlH4 and 1/3 eqiuv. AlCl3 in tetrahydrofuran results in new hydrides [(dbhmp-bian)AlH2(THF)]-[Li(THF)4]+ and (dbhmp-bian)AlH(THF), respectively. Both complexes were characterized by spectroscopic (IR, NMR) and X-ray diffraction methods.
References
1.

Hydroalumination and hydrogallation of alkynes: New insights into the course of well-known reactions
UHL W.
Coordination Chemistry Reviews,
2008
2.

Li W., Ma X., Walawalkar M.G., Yang Z., Roesky H.W.
Coordination Chemistry Reviews,
2017
3.

Liu Y., Li J., Ma X., Yang Z., Roesky H.W.
Coordination Chemistry Reviews,
2018
4.

Yang Z., Hao P., Liu Z., Ma X., Roesky H.W., Sun K., Li J.
Organometallics,
2012
5.

Keyes L.K., Todd A.D., Giffin N.A., Veinot A.J., Hendsbee A.D., Robertson K.N., Geier S.J., Masuda J.D.
RSC Advances,
2017
6.
![Syntheses and Structural Characterization of a LAl(N3)N[μ-Si(N3)(tBu)]2NAl(N3)L and a Monomeric Aluminum Hydride Amide LAlH(NHAr) (L = HC[(CMe)(NAr)]2, Ar = 2,6-iPr2C6H3)](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhu H., Yang Z., Magull J., Roesky H.W., Schmidt H., Noltemeyer M.
Organometallics,
2005
7.
![Reactivity Studies of LAlH2 [L = HC(CMeNAr)2, Ar = 2, 6‐iPr2C6H3] with 2‐[(2‐Hydroxybenzylidene)amino]‐3‐mercaptopropionic Acid and Benzene‐1, 2‐diamine](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ma X., Hao P., Li J., Roesky H.W., Yang Z.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2013
8.

Zheng W., Roesky H.W.
Journal of the Chemical Society Dalton Transactions,
2002
9.

Chen I., Ho S., Chen Y., Lin C., Hu C., Tu C., Datta A., Huang J., Lin C.
Dalton Transactions,
2009
10.

Kuo P., Chen I., Chang J., Lee M., Hu C., Hung C., Lee H., Huang J.
European Journal of Inorganic Chemistry,
2004
11.

Yang Z., Zhong M., Ma X., Nijesh K., De S., Parameswaran P., Roesky H.W.
Journal of the American Chemical Society,
2016
12.

Yang Z., Zhong M., Ma X., De S., Anusha C., Parameswaran P., Roesky H.W.
Angewandte Chemie - International Edition,
2015
13.

Jakhar V.K., Barman M.K., Nembenna S.
Organic Letters,
2016
14.

Franz D., Sirtl L., Pöthig A., Inoue S.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2016
15.

Myers T.W., Berben L.A.
Journal of the American Chemical Society,
2013
16.

Sherbow T.J., Carr C.R., Saisu T., Fettinger J.C., Berben L.A.
Organometallics,
2015
17.

Berben L.A.
Chemistry - A European Journal,
2014
18.

Sokolov V.G., Koptseva T.S., Moskalev M.V., Piskunov A.V., Samsonov M.A., Fedushkin I.L.
Russian Chemical Bulletin,
2017
19.

Sokolov V.G., Koptseva T.S., Moskalev M.V., Baranov E.V., Fedyushkin I.L.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2019
20.

Guo L., Kong W., Xu Y., Yang Y., Ma R., Cong L., Dai S., Liu Z.
Journal of Organometallic Chemistry,
2018
21.

Wang F., Chen C.
Polymer Chemistry,
2019
22.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
23.
10.1016/j.mencom.2020.01.031_bib0115
Sheldrick
SHELXTL, Version 6.14,
2003
24.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009