Keywords
activation energy of thermal fluorescence quenching
fluorescence
pH
photoexcitation energy
tryptophan
Abstract
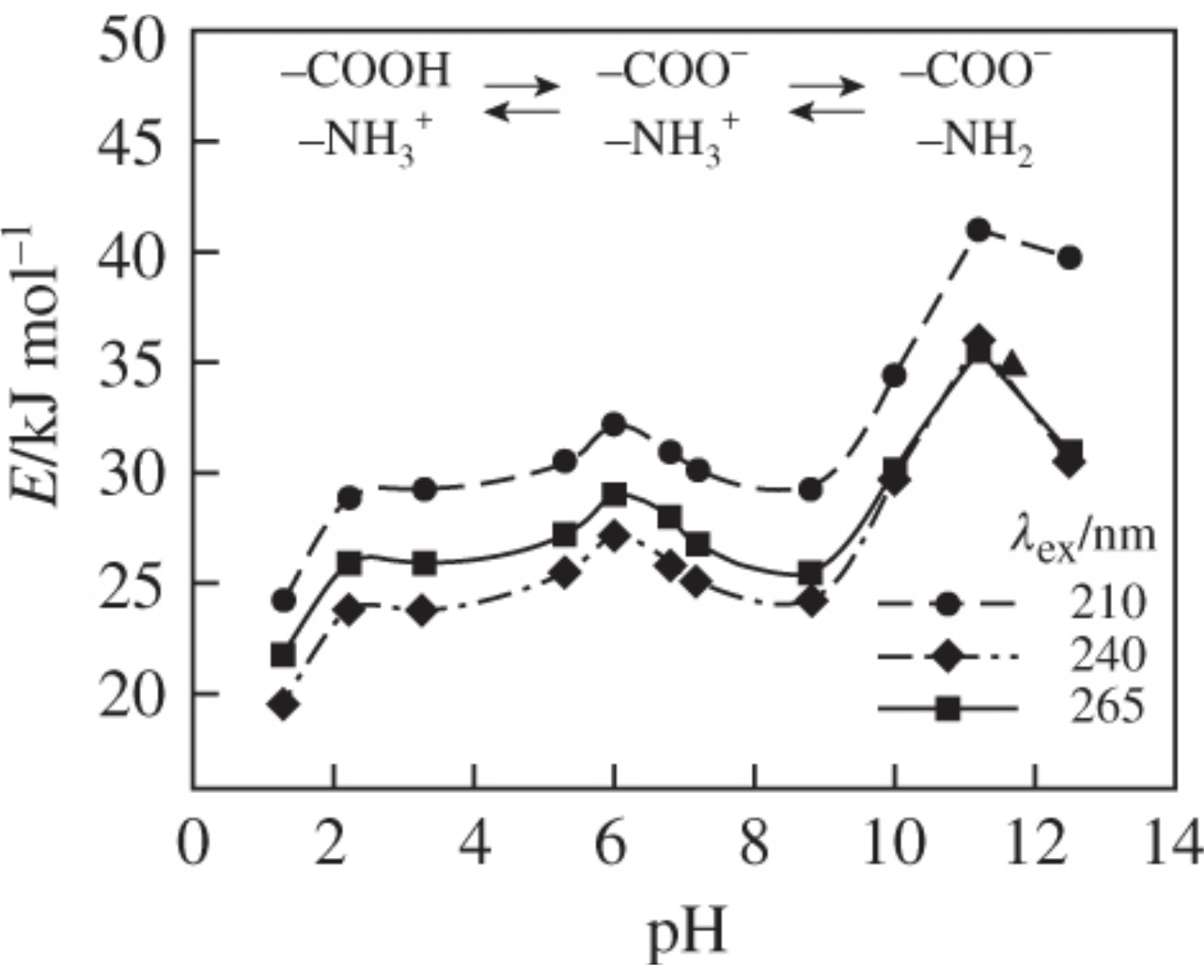
The activation energy of temperature quenching (E) of tryptophan fluorescence at pH 1–13 exhibited a maximum corresponding to the isoelectric point (pHi ≈ 6) of tryptophan at all exciting light wavelengths. The values of E nonmonotonically changed with the excitation wavelength. This behavior was explained by photoinduced electron transfer from the higher singlet-excited Sn (n>1) states of tryptophan.
References
1.
10.1016/j.mencom.2020.01.019_bib0005
Palego
J. Amino Acids,
2016
2.
Ageeva A.A., Babenko S.V., Polyakov N.E., Leshina T.V.
Mendeleev Communications,
2019
3.

Teale F.W., Weber G.
Biochemical Journal,
1957
4.

Tatischeff I., Klein R.
Photochemistry and Photobiology,
1975
5.
10.1016/j.mencom.2020.01.019_bib0025
Agarwal
Textbook of Biochemistry,
2007
6.

White A.
Biochemical Journal,
1959
7.

Robbins R.J., Fleming G.R., Beddard G.S., Robinson G.W., Thistlethwaite P.J., Woolfe G.J.
Journal of the American Chemical Society,
1980
8.
10.1016/j.mencom.2020.01.019_bib0040
Eisinger
Chem. Phys.,
1969
9.

Gally J.A., Edelman G.M.
Biochimica et Biophysica Acta - General Subjects,
1962
10.

Kirby E.P., Steiner R.F.
The Journal of Physical Chemistry,
1970
11.

Weller A.
Pure and Applied Chemistry,
1968
12.

Wawilow S.J.
European Physical Journal A,
1927
13.

Steen H.B.
Journal of Chemical Physics,
1974
14.

Ostakhov S.S., Kazakov V.P., Alyab’ev A.S., Osina I.O.
Doklady Physical Chemistry,
2007
15.

Kazakov V.P., Ostakhov S.S., Alyab’ev A.S., Farrakhova G.G.
High Energy Chemistry,
2005
16.
10.1016/j.mencom.2020.01.019_bib0080
Bowen
J. Chem. Soc.,
1958
17.
10.1016/j.mencom.2020.01.019_bib0085
Jakubke
Aminokisloty, peptidy, belki (Amino Acids, Peptides, Proteins),
1985
18.

Bent D.V., Hayon E.
Journal of the American Chemical Society,
1975
19.

Ostakhov S.S., Kazakov V.P., Osina I.O.
Doklady Physical Chemistry,
2008
20.

Kazakov V.P., Ostakhov S.S., Farrakhova G.G.
High Energy Chemistry,
2008