Keywords
arylphosphonates
esters
hydrolysis
mechanism.
phosphonic acids
rate constants
reactivity
Abstract
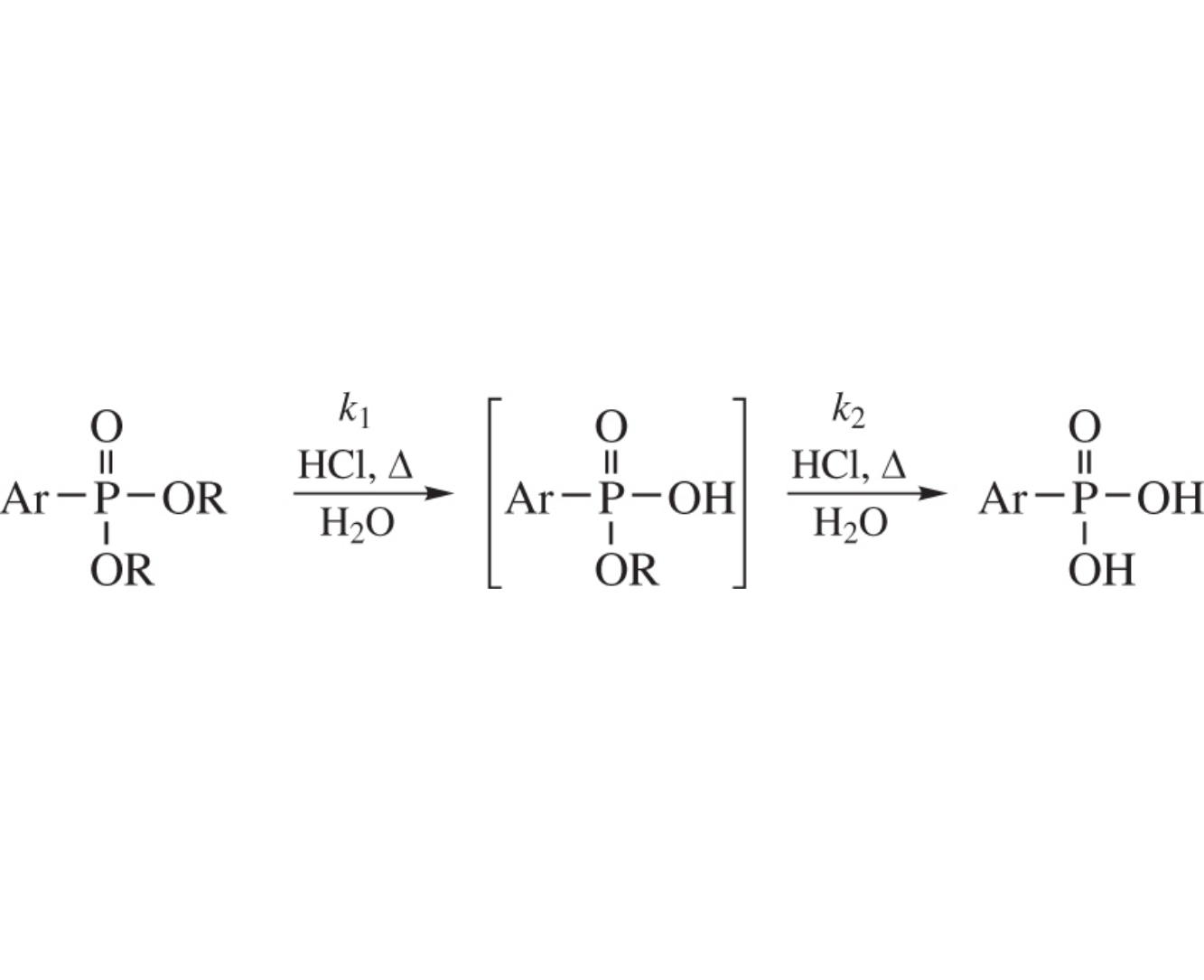
The HCl-catalyzed hydrolysis of dialkyl arylphosphonates monitored by 31P NMR spectroscopy has revealed two consecutive steps characterized by pseudo first order rate constants k1 and k2. A reactivity order for the two steps and for the overall two step hydrolysis has been derived depending on the alkoxy and aryl substituents. Besides the AAc2 mechanism, the AAl1 route has been substantiated for the PriO substituent.
References
1.
10.1016/j.mencom.2020.01.012_bib0005
Kosolapoff
1973
2.
10.1016/j.mencom.2020.01.012_bib0010
Methoden der organischen Chemie (Houben-Weyl), Band E2, Phosphor- Verbindungen II,
1982
3.

Desai J., Wang Y., Wang K., Malwal S.R., Oldfield E.
ChemMedChem,
2016
4.

Tcarkova K.V., Artyushin O.I., Bondarenko N.A.
Phosphorus, Sulfur and Silicon and the Related Elements,
2016
5.

Gavande N., Yamamoto I., Salam N.K., Ai T., Burden P.M., Johnston G.A., Hanrahan J.R., Chebib M.
ACS Medicinal Chemistry Letters,
2010
6.

Haake P., Hurst G.
Journal of the American Chemical Society,
1966
7.

Keglevich G., Grün A., Bölcskei A., Drahos L., Kraszni M., Balogh G.T.
Heteroatom Chemistry,
2012
8.

Rahil J., Haake P.
Journal of Organic Chemistry,
1981
9.
![1-Oxo-2-oxa-1-phosphabicyclo[2.2.2]octane: A New Mechanistic Probe for the Basic Hydrolysis of Phosphate Esters](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Wróblewski A.E., Verkade J.G.
Journal of the American Chemical Society,
1996
10.

Ray R., Boucher L.J., Broomfield C.A., Lenz D.E.
Biochimica et Biophysica Acta - General Subjects,
1988
11.
10.1016/j.mencom.2020.01.012_bib0055
Salomon
Tetrahedron Lett.,
1995
12.

Jansa P., Hradil O., Baszczyňski O., Dračínský M., Klepetářová B., Holý A., Balzarini J., Janeba Z.
Tetrahedron,
2012
13.

Keglevich G., Rádai Z., Harsági N., Szigetvári Á., Kiss N.Z.
Heteroatom Chemistry,
2017
14.

Henyecz R., Kiss A., Mórocz V., Kiss N.Z., Keglevich G.
Synthetic Communications,
2019