Keywords
azepine–pyrazine recyclization
baryl(heteroaryl)ethylamines
bioactive amines
fused dihydroazepines
ring opening reactions
Abstract
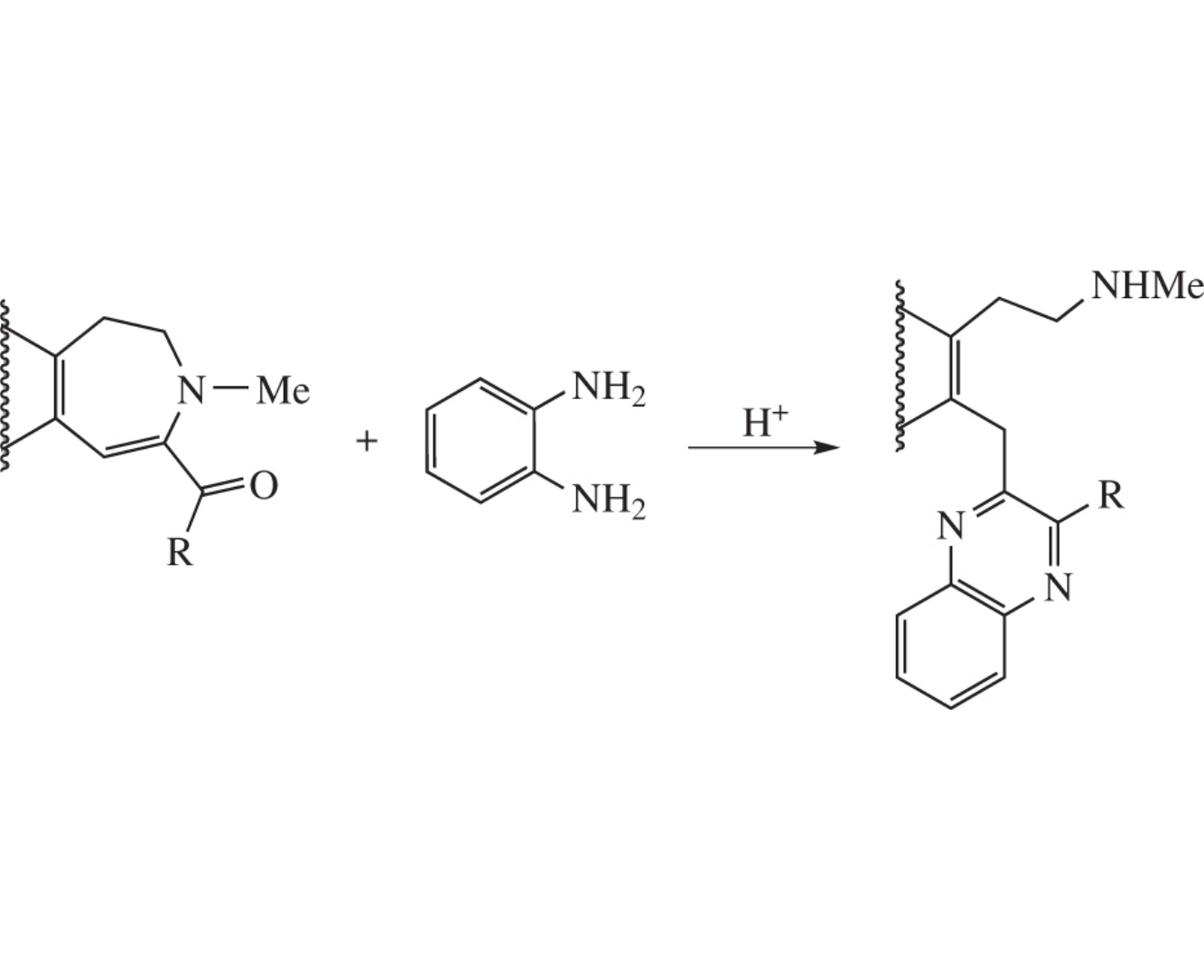
Nucleophilic heterocyclic ring opening in fused 7-acyl- H+ 1,2-dihydroazepines with o-phenylenediamine affords b-(hetero)arylethylamines.
References
1.

The Potential of Trace Amines and Their Receptors for Treating Neurological and Psychiatric Diseases
Berry M.
Reviews on Recent Clinical Trials,
2007
2.

Avery M.C., Krichmar J.L.
Frontiers in Neural Circuits,
2017
3.

Gainetdinov R.R., Hoener M.C., Berry M.D.
Pharmacological Reviews,
2018
4.
10.1016/j.mencom.2020.01.009_bib0020
Irsfeld
Webmedcentral,
2013
5.
10.1016/j.mencom.2020.01.009_bib0025
Melatonin and Melatonergic Drugs in Clinical Practice,
2014
6.
10.1016/j.mencom.2020.01.009_bib0030
Shulgin
2011
7.
10.1016/j.mencom.2020.01.009_bib0035
Wishart
Nucleic Acids Res.,
2017
8.

Smith J.R., Norman R.O., Rose M.E., Curran A.C.
Journal of the Chemical Society Perkin Transactions 1,
1979
9.
![A novel synthesis of benzo- and indolo[a]quinolizidines by intramolecular diels-alder reaction of l-azadienes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ihara M., Kirihara T., Kawaguchi A., Fukumoto K., Kametani T.
Tetrahedron Letters,
1984
10.

Venkov A.P., Statkova-Abeghe S.
Synthetic Communications,
1996
11.
10.1016/j.mencom.2020.01.009_bib0055
Clark
J. Chem. Soc., Perkin Trans.,
2001
12.

Chen F., Zhang Y., Yu L., Zhu S.
Angewandte Chemie - International Edition,
2017
13.

Kartsev V.G., Zubenko A.A., Morkovnik A.S., Divaeva L.N.
Tetrahedron Letters,
2015
14.

Zubenko A.A., Kartsev V.G., Morkovnik A.S., Divaeva L.N., Suponitsky K.Y.
ChemistrySelect,
2016
15.

Zubenko A.A., Divaeva L.N., Morkovnik A.S., Kartsev V.G., Drobin Y.D., Serbinovskaya N.M., Fetisov L.N., Bodryakov A.N., Bodryakova M.A., Lyashenko L.A.
Russian Journal of Bioorganic Chemistry,
2017
16.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018
17.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Suponitsky K.Y., Klimenko A.I.
Mendeleev Communications,
2018
18.
J. E. Pero, H.D. G. F. Lehman, M.E. Layton, M.A. Rossi and M. J. Kelly, III, Patent WO 2014066491A1, 2014.
19.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Kuzmina L.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018
20.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
21.

Welsch M.E., Snyder S.A., Stockwell B.R.
Current Opinion in Chemical Biology,
2010
22.

Watanabe K., Oguri H., Oikawa H.
Current Opinion in Chemical Biology,
2009
23.

Lee J.S., Waring M.J.
Biochemical Journal,
1978