Keywords
arylacetylenes
cascade reactions
dioxaphospholes
ethenylphosphonic acids
oxaphosphinines
phosphoranes
Abstract
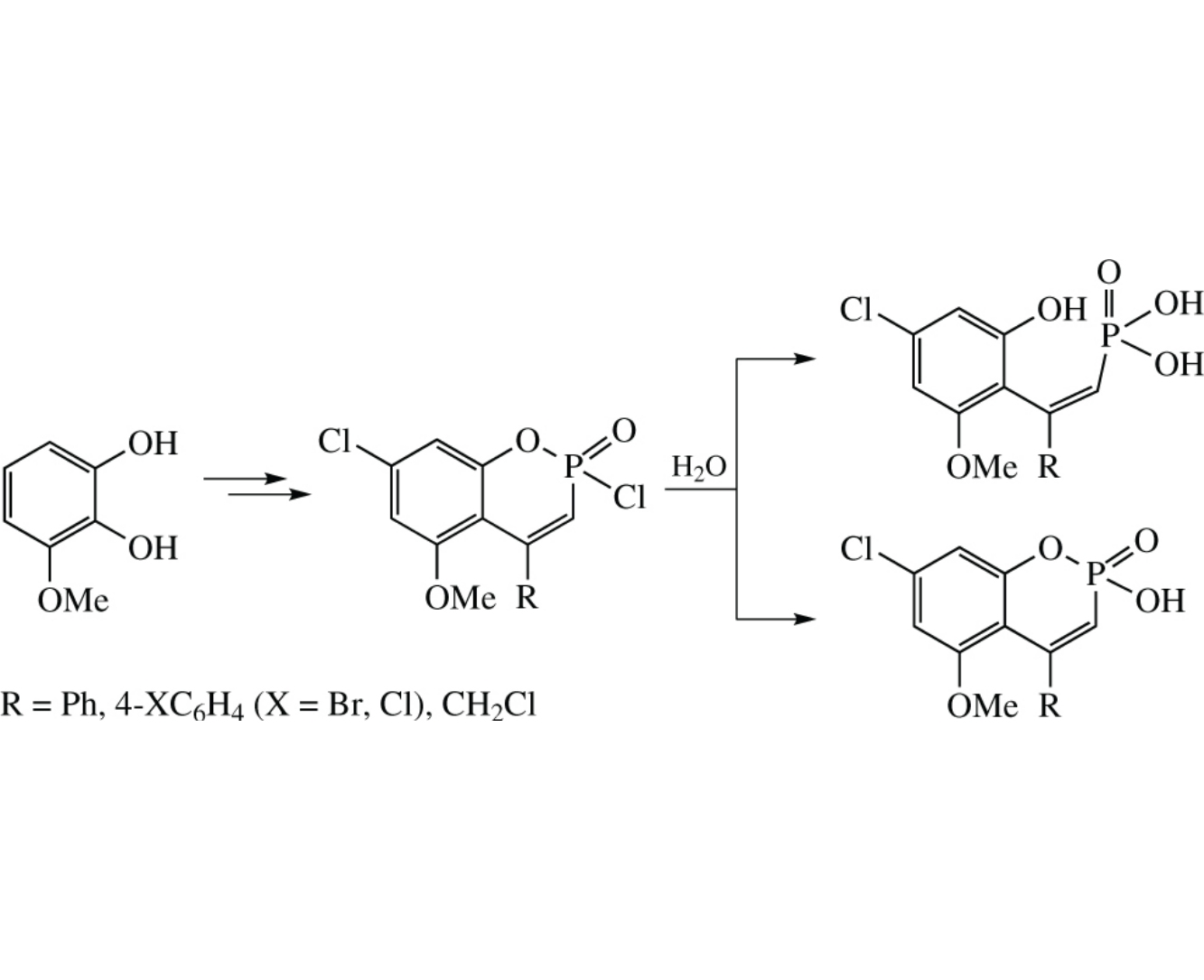
2,2,2-Trichloro-4-methoxy-1,3,2-benzodioxaphosphole reacts with arylacetylenes or 3-chloropropyne to give 2,7-dichloro-5-methoxy-4-aryl(haloalkyl)-1,2-benzoxaphosphinine 2-oxides. Hydrolysis of the latter leads to the opening of the oxaphosphinine ring and formation of (E)-2-(4-chloro-2- methoxy-6 hydroxyphenyl)ethenylphosphonic acid.
References
1.
10.1016/j.mencom.2020.01.011_sbref0005a
The Chemistry of Phenols,
2003
2.

Crawford J.
Progress in Polymer Science,
1999
3.

Das P.K., DesLauriers P.J., Fahey D.R., Wood F.K., Cornforth F.J.
Polymer Degradation and Stability,
1995
4.
10.1016/j.mencom.2020.01.011_sbref0005d
Zweifel
Plastics Additives Handbook,
2009
5.

Vulic I., Vitarelli G., Zenner J.M.
Polymer Degradation and Stability,
2002
6.

Pospíšil J.
Polymer Degradation and Stability,
1988
7.

Kuimov V.A., Malysheva S.F., Artem’ev A.V., Kenzhetaeva S.O., Belogorlova N.A., Komarova T.N., Gusarova N.K., Trofimov B.A.
Russian Journal of Organic Chemistry,
2014
8.
10.1016/j.mencom.2020.01.011_sbref0010a
Gardziella
Phenolic Resins: Chemistry, Applications, Standardization, Safety and Ecology,
2000
9.
10.1016/j.mencom.2020.01.011_sbref0010b
Phenolic Resins: A Century of Progress,
2010
10.
10.1016/j.mencom.2020.01.011_sbref0010c
Knop
Chemistry and Application of Phenolic Resins,
1979
11.

Hirano K., Asami M.
Reactive and Functional Polymers,
2013
12.
10.1016/j.mencom.2020.01.011_bib0015
Phenolic Compounds in Food and Their Effects on Health, I ACS Symposium Series,
1992
13.
10.1016/j.mencom.2020.01.011_bib0020
Phenolic Compounds – Natural Sources, Importance and Applications,
2017
14.
10.1016/j.mencom.2020.01.011_sbref0025a
Corbridge
Phosphorus: Chemistry, Biochemistry and Technology,
2013
15.
10.1016/j.mencom.2020.01.011_sbref0025b
Phosphorus-Carbon Heterocyclic Chemistry: The Rise of a New Domain,
2001
16.
10.1016/j.mencom.2020.01.011_sbref0025c
Mann
Heterocyclic Derivatives of Phosphorus, Arsenic, Antimony and Bismuth,
1970
17.

Gloede J.
Zeitschrift für Chemie,
2010
18.
10.1016/j.mencom.2020.01.011_sbref0025e
1,4-Cycloaddition Reaction: The Diels–Alder Reaction in Heterocyclic Syntheses,
1967
19.

Mironov V.F., Nemtarev A.V.
Review Journal of Chemistry,
2011
20.

Nasibullin I.O., Nemtarev A.V., Mironov V.F.
Russian Journal of General Chemistry,
2014
21.

Nasibullin I.O., Nemtarev A.V., Mironov V.F.
Russian Journal of Organic Chemistry,
2014
22.

Nemtarev A.V., Nasibullin I.O., Mironov V.F., Cherkasov V.K.
Phosphorus, Sulfur and Silicon and the Related Elements,
2015
23.

Nasibullin I.O., Nemtarev A.V., Cherkasov V.K., Mironov V.F.
Russian Chemical Bulletin,
2015
24.

Nemtarev A.V., Mironov V.F., Aniskin A.S., Baranov D.S., Mironova E.V., Krivolapov D.B., Musin R.Z., Vasilevskii S.F., Druzhkov N.O., Cherkasov V.K.
Russian Chemical Bulletin,
2013
25.

Nemtarev A.V., Varaksina E.N., Mironov V.F., Musin R.Z., Konovalov A.I.
Russian Journal of Organic Chemistry,
2007
26.

Nemtarev A.V., Varaksina E.N., Mironov V.F., Musin R.Z., Konovalov A.I.
Russian Journal of Organic Chemistry,
2007
27.

Nemtarev A.V., Mironov V.F., Varaksina E.N., Gubaidullin A.T., Krivolapov D.B., Musin R.Z., Litvinov I.A.
Russian Chemical Bulletin,
2014
28.
Nemtarev A.V., Varaksina E.N., Mironov V.F., Musin R.Z., Konovalov A.I.
Mendeleev Communications,
2006
29.

Garrigues B., Munoz A., Koenig M., Sanchez M., Wolf R.
Tetrahedron,
1977
30.

Cong C.B., Gence G., Garrigues B., Koenig M., Munoz A.
Tetrahedron,
1979
31.
10.1016/j.mencom.2020.01.011_bib0055
Kuhl
Phosphorus-31 NMR Spectroscopy,
2008
32.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
33.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015