Keywords
Lewis acids
octamethylcyclotetrasiloxane
polysiloxanes
ring-opening polymerization
trifluoroacetate complexes
Abstract
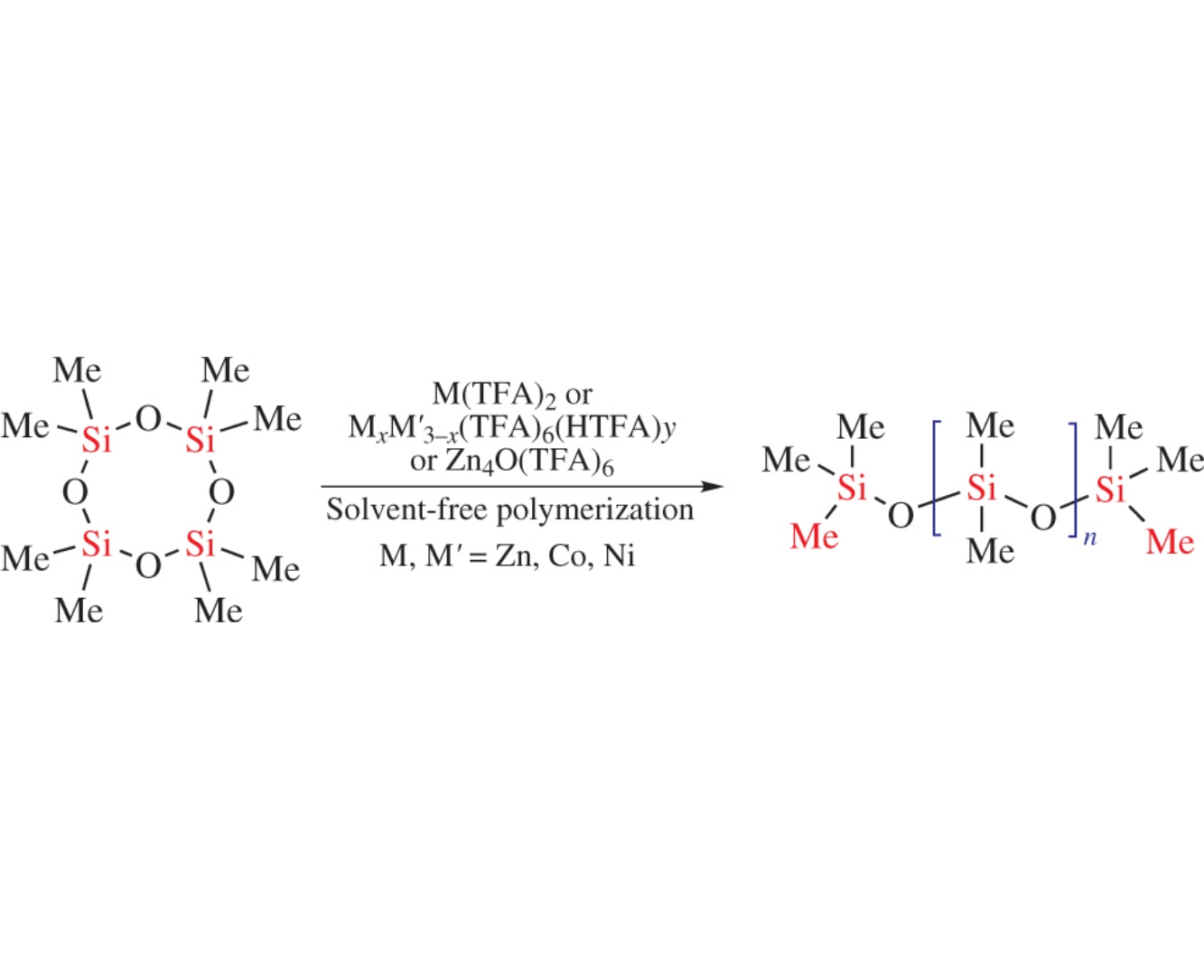
3d-Metal trifluoroacetate complex-catalyzed ring opening polymerization of octamethylcyclotetrasiloxane affords polydimethylsiloxanes whose molecular weight can be controlled by the nature of 3d metal complexes, in particular, by their structural type, and by the polymerization conditions. The best results were achieved using tetranuclear zinc oxotrifluoroacetate as the catalyst at a temperature of 75°C and a reaction time of 35h.
References
1.

2.
10.1016/j.mencom.2020.01.014_bib0010
Moretto
Silicones, in Ullmann's Encyclopedia of Industrial Chemistry,
2005
3.
10.1016/j.mencom.2020.01.014_bib0015
Noll
Chemistry and Technology of Silicone,
1968
4.
10.1016/j.mencom.2020.01.014_bib0020
Concise Encyclopedia of High Performance Silicones,
2014
5.

Kamino B.A., Bender T.P.
Chemical Society Reviews,
2013
6.

Skorotetcky M.S., Borshchev O.V., Surin N.M., Meshkov I.B., Muzafarov A.M., Ponomarenko S.A.
Silicon,
2014
7.

Kirikova M.N., Agina E.V., Bessonov A.A., Sizov A.S., Borshchev O.V., Trul A.A., Muzafarov A.M., Ponomarenko S.A.
Journal of Materials Chemistry C,
2016
8.

Palaganas J., de Leon A.C., Mangadlao J., Palaganas N., Mael A., Lee Y.J., Lai H.Y., Advincula R.
Macromolecular Materials and Engineering,
2017
9.

Wu A.S., Small IV W., Bryson T.M., Cheng E., Metz T.R., Schulze S.E., Duoss E.B., Wilson T.S.
Scientific Reports,
2017
10.

Basu S., Khan A.L., Cano-Odena A., Liu C., Vankelecom I.F.
Chemical Society Reviews,
2010
11.
10.1016/j.mencom.2020.01.014_bib0055
Rao
Sep. Purif. Technol.,
2011
12.
10.1016/j.mencom.2020.01.014_bib0060
Excipient Applications in Formulation Design and Drug Delivery,
2015
13.

Kim B., Hong D., Chang W.V.
Journal of Applied Polymer Science,
2015
14.
10.1016/j.mencom.2020.01.014_bib0070
Mark
The Polysiloxanes,
2015
15.
10.1016/j.mencom.2020.01.014_bib0075
2005
16.
10.1016/j.mencom.2020.01.014_bib0080
Voronkov
Siloxane Bond: Physical Properties and Chemical Transformations,
1978
17.

Embery C.J., Matisons J.G., Clarke S.R.
ACS Symposium Series,
2003
18.
Lukin R.Y., Emelyanov D.A., Kachmarzhik A.D., Sukhov A.V., Sinyashin O.G., Yakhvarov D.G.
Mendeleev Communications,
2019
19.
Vysochinskaya Y.S., Anisimov A.A., Milenin S.A., Korlyukov A.A., Dolgushin F.M., Kononova E.G., Peregudov A.S., Buzin M.I., Shchegolikhina O.I., Muzafarov A.M.
Mendeleev Communications,
2018
20.
Anisimov A.A., Polshchikova N.V., Vysochinskaya Y.S., Zader P.A., Nikiforova G.G., Peregudov A.S., Buzin M.I., Shchegolikhina O.I., Muzafarov A.M.
Mendeleev Communications,
2019
21.

Mojsiewicz-Pieńkowska K., Jamrógiewicz M., Szymkowska K., Krenczkowska D.
Frontiers in Pharmacology,
2016
22.

Yashiro T., Kricheldorf H.R., Schwarz G.
Macromolecular Chemistry and Physics,
2010
23.

Morozov I.V., Karpova E.V., Glazunova T.Y., Boltalin A.I., Zakharov M.A., Tereshchenko D.S., Fedorova A.A., Troyanov S.I.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2016
24.
10.1016/j.mencom.2020.01.014_bib0120
Organosilicon Chemistry V: From Molecules to Materials,
2003