Keywords
closo-borate anions
carbonyl derivatives
DFT
QTAIM analysis
reactivity descriptors
Abstract
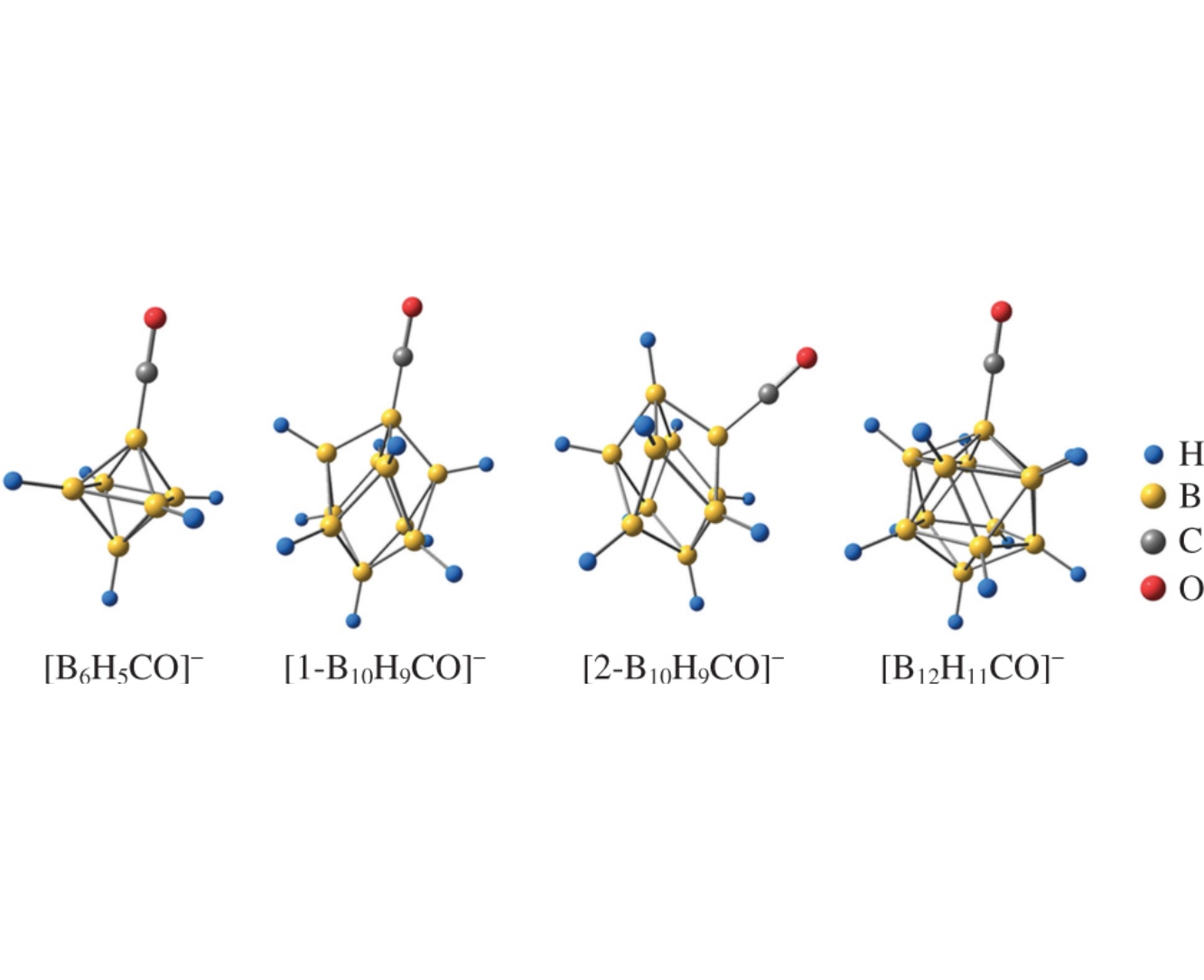
The structure, bonding, and reactivity of the monocarbonyl derivatives of closo-borate anions [BnHn–1CO]– (n=6, 10, 12) have been analyzed. The B–B, B–H, B–C and C–O bonding interactions in such anions have been theoretically examined using the Quantum Theory of Atoms in Molecules (QTAIM). Several local and integral topological properties of the electron density involved in these interactions have been computed, and different chemical reactivity descriptors have been calculated via conceptual density functional theory (DFT).
References
1.
![Theoretical QTAIM, ELI-D, and Hirshfeld Surface Analysis of the Cu–(H)B Interaction in [Cu2(bipy)2B10H10]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vologzhanina A.V., Korlyukov A.A., Avdeeva V.V., Polyakova I.N., Malinina E.A., Kuznetsov N.T.
Journal of Physical Chemistry A,
2013
2.

Kochnev V.K., Kuznetsov N.T.
Computational and Theoretical Chemistry,
2016
3.

Zhang Y., Sun Y., Lin F., Liu J., Duttwyler S.
Angewandte Chemie - International Edition,
2016
4.

Kaszyński P., Ringstrand B.
Angewandte Chemie - International Edition,
2015
5.

Olid D., Núñez R., Viñas C., Teixidor F.
Chemical Society Reviews,
2013
6.

Semioshkin A.A., Sivaev I.B., Bregadze V.I.
Dalton Transactions,
2008
7.
![Derivatives of closo-decaborate anion [B10H10]2− with exo-polyhedral substituents](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Zhizhin K.Y., Zhdanov A.P., Kuznetsov N.T.
Russian Journal of Inorganic Chemistry,
2010
8.

Duchêne L., Kühnel R.-., Rentsch D., Remhof A., Hagemann H., Battaglia C.
Chemical Communications,
2017
9.

Shakirova O.G., Lavrenova L.G., Bogomyakov A.S., Zhizhin K.Y., Kuznetsov N.T.
Russian Journal of Inorganic Chemistry,
2015
10.

Shakirova O.G., Daletskii V.A., Lavrenova L.G., Trubina S.V., Erenburg S.B., Zhizhin K.Y., Kuzhetsov N.T.
Russian Journal of Inorganic Chemistry,
2013
11.

Losytskyy M.Y., Kovalska V.B., Varzatskii O.A., Kuperman M.V., Potocki S., Gumienna-Kontecka E., Zhdanov A.P., Yarmoluk S.M., Voloshin Y.Z., Zhizhin K.Y., Kuznetsov N.T., Elskaya A.V.
Journal of Luminescence,
2016
12.

Kuperman M., Chernii S., Varzatskii O., Zhdanov A., Bykov A., Zhizhin K., Yarmoluk S., Kovalska V.
ChemistrySelect,
2017
13.
![Synthesis of monosubstituted derivatives of closo-decahydrodecaborate(2-). X-ray crystal structures of [closo-2-B10H9CO]- and [closo-2-B10H9NCO]2-](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Shelly K., Knobler C.B., Hawthorne M.F.
Inorganic Chemistry,
1992
14.
10.1016/j.mencom.2020.01.029_bib0070
Muetterties
Inorg. Chem.,
1963
15.

Qin X., Wu H., Jiao H.
Journal of Molecular Structure THEOCHEM,
2007
16.

Qin X., Wu H., Jiao H.
Journal of Molecular Structure THEOCHEM,
2007
17.

Mierzwa G., Gordon A.J., Berski S.
Polyhedron,
2019
18.

Alikhani M.E.
Physical Chemistry Chemical Physics,
2013
19.

Neese F.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2011
20.

Bader R.F., Legare D.A.
Canadian Journal of Chemistry,
1992
21.

Dobrott R.D., Lipscomb W.N.
Journal of Chemical Physics,
1962
22.

23.

Domingo L., Ríos-Gutiérrez M., Pérez P.
Molecules,
2016
24.

Domingo L.R., Aurell M.J., Pérez P., Contreras R.
Tetrahedron,
2002