Keywords
alkynes
Annulation
nucleophilic addition
phenanthridine
zwitterions
Abstract
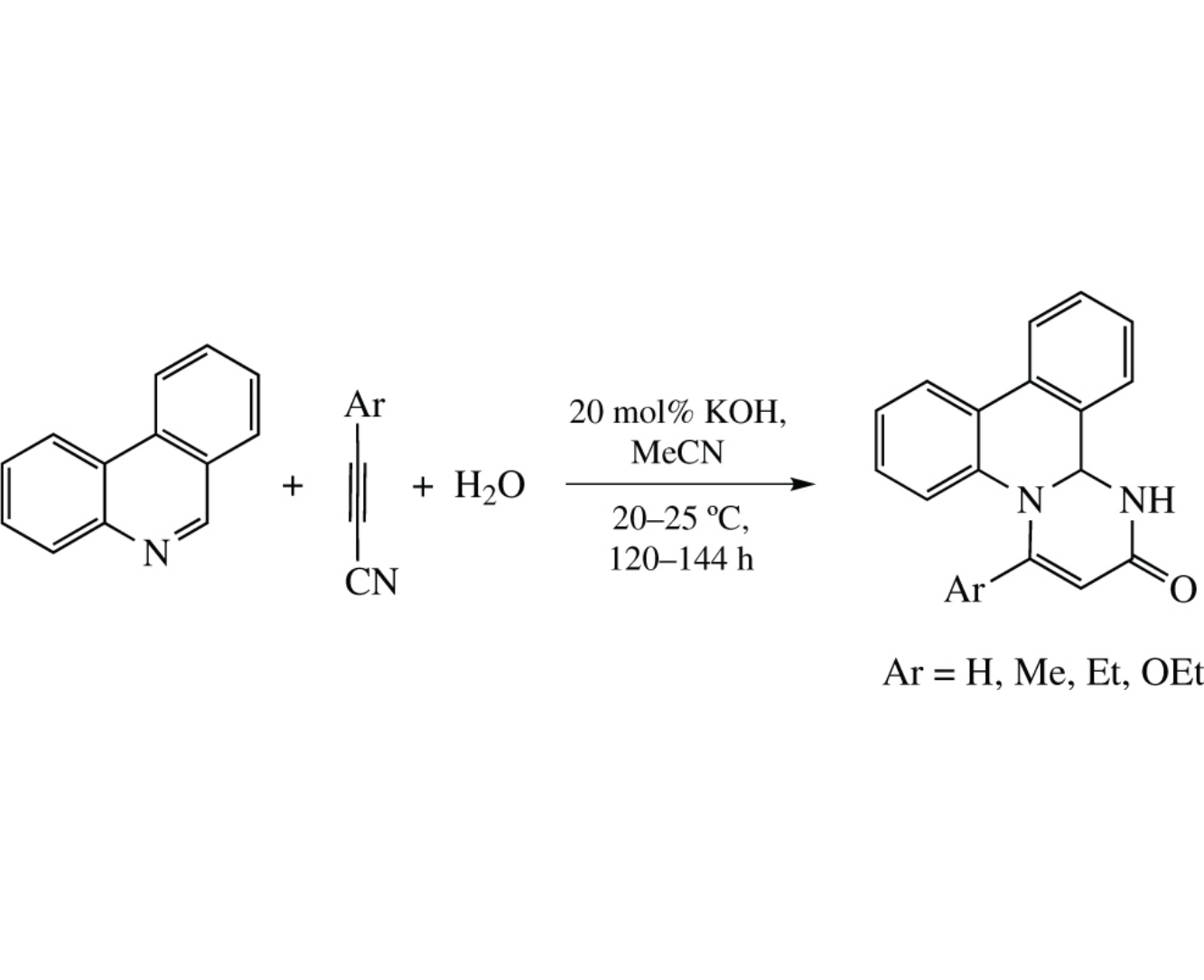
3-Arylpropynenitriles are readily annulated with phenanthridine in the KOH/H2O/MeCN system at room temperature to afford 4-aryl-1,13b dihydropyrimido[1,2-f]phenanthridin-2-ones. The moderate yield of the products can be rationalized by the anionic oligomerization of an intermediate zwitterion adduct of the reactants. The reaction represents a single-stage access to a new family of pharmaceutically prospective compounds.
References
1.

Tumir L., Radić Stojković M., Piantanida I.
Beilstein Journal of Organic Chemistry,
2014
2.

Tang W., Hemm I., Bertram B.
Planta Medica,
2003
3.

Abdel-Halim O.B., Morikawa T., Ando S., Matsuda H., Yoshikawa M.
Journal of Natural Products,
2004
4.

Matsumoto K., Choshi T., Hourai M., Zamami Y., Sasaki K., Abe T., Ishikura M., Hatae N., Iwamura T., Tohyama S., Nobuhiro J., Hibino S.
Bioorganic and Medicinal Chemistry Letters,
2012
5.

Lyakhova E.G., Kolesnikova S.A., Kalinovsky A.I., Afiyatullov S.S., Dyshlovoy S.A., Krasokhin V.B., Minh C.V., Stonik V.A.
Tetrahedron Letters,
2012
6.
![Phenanthridine synthesis via [2+2+2] cyclotrimerization reactions](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Sripada L., Teske J.A., Deiters A.
Organic and Biomolecular Chemistry,
2008
7.
![A Two-Step Synthesis of Cytostatically Active Benzo[c]phenanthridine Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Clement B., Weide M., Wolschendorf U., Kock I.
Angewandte Chemie - International Edition,
2005
8.
10.1016/j.mencom.2020.01.004_bib0040
Zupko
In Vivo,
2009
9.

Baumann M., Baxendale I.R.
Beilstein Journal of Organic Chemistry,
2013
10.

Flores Toque H.A., Priviero F.B., Teixeira C.E., Perissutti E., Fiorino F., Severino B., Frecentese F., Lorenzetti R., Baracat J.S., Santagada V., Caliendo G., Antunes E., De Nucci G.
Journal of Medicinal Chemistry,
2008
11.
Pharmaceutical Substances: Syntheses, Patents, Applications, eds. A. Kleemann, J. Engel, B. Kutscher and D. Reicher, Thieme, Stuttgart, 2001, vol. 1.
12.

Sharif E.U., O'Doherty G.A.
European Journal of Organic Chemistry,
2012
13.

Bevk D., Teresa Gand�egui M., Alvarez-Builla J., Florencio F.
Heterocycles,
1994
14.

Singh D., Sinha P., Bansal R.
Current Green Chemistry,
2014
15.
![Efficient Synthesis of Pyrrolo[1,2-f]phenanthridine Derivatives via Dipolar Cycloaddition of Phenanthridine, Activated Acetylenes, and Ethyl Bromopyruvate](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mehrabi H., Pishahang J.
Synthetic Communications,
2013
16.
![Synthesis of Functionalized Pyrido[1,2-f]phenanthridines from Phenanthridine, Activated Acetylenes, and Arylidenemalononitriles](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mehrabi H., Hatami-pour M.
Synthetic Communications,
2012
17.

Teimouri M.B., Abbasi T., Ahmadian S., Poor Heravi M.R., Bazhrang R.
Tetrahedron,
2009
18.
![Efficient Synthesis of [1,3]Oxazino[3,2-f]phenanthridine Derivatives by a Novel 1,4-Dipolar Cycloaddition Involving a Phenanthridine-Dimethyl Acetylenedicarboxylate Zwitterion and Aromatic Aldehydes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
19.

Mehrabi H., Hatami-Pour M.
Chinese Chemical Letters,
2014
20.
![Synthesis of Fused [1,3]Oxazines from Ethyl Trifluoroacetate, Activated Acetylenes and N-Heterocycles](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Mohammadi M., Khandan-Barani K., Hassanabadi A.
Journal of Chemical Research,
2017
21.

Andriyankova L.V., Mal'kina A.G., Nikitina L.P., Belyaeva K.V., Ushakov I.A., Afonin A.V., Nikitin M.V., Trofimov B.A.
Tetrahedron,
2005
22.

Skvortsov Y.M., Mal'kina A.G., Volkov A.N., Trofimov B.A., Oleinikova E.B., Kazin I.V., Gedymin V.V.
Russian Chemical Bulletin,
1978
23.

Liu P., Clark R.J., Zhu L.
Journal of Organic Chemistry,
2018
24.

Belyaeva K.V., Nikitina L.P., Afonin A.V., Vashchenko A.V., Trofimov B.A.
Russian Chemical Bulletin,
2017
25.
![Cyanoacetylenes as Triggers and Partners in KOH-Assisted Assemblies of Quinoline-Based Dihydropyrimido[1,2-a]quinolin-3-ones on Water](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Belyaeva K.V., Nikitina L.P., Mal’kina A.G., Afonin A.V., Vashchenko A.V., Trofimov B.A.
Journal of Organic Chemistry,
2019
26.
Mal’kina A.G., Shemyakina O.A., Nosyreva V.V., Albanov A.I., Klyba L.V., Zhanchipova E.R., Trofimov B.A.
Mendeleev Communications,
2006
27.

Trofimov B.A., Andriyankova L.V., Mal'kina A.G., Belyaeva K.V., Nikitina L.P., Dyachenko O.A., Kazheva O.N., Chekhlov A.N., Shilov G.V., Afonin A.V., Ushakov I.A., Baikalova L.V.
European Journal of Organic Chemistry,
2007
28.

A one-pot, water compatible synthesis of pyrimidine nucleobases under plausible prebiotic conditions
Okamura H., Becker S., Tiede N., Wiedemann S., Feldmann J., Carell T.
Chemical Communications,
2019
29.
Trofimov B.A., Andriyankova L.V., Belyaeva K.V., Mal’kina A.G., Nikitina L.P., Afonin A.V., Ushakov I.A.
Mendeleev Communications,
2009